Abstract
Background:
Rotator cuff (RC) tendinopathy is one of the most common causes of shoulder pain. Platelet-rich plasma (PRP) has been frequently used in clinical scenarios, but its efficacy remains inconsistent.
Purpose:
To investigate the different responses of human tenocytes from torn RCs to leukocyte-rich PRP (LR-PRP) and leukocyte-poor PRP (LP-PRP) in a 2-chamber coculture device.
Study Design:
Controlled laboratory study.
Methods:
PRP was prepared using different platelet and leukocyte concentrations according to 5 groups: (1) LR-PRP with 5000 platelets/µL, (2) LR-PRP with 10,000 platelets/µL, (3) LP-PRP with 5000 platelets/µL, (4) LP-PRP with 10,000 platelets/µL, and (5) control with only culture medium supplementation and without PRP stimulation. Platelet-derived growth factor–AB (PDGF-AB) and transforming growth factor–β1 (TGF-β1) were measured in LR-PRP and LP-PRP via enzyme-linked immunosorbent assay. Microscopy, water-soluble tetrazolium salt assay, and quantitative real-time polymerase chain reaction were used to investigate the morphology, proliferation, and gene expression of RC tenocytes exposed to different PRP formulations. Data were collected from at least 3 independent measurements. The results were analyzed via 1-way analysis of variance, followed by the post hoc Bonferroni test.
Results:
The ratio of leukocytes to 5000 platelets/µL was 29.5 times higher in LR-PRP than in LP-PRP (P < .05). In the 5000 platelets/µL groups, the levels of TGF-β1 and PDGF-AB were both significantly higher in LR-PRP versus LP-PRP (TGF-β1: 367.0 ± 16.5 vs 308.6 ± 30.3 pg/mL, respectively [P = .043]; PDGF-AB: 172.1 ± 1.8 vs 94.1 ± 4.2 pg/mL, respectively [P < .001]). Compared with the control group, RC tenocyte proliferation was 1.42 ± 0.01 and 1.41 ± 0.03 times higher in the LR-PRP groups with 5000 platelets/µL and 10,000 platelets/µL, respectively (P < .05). The expression of tenocyte-related genes was higher in tenocytes cultured in LR-PRP.
Conclusion:
Both the LR-PRP groups with 5000 platelets/µL and 10,000 platelets/µL induced more growth factor release and increased RC tenocyte proliferation than did the LP-PRP groups.
Clinical Relevance:
In RC repair, LR-PRP may be better than LP-PRP for increasing the proliferation of tenocytes.
A rotator cuff (RC) tear is a common cause of persistent shoulder pain. 24 Even with surgical interventions, studies have revealed the high incidence of recurrent tendon defects after RC repair. 32 One important reason for poor healing and high retear rates is the minimal blood supply to the repair construct. 31 Tenocytes are the basic cellular components of tendon tissue and contribute to synthesize all components of the extracellular matrix for the intrinsic repair process. 26 A low cell density in tendon tissue and a minimal blood supply lead to limited extracellular matrix restoration for tendon healing. 3
Platelet-rich plasma (PRP) has been an attractive approach for stimulating and accelerating the soft tissue healing process. 11 PRP with a 3 to 5 times higher platelet concentration compared with whole blood is frequently used in a clinical scenario, which releases more growth factors and cytokines than whole blood. 5 Platelet-derived growth factors (PDGFs), including PDGF-AB and transforming growth factor–β1 (TGF-β1), have recently shown promise in promoting tendon repair and regeneration. 2,36
In the past decade, a number of in vitro studies have reported that PRP enhances the proliferation of human muscle, bone, and tendon cells. 2,16,22 In contrast, other studies have revealed no statistically significant benefit from PRP treatment. 30,35 Currently, the efficacy of PRP therapy is still unclear and remains controversial among medical experts, especially when PRP is used in conjunction with RC repair. This might be attributed to the large variations in the preparation procedure and composition of the applied PRP. Some authors have suggested that red blood cells (RBCs) and white blood cells (WBCs) may be detrimental, as these cells participate in inflammatory reactions that can lead to damage in the treated tissue. 10,23 However, nonplatelet cellular components in platelet concentrates are important for proper platelet function. 27 The lack of RBCs and leukocytes results in a deficiency in platelet function concerning growth factor release and the ability of the releasate to stimulate tenocyte proliferation. 27 A recent study showed better effects with a leukocyte-rich PRP (LR-PRP) injection over a corticosteroid injection for chronic gluteal tendinopathy, 12 while another animal study revealed that leukocyte-poor PRP (LP-PRP) promoted tendon healing in chronic Achilles tendinopathy than LR-PRP did. 38 PRP injections for RC tears are reported in less high-quality studies, but a randomized controlled trial found that there was an improvement in pain with 2 injections of LR-PRP, separated by 4 weeks, compared with a placebo injection. 29 More studies are needed to identify the best preparation and composition of PRP for different clinical conditions, 13 especially for RC tears.
In this study, we used a 2-chamber coculture device to investigate the responses of human tenocytes from torn RC tissue to LR-PRP and LP-PRP formulations. The purpose of this study was to evaluate tenocyte proliferation and growth factor release via LR-PRP and LP-PRP treatment. We hypothesized that different clinical scenarios might have their own best PRP compositions for treatment and that LR-PRP would be more effective in RC tenocyte proliferation and growth factor release.
Methods
Isolation of Tenocytes
After obtaining institutional review board approval for this study, we isolated tendon tissue from the edge of torn human supraspinatus tendons during arthroscopic RC repair of 3 patients with moderate degenerative RC tears. All patients had informed consent. All patients were male, with ages ranging from 49 to 65 years (Table 1). None of the patients had diabetes, smoking, systemic illness, recent histoy of cancer, and undergone any form of anticoagulant, antibacterial, or immunosuppressive therapy within the previous 6 months.
Descriptive Data of Patients

We used a previously published protocol to isolate tenocytes. 28 After the first passage, adherent cells were trypsinized, and cells were seeded at 2 × 104 cells/cm2 in the coculture device. Normal tenocyte morphological characteristics were confirmed via microscopy. The cell number was determined using an automated cell counter (Countess II FL; Thermo Fisher Scientific). Tenocytes above 3 passages were discarded because a phenotypic drift was reported. 21 The tenocyte genotype was confirmed using quantitative real-time polymerase chain reaction (qRT-PCR) for the tenocyte markers, including type I collagen (COL1), type III collagen (COL3), decorin (DCN), tenascin-C (TNC), and scleraxis (SCX).
LR-PRP and LP-PRP Preparation
PRP was prepared with different platelet and leukocyte concentrations according to 4 PRP groups plus a control group: (1) LR-PRP with 5000 platelets/µL, (2) LR-PRP with 10,000 platelets/µL, (3) LP-PRP with 5000 platelets/µL, (4) LP-PRP with 10,000 platelets/µL, and (5) control with only culture medium supplementation and without PRP stimulation. We withdrew 20 mL of autologous blood from each patient during RC repair. For the LR-PRP preparation, 5 mL of whole blood was initially subjected to centrifugation at 200g for 10 minutes to pellet the RBCs, which were discarded. The supernatant was then subjected to a second centrifugation at 600g for 8 minutes. Upper plasma was aspirated, and 0.3 mL of plasma was used to resuspend the resultant cell pellet to generate LR-PRP. 38 For the preparation of LP-PRP, after the first centrifugation at 200g, the second centrifugation at 100g was applied to discard the WBCs and residual RBCs. Subsequently, a third centrifugation at 600g was applied to enrich the platelets, and 0.3 mL of LP-PRP was obtained via resuspension of the platelets in plasma. 38 The procedure is summarized in Figure 1. The platelet and WBC counts in whole blood and PRP of the 3 patients are listed in Table 2. The platelet count was determined via a cell counter first and diluted to concentrations of 5000 and 10,000 platelets/µL accordingly.

Schematic diagram of the preparation of (A) leukocyte-rich platelet-rich plasma (LR-PRP) and (B) leukocyte-poor platelet-rich plasma (LP-PRP).
No. of Platelets and WBCs in Whole Blood and PRP of Patients a

a LP-PRP, leukocyte-poor platelet-rich plasma; LR-PRP, leukocyte-rich platelet-rich plasma; PRP, plate-rich plasma; WBC, white blood cell.
b Per 103 cells/μL.
Stimulation of Tenocytes via PRP
PRP and tenocytes were obtained from each patient during surgery and were then combined. Cells at a density of 1.5 × 104 cells/cm2 suspended in 120-µL culture medium were respectively applied to 1 of the 2 subchambers (cell chamber) of the coculture device (Figure 2). Then, PRP that contained 5000 platelets/µL and 10,000 platelets/µL in liquid form was applied to the remaining subchamber (PRP chamber). It is noted that the level of the solution was not added over the height of the barrier. Then, the coculture device was placed in an incubator for an hour. After 1 hour, cells attached to the surface of the cell chamber, and PRP became a gel-like form in the PRP chamber. Another 500-µL culture medium was applied to the PRP chamber to overcome the polydimethylsiloxane barrier, causing an exchange of culture medium between the cell chamber and PRP chamber as previously described. 4 Hence, tenocytes cultured in the cell chamber were able to receive platelet-derived growth factors via diffusion. Finally, cells were cultured in the incubator for 3 days. After the culture course, cell proliferation was investigated using bioassay. The procedure is summarized in Figure 3.

Photograph of the coculture device.

Design and experimental setup to investigate the proliferation of tenocytes under the stimulation of leukocyte-rich platelet-rich plasma (LR-PRP) and leukocyte-poor platelet-rich plasma (LP-PRP). WST-1, water-soluble tetrazolium salt.
Quantification of Cells
Briefly, the cell number was quantified using water-soluble tetrazolium salt (WST-1) assay (Roche) as previously described. 6
Gene Analysis by qRT-PCR
RNA was isolated from cultured dishes as previously described. 21 RNA quantity and purity (A260/280) were measured using μQuant software (BioTek Instruments). RNA was reverse-transcribed into complementary DNA (cDNA) using 1 µg of mRNA and a High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific). qRT-PCR was performed using 10 to 100 ng of cDNA as a template and the StepOne Real-Time PCR System (Thermo Fisher Scientific). The resultant cycle threshold values were normalized and analyzed using the standard curve method. TaqMan Gene Expression Assays (Thermo Fisher Scientific) were used for the following genes: COL1, COL3, TNC, DCN, SCX, and transcription factor SRY-box transcription factor–9 (SOX9), relative to glyceraldehyde 3-phosphate dehydrogenase as the endogenous control.
Evaluation of Growth Factors in Coculture Device
TGF-β1 and PDGF-AB were analyzed by using a commercial immunoassay kit (Human TGF-β1 and Human PDGF-AB Quantikine ELISA Kit; R&D Systems). The analytical protocol followed the manufacturer’s instructions. The supernatant was pipetted in a kit-supplied microplate that was precoated with the primary antibody and incubated for 3 hours at room temperature. Then, the microplate was washed using the provided buffer. Next, the detection antibody was added and incubated for 1.5 hours at room temperature. After the microplate was washed , the substrate solution was added and incubated for 20 minutes. The optical density of the reactive solution was quantified using a microplate reader (ELx800; BioTek Instruments). In addition, a serial dilution of the provided standard TGF-β1 and PDGF-AB solution was utilized to set up the calibration curve. Thus, the actual concentrations of TGF-β1 and PDGF-AB could be calculated based on the calibration curve.
Statistical Analysis
Data (presented as mean ± SD) for the 4 PRP groups were collected from at least 3 independent measurements. The LR-PRP and LP-PRP groups were compared within the different platelet concentrations (5000 and 10,000 platelets/μL) as well as with the control group using 1-way analysis of variance, followed by the post hoc Bonferroni test. Statistical significance was indicated at P < .05. All statistical analyses were performed using SPSS 21.0 for Windows (IBM Corp).
Results
Different PRP Analyses
The concentrations of platelets and leukocytes were analyzed to confirm the successful preparation of LR-PRP (platelet count, 1190.7 ± 773.6 × 103 platelets/μL) and LP-PRP (platelet count, 548.7 ± 392.9 × 103 platelets/μL) compared to whole blood (platelet count, 183.7 ± 80.0 × 103 platelets/μL) (Figure 4A and Table 2). Although the concentrations of leukocytes and platelets between LR-PRP (leukocyte count, 4.7 ± 3.8 × 103 leukocytes/μL) and LP-PRP (leukocyte count, 0.9 ± 3.8 × 103 leukocytes/μL) were not significantly different (P = .17 and .51, respectively), the ratio of leukocytes to 5000 platelets/µL was 29.5 times higher in LR-PRP versus LP-PRP (P < .05) (Figure 4, B and C).
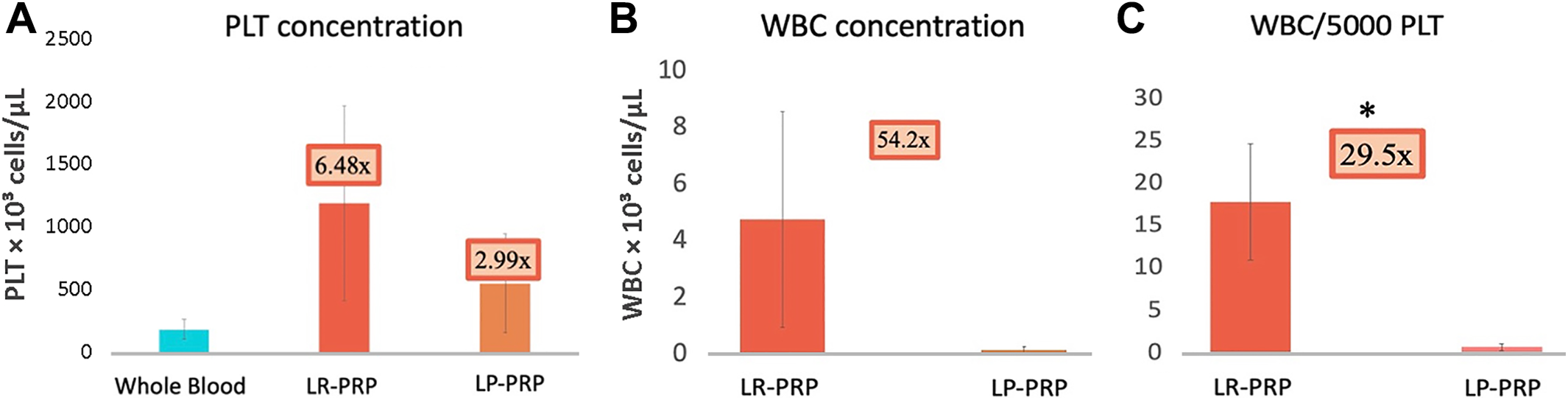
Compositions of leukocyte-rich platelet-rich plasma (LR-PRP) and leukocyte-poor platelet-rich plasma (LP-PRP) prepared from human whole blood. (A) The platelet concentration in LR-PRP was about 2 times higher than that in LP-PRP (P = .51). (B) The leukocyte concentration in LR-PRP was about 5 times higher than that in LP-PRP (P = .17). (C) The ratio of leukocytes to 5000 platelets/µL was 29.5 times higher in LR-PRP compared with LP-PRP (P < .05). PLT, platelet; WBC, white blood cell. * P < 0.05.
Growth Factor Levels in LR-PRP and LP-PRP
In the 5000 platelets/µL groups, the concentration of TGF-β1 in LR-PRP was 1.2 times higher than in LP-PRP (367.0 ± 16.5 vs 308.6 ± 30.3 pg/mL; P = .043), while PDGF-AB was 1.8 times higher in LR-PRP than in LP-PRP (172.1 ± 1.8 vs 94.1 ± 4.2 pg/mL; P < .001) (Figure 5).

Growth factor levels in leukocyte-rich platelet-rich plasma (LR-PRP) with 5000 platelets/µL and leukocyte-poor platelet-rich plasma (LP-PRP) with 5000 platelets/µL prepared from human whole blood. (A) The concentration of transforming growth factor–β1 (TGF-β1) in LR-PRP was 1.2 times higher than that in LP-PRP. (B) The concentration of platelet-derived growth factor–AB (PDGF-AB) in LR-PRP was 1.8 times higher than that in LP-PRP. *P < .05.
Effects of LR-PRP and LP-PRP on Tenocyte Proliferation
On morphological analysis of our 5 experimental groups (Figure 6, A-E), both LR-PRP groups showed increased cell numbers, indicating better tenocyte proliferation, while in the LP-PRP group with 5000 platelets/µL, tenocytes were spread and elongated with decreased cell sizes.
As for WST-1 assay, the LR-PRP groups with 5000 platelets/µL and 10,000 platelets/µL showed 1.42 ± 0.01 and 1.41 ± 0.03 times higher tenocyte proliferation rates, respectively, compared with the control group (P < .05). No significant difference was found between the LR-PRP group with 10,000 platelets/µL and the control group (Figure 6F). In addition, the LR-PRP group with 5000 platelets/µL had a significantly higher proliferation rate than the LP-PRP group with 5000 platelets/µL had (1.42 ± 0.01 and 1.06 ± 0.02 times higher proliferation rate, respectively, compared with the control group; P < .05) (Figure 6F).

Effects of leukocyte-rich platelet-rich plasma (LR-PRP) and leukocyte-poor platelet-rich plasma (LP-PRP) on tenocyte morphology and proliferation on day 3. (A) Control tenocytes were well spread and elongated with an irregular shape. (B) Tenocytes cultured in LR-PRP with 5000 platelets/µL had increased cell numbers. (C) Tenocytes cultured in LR-PRP with 10,000 platelets/µL had increased cell sizes (arrows) and numbers. (D) Tenocytes cultured in LP-PRP with 5000 platelets/µL were spread and elongated (arrowheads) with decreased cell sizes. (E) Tenocytes cultured in LP-PRP with 10,000 platelets/µL had increased cell sizes. (F) WST-1 assay indicated that tenocyte proliferation in the LR-PRP group with 5000 platelets/µL was 1.42 times higher than that in the control group and 1.34 times higher than that in the LP-PRP group with 5000 platelets/µL. *P < .05. FBS, fetal bovine serum; O.D., Optical density; WST-1, water-soluble tetrazolium salt.
qRT-PCR for Gene Analysis
Tenocyte-related genes, such as TNC, COL1, COL3, and SCX, were upregulated in the LR-PRP and LP-PRP groups, with LR-PRP showing a higher expression of these genes. The expression of DCN, an extracellular matrix–related gene, was increased in the LR-PRP and LP-PRP groups. However, the expression of non–tendon related gene SOX9 was increased in LP-PRP and decreased in LR-PRP (Figure 7).

Effects of leukocyte-rich platelet-rich plasma (LR-PRP) and leukocyte-poor platelet-rich plasma (LP-PRP) on the gene expression of tendon- and non–tendon related cell markers. The expression levels of tenascin-C (TNC), type I collagen (COL1), type III collagen (COL3), scleraxis (SCX), and decorin (DCN) were higher in tenocytes in the PRP groups than in the control group, with LR-PRP higher than LP-PRP. The expression of non–tenocyte related gene, SRY-box transcription factor–9 (SOX9), was increased in tenocytes treated with LP-PRP but not with LR-PRP.
Discussion
We found in the current study that the tenocytes harvested from the torn edge of the RC and stimulated via LR-PRP with 5000 platelets/µL had a 1.42 ± 0.01 times higher proliferation rate than that of the control group and 1.34 times higher rate than that in the LP-PRP group with 5000 platelets/µL. This result was compatible with the growth factor concentrations in these 2 types of PRP. In addition, tenocyte-related genes had higher expression levels in tenocytes stimulated via LR-PRP than those stimulated via LP-PRP, while non–tenocyte related genes had higher expression levels in the LP-PRP groups. Our results suggest that LR-PRP has a beneficial effect in enhancing the proliferation of tenocytes from the torn RC.
In our PRP preparation protocol, although the concentrations of leukocytes in LR-PRP and LP-PRP were not significantly different, the ratio of WBCs to platelets in LR-PRP was significantly higher, which meant that the concentration of leukocytes in LR-PRP would be higher than that in LP-PRP when the concentration of platelets was a fixed value. Therefore, LR-PRP used in our experiments had a higher leukocyte concentration than LP-PRP had.
PRP has been widely used to promote wound and soft tissue healing because of its high concentrations of platelets and growth factors. 34 Chung et al 7 found that the local administration of PRP on a repaired supraspinatus tendon facilitated biological tendon-to-bone healing and increased the load to failure of the repaired rabbit RC. To the contrary, a single dose of platelet-poor plasma or 2 doses of PRP applied at the time of a rabbit medial collateral ligament injury did not improve ligament healing. 19 Therefore, the roles of different cytokines within PRP are still controversial. 18 Our study indicated that LR-PRP released more TGF-β1 and PDGF-AB than LP-PRP did. A previous clinical study revealed that a TGF-β1 level >61.79 μg/mL in PRP had better clinical outcomes for RC tendinopathy than the control group. 17 TGF-β1 inhibits matrix metalloproteinase–9 and -13 expression and increases collagen accumulation, improving tendon strength and tendon healing. 1,39 Based on these data, it is possible that higher levels of leukocytes and TGF-β1 in LR-PRP might have better efficacy in RC repair compared with LP-PRP.
Our research also indicated that LR-PRP stimulated RC tenocyte proliferation better compared with LP-PRP. In addition, tenocytes stimulated via LR-PRP demonstrated higher levels of tenocyte-related genes, while LP-PRP expressed a higher level of the chondrogenic gene SOX9. However, an in vitro study using rabbit tendon stem cells (TSCs) by Zhang et al 40 achieved the opposite result. In their study, LR-PRP had a negative effect on TSCs by inhibiting the proliferation of TSCs, accelerating nontenocyte differentiation, and inducing the apoptosis of TSCs. An animal study using a rabbit Achilles tendinopathy model to compare the efficacy of LR-PRP and LP-PRP concluded that LP-PRP improves tendon healing and is a preferable option for the clinical treatment of tendinopathy. 38 With a previous clinical study suggesting that high interleukin–1β and TGF-β1 levels are beneficial to the clinical outcomes of RC tendinopathy, we assume that the different conclusions of these studies may result from the different injury sites, such as the RC and Achilles tendon, and different cell origins, such as rabbits and humans.
Regarding human RC tears and different PRP formulations, Cross et al 8 demonstrated that LP-PRP promotes normal collagen matrix synthesis and decreases cytokines associated with matrix degradation and inflammation to a greater extent than LR-PRP does. To the contrary, Jiang et al 15 found a higher gene expression and more protein synthesis of COL1 with LR-PRP than with LP-PRP, which led to more mature collagen fibers, a larger fiber diameter, a higher failure load, and higher tensile stress. A network meta-analysis including 13 randomized controlled trials by Hurley et al 14 demonstrated that LP-PRP was found to significantly reduce the rate of retears and/or incomplete tendon healing after arthroscopic RC repair. It also improved visual analog scale for pain, Constant, and University of California, Los Angeles, scores. LR-PRP did not result in any significant improvements over the control group, except for the visual analog scale score for pain. However, post hoc analysis did not reveal significant improvements of LP-PRP over LR-PRP in any category. 14 Therefore, the evidence to support an optimal leukocyte concentration of PRP remains controversial, as the meta-analysis failed to record the platelet count, leukocyte to platelet ratio, growth factor concentrations, and whether PRP was activated. All these variables may be influenced by patient characteristics and preparation methods.
Limitations
There are limitations to our study. First, better tenocyte proliferation and gene expression in vitro cannot represent better clinical outcomes in patients with RC tears. Although a randomized controlled trial 29 found an improvement in pain with 2 injections of LR-PRP compared with a placebo injection, there are few studies that have directly compared LR-PRP and LP-PRP in RC tears. Further clinical studies should focus on the efficacy of LR-PRP and LP-PRP in RC tears. Second, the application timing of LR-PRP and LP-PRP is another key factor that may influence outcomes. For the treatment of acute tendon injuries, leukocytes in LR-PRP may be helpful when inflammation is at a critical stage by inducing a catabolic response and in fighting off infections. 25 However, the prolonged duration of the infiltration of excessive neutrophils may impair the healing process as chronic inflammation replaces acute inflammation. 9 Therefore, it is suggested that the application of LR-PRP and LP-PRP be dependent on the different types of tendon injuries and healing stages. 33 A recent study found that the intratendon delivery of LR-PRP at an early stage showed a beneficial effect for the repair of tendinopathy but not at a late stage. 20 Future studies should focus on the application of different PRP compositions to different stages of the tendon healing process.
A third limitation was that only 3 male patients were included in this study, as PRP from male patients contains a higher level of cytokines and growth factors compared with that from female patients. 37 Also, the ratio of growth factors to proinflammatory cytokines is higher in younger groups compared with older groups. 37 We enrolled 3 middle-aged male patients to control the study cohort as much as possible. Further studies should focus on sex-specific analysis in the relationship between RC injuries and the different preparations of PRP.
Conclusion
The findings of this study indicate that LR-PRP induced more growth factor release and increased RC tenocyte proliferation compared with LP-PRP.
Footnotes
Final revision submitted November 8, 2021; accepted December 31, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: Financial support for this study was received from the Ministry of Science and Technology and Chang Gung Memorial Hospital (grants 107-2314-B-182A-150-MY3, CMRPG5K0091, and CMRPG5K0211). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Chang Gung Medical Foundation (No. 201702198B0).
