Abstract
Background:
Rotator cuff repair is a common orthopaedic procedure that provides pain relief for many patients, but unfortunately, an estimated 20% to 70% of repair procedures will fail. Previous research has shown that elongation (ie, retraction) of a repaired tendon is common even in patients with a repair construct that appears intact on magnetic resonance imaging. However, it is unknown how this repair tissue functions under dynamic conditions.
Purpose:
To quantify static retraction and maximum dynamic elongation of repair tissue after rotator cuff repair.
Study Design:
Case series; Level of evidence, 4.
Methods:
Data from 9 patients were analyzed for this study. During surgery, a 3.1-mm tantalum bead was sutured to the supraspinatus tendon, medial to the repair site. Glenohumeral kinematics were assessed at 1 week (static) and 3 months (static and during scapular-plane abduction) after surgery using a biplanar videoradiographic system. The 3-dimensional position of the bead was calculated relative to the tendon’s insertion on the humerus (ie, bead-to-insertion distance). Static retraction was calculated as the change in the bead-to-insertion distance under static conditions between 1 week and 3 months after surgery, and maximum dynamic elongation was calculated as the maximal positive change in the bead-to-insertion distance during dynamic motion relative to the start of motion. The magnitudes of static retraction and maximum dynamic elongation were assessed with 1-sample t tests.
Results:
At 3 months after surgery, static retraction occurred in all patients by a mean of 10.0 ± 9.1 mm (P = .01 compared with no elongation). During scapular-plane abduction, maximum dynamic elongation averaged 1.4 ± 1.0 mm (P < .01 compared with no elongation). Descriptively, dynamic elongation consistently took 1 of 2 forms: an initial increase in the bead-to-insertion distance (mean, 2.0 ± 0.6 mm) before decreasing until the end of motion or an immediate and substantial decrease in the bead-to-insertion distance at the onset of motion.
Conclusion:
Repair tissue elongation (static retraction and maximum dynamic elongation) appeared to be a common and significant finding at 3 months after arthroscopic rotator cuff repair. Dynamic elongation of repair tissue during scapular-plane abduction exhibited 1 of 2 distinct patterns, which may suggest different patterns of supraspinatus mechanical and neuromuscular function.
Rotator cuff tears are common, 30,39,51 with approximately 250,000 surgical repair procedures performed in the United States each year. 13 Surgical repair typically provides short- to medium-term pain relief, but healing of repair tissue after rotator cuff repair remains a significant clinical problem. § Factors that are believed to affect repair tissue healing include tear size and chronicity, muscle atrophy, fatty infiltration, patient age, repair technique, and postoperative rehabilitation. 11,14,22,28,47,50 Despite our understanding of these factors, repair failure remains common, with reported rates ranging from approximately 20% to 70%. ∥
In addition to the relatively high retear rates, clinical outcomes after rotator cuff repair are often variable and are not always consistent with repair tissue integrity. For example, some studies have reported differences in shoulder strength between patients with intact and failed repair constructs, 7,12,18,27,35 whereas other studies have reported no difference. 2,33,42,47 Similarly, some studies have reported a difference in patient-reported outcome scores (eg, Constant score, American Shoulder and Elbow Surgeons score, subjective pain rating) between those with intact and failed repair constructs, 7,8,18,27,35 whereas other studies failed to detect a difference in these outcomes. 2,12,19,33,41,43 This disconnect between repair tissue integrity and clinical outcome emphasizes our incomplete understanding regarding the treatment of rotator cuff tears, postoperative repair tissue healing, and long-term shoulder function.
While it would be advantageous to accurately predict a patient’s outcome after rotator cuff repair, there are conflicting reports regarding the ability to predict outcomes based on conventional clinical data. For example, previous studies have reported that tear size is associated with repair integrity, 2,12,27,41 while others have reported that no association exists. 19,33,35,43 Similarly, studies have reported that patient age, tear retraction, muscle atrophy, and fatty infiltration are negatively associated with repair tissue healing and clinical outcomes. ¶ However, it has also been suggested that perhaps these factors are not strong predictors of clinical outcomes 25 and that patients' expectations may be a stronger predictor than any anatomic characteristic of the rotator cuff tear. 17 Indeed, a comprehensive review by McElvany et al 38 reported that “no conclusions could be reached regarding the effect of patient factors, tear characteristics, or treatment methods on clinical outcome” (p. 495).
Recent research suggests that repair tissue retraction may provide insight into postoperative healing and shoulder function that is not adequately provided by conventional clinical data. 37 Specifically, McCarron and colleagues 37 implanted tantalum markers into the supraspinatus tendon at the time of rotator cuff repair and then tracked the 3-dimensional (3D) marker positions over time using static computed tomography (CT). Their study demonstrated that even though only 30% of patients had a recurrent tendon defect, all repair constructs retracted in the first year (mean, 16.1 ± 5.3 mm [range, 6-23 mm]). That study showed that repair tissue retraction is common and may help explain why shoulder function is compromised after surgical repair. Unfortunately, this CT-based approach had a measurement error of ±3 mm 15 and limits the investigation to static conditions in which forces on repair tissue may not reflect those experienced during dynamic conditions.
The objective of this study was to use a biplanar videoradiographic system with high accuracy (±0.1 mm under dynamic in vivo conditions 48 ) to quantify static retraction and maximum dynamic elongation of repair tissue at 3 months after rotator cuff repair. We hypothesized that repair tissue would retract over time after surgery.
Methods
Participants
A total of 12 patients were recruited to participate in this study between January 2019 and March 2020. Eligible participants were aged 50 to 70 years with a full-thickness rotator cuff tear scheduled for arthroscopic repair. Reasons for exclusion and participant flow through the study stages are provided in Figure 1. Each participant provided written informed consent before data collection. The data collection protocol received institutional review board approval, and informed consent was obtained from each patient before participation.

CONSORT (Consolidated Standards of Reporting Trials) flow diagram showing participant enrollment, follow-up, and analysis. BMI, body mass index; MRI, magnetic resonance imaging; pre-op, preoperative.
Data Collection
Preoperative shoulder function was assessed approximately 1 week before surgery. Active shoulder range of motion (ROM) was assessed using a goniometer during abduction and flexion. Isometric shoulder strength was assessed using an isokinetic dynamometer (Biodex System 2; Biodex Medical Systems) during abduction, flexion, and internal and external rotation as previously described. 6 Shoulder strength was subsequently normalized by describing it as a percentage of normative values that were determined based on the regression equations developed by Hughes et al. 26 Specifically, characteristic variables such as age, sex, body mass, and dominance of the side tested were used to determine a predicted value for each participant and strength measure (ie, abduction, flexion, and internal and external rotation). Patient-reported pain (worst in the previous week) and function were assessed using the visual analog scale and Western Ontario Rotator Cuff Index (WORC), respectively.
Preoperative magnetic resonance imaging (MRI) was performed on various 1.5-T scanners and included axial and oblique sagittal proton density–weighted images with fat suppression, oblique coronal and oblique sagittal T1-weighted images without fat suppression, and oblique coronal T2-weighted images with fat suppression. A board-certified musculoskeletal radiologist with 15 years of experience evaluated each MRI scan and assessed tear size, fatty infiltration using modified Goutallier grades, 20,24 and tangent sign. 52
Rotator cuff repair was performed arthroscopically by 1 of 3 fellowship-trained orthopaedic surgeons (V.M., E.C.M., or S.M.). For each patient, the insertion site of the supraspinatus tendon was debrided, and suture anchors were implanted. Sutures were placed in the retracted tendon to reapproximate the tendon back to its insertion, and minimal acromioplasty was performed to remove any anterior subacromial spurring. The repair technique (ie, number of rows and anchors) was determined based on surgeon discretion. A double-row repair technique was used in 66.7% of cases with a median of 2 anchors (range, 1-4 anchors). After repair, a 3.1-mm tantalum bead was attached to the bursal surface of the supraspinatus tendon (Figure 2). This was accomplished by passing 2-0 FiberWire suture (Arthrex) through a 0.5-mm laser-drilled hole in the tantalum bead and then suturing the bead medial to the repair site.

Intraoperative image showing the implantation of a 3.1-mm tantalum bead to the bursal surface of the supraspinatus tendon.
After surgery, patients were discharged with a shoulder abduction sling and standard postoperative medications and precautions. Patients were also referred for physical therapy, which consisted of the following guidelines: (1) passive ROM only for the first 6 weeks after surgery, (2) progression to active-assisted ROM after postoperative week 6, (3) progression to active ROM by postoperative week 8, (4) isometric strengthening initiated in postoperative weeks 6 to 8, and (5) progression to resisted exercises after postoperative weeks 10 to 12. Rehabilitation progression was guided by patient tolerance and the avoidance of compensatory movement patterns (eg, shoulder shrugging).
At 1 week after surgery, glenohumeral kinematics and the 3D position of the implanted tantalum bead were determined under static conditions using a high-speed biplanar videoradiographic system that consisted of 2 high-voltage pulsed x-ray generators (model CPX 3100CV; EMD Technologies), two 40-cm image intensifiers (model P9447H110; Thales), and 2 cameras (Phantom VEO 340; Vision Research). 34 These data served as the baseline values by which to assess tendon elongation after surgery. Before imaging, participants were seated with their shoulder centered in the 3D imaging volume, draped with a protective lead apron, and asked to carefully remove their involved shoulder from the sling. The arm was placed comfortably at their side while static radiographic images were acquired.
At 3 months after surgery, glenohumeral kinematics and the 3D position of the implanted tantalum bead were once again determined under static conditions. In addition, biplanar videoradiographic images were acquired while the participants performed humeral abduction in the scapular plane through their existing ROM, which allowed for the assessment of repair tissue elongation under dynamic conditions. Finally, CT and MRI scans were also acquired at 3 months after surgery of each participant’s involved shoulder. The CT scan was acquired with the following parameters: helical scan mode; tube voltage, 120 kVp; tube current, 200-400 mA (auto); slice thickness, 1.25 mm; and field of view, 34 cm. Clinical MRI examinations (described previously) were performed to assess for structural integrity of the repair site and were interpreted by the same radiologist who evaluated the preoperative MRI scans. Importantly, the radiologist was blinded to the preoperative MRI results and the tendon elongation data.
Data Processing
Glenohumeral kinematics was calculated using methods described previously. 34 Briefly, the humerus and scapula were segmented from the CT scans and reconstructed into 3D bone models using Mimics software (Materialise). Glenohumeral kinematics was tracked by registering the volumetric bone models to calibrated distortion-corrected radiographic images using custom software. Likewise, the 3D bead position was tracked using custom software.
Given that glenohumeral kinematics and the 3D position of the implanted tantalum bead were both known relative to a common coordinate system, the distance between the bead and the humeral supraspinatus tendon insertion (ie, bead-to-insertion distance) could be calculated and used to estimate repair tissue length. To accomplish this, supraspinatus tendon insertion landmarks were identified on the 3D model of the humerus on the superior facet of the greater tuberosity along the anatomic neck (Figure 3). Next, for each frame of the motion trial, the shortest 3D contour line was calculated between the tantalum bead and each of the tendon insertion landmarks. Each contour line was calculated such that it followed the curvature of the humeral surface, which ensured that the bead-to-insertion distances remained physiologically plausible by not allowing the shortest distance vector to penetrate the bone’s surface (Figure 3B). The average of these individual contour line distances was then calculated to produce a single composite estimate of repair tissue length for each patient.
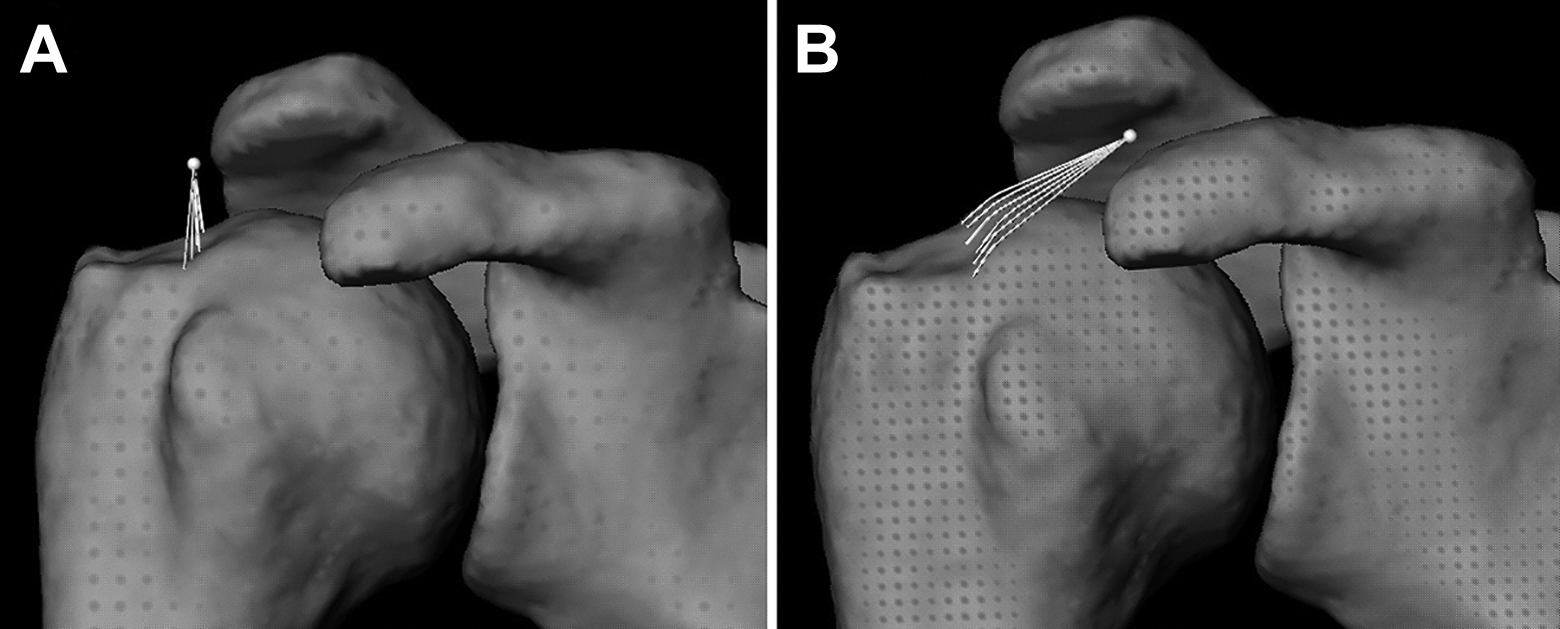
The location of the tantalum bead (white dot) placed during arthroscopic rotator cuff repair in patient 3 at (A) 1 week and (B) 3 months postoperatively. Note the location of the tendon insertion landmarks on the superior facet of the greater tuberosity along the anatomic neck and the 3-dimensional contour line (white lines) between each landmark and the bead. In this patient, static retraction of repair tissue at 3 months after surgery measured 11.9 mm, despite being classified as intact on magnetic resonance imaging.
Next, the bead-to-insertion distance data were examined, as they could have been influenced by factors other than repair tissue retraction. Specifically, previous work suggests that the position of a bead implanted into the supraspinatus tendon may move up to 10 mm in the anterior/posterior or superior/inferior directions as a result of changing glenohumeral joint positions. 15 Although the humerothoracic shoulder position was standardized during static image acquisition in the current study, the glenohumeral position may not have been precisely replicated across time. Consequently, patients whose bead position moved >10 mm in the anterior/posterior or superior/inferior directions relative to the humeral coordinate system under static conditions between 1 week and 3 months after surgery were excluded from further analysis (n = 3), as it suggests that the bead may not have remained firmly affixed to the tendon. All the excluded patients were deemed to have an intact repair construct on MRI.
Repair tissue deformation was described as follows for the remaining 9 patients. During the static trials, the change in the bead-to-insertion distance between 1 week and 3 months after surgery was calculated and termed “static retraction,” as it represented the amount that repair tissue had retracted away from the anatomic insertion over time. Finally, the positive change in the bead-to-insertion distance was calculated during the dynamic trials relative to the first frame of the motion trial, which estimated how much repair tissue elongated during the motion. The maximum amount of positive deformation (ie, elongation) that occurred during the motion trial was termed “maximum dynamic elongation.”
Statistical Analysis
Patient characteristic data were described using means and standard deviations or proportions, as appropriate. Differences between preoperative and 3-month postoperative measures of pain, self-reported function, active ROM, and normalized strength were assessed using paired t tests. Static retraction and maximum dynamic elongation were described using descriptive statistics. The magnitudes of static retraction and maximum dynamic elongation were assessed with 1-sample, 2-tailed t tests, which examined whether tendon deformation was significantly different from 0 (ie, no change). All statistical analyses were performed using R (R Core Team). Statistical significance was defined as P < .05.
Results
Characteristic data for the 9 patients included in the analysis are presented in Table 1. Compared with preoperative scores, patients experienced significant improvements in pain and self-reported function at 3 months after surgery (Table 2). However, changes in objective measures of function (ie, ROM and normalized strength) were not statistically significant. The MRI findings indicated that the rotator cuff repair construct was intact at 3 months after surgery in 8 of 9 patients.
Participant Characteristics, Preoperative Tear Characteristics, and Preoperative Physical Therapy

Pain and Function Outcomes Before and After Surgery a

a Data are presented as mean ± SD. Bolded P values indicate a statistically significant difference between preoperative and postoperative values (P < .05). ROM, range of motion; VAS, visual analog scale; WORC, Western Ontario Rotator Cuff Index.
At 3 months after surgery, static retraction of repair tissue had occurred in all patients by a mean of 10.0 ± 9.1 mm (range, 1.5-29.9 mm; P = .01) (Table 3). During scapular-plane abduction, the mean maximum dynamic elongation was 1.4 ± 1.0 mm (range, 0.0-2.8 mm; P < .01) (Table 3). The pattern of dynamic elongation during shoulder motion consistently took 1 of 2 forms. In 6 of 9 patients, the bead-to-insertion distance initially increased by a mean of 2.0 ± 0.6 mm, with maximum elongation occurring at a mean glenohumeral elevation angle of 41.5° ± 14.3°, and then decreased until the end of shoulder motion (Figure 4). All patients who presented with this pattern had an intact repair construct at 3 months after surgery. In the remaining 3 patients, the bead-to-insertion distance decreased immediately at the start of motion and almost exclusively continued to decrease throughout the motion (Figure 4). Of these patients, 2 had an intact repair site at 3 months after surgery, and the third was found to have a recurrent defect. Given that the supraspinatus tendon is assumed to lengthen in response to muscle contraction associated with motion, the 9 patients were separated into 2 subgroups for further analysis: functionally sufficient (n = 6) and functionally insufficient (n = 3). Specifically, patients whose tendons were classified as functionally sufficient exhibited an initial increase in the bead-to-insertion distance, followed by a decrease, while those with tendons classified as functionally insufficient exhibited an immediate decrease in the bead-to-insertion distance at the start of motion.
Repair Tissue Elongation and Repair Integrity by Patient


Changes in the bead-to-insertion distance in all patients at 3 months postoperatively relative to (A) 1 week postoperatively and (B) the first frame of the motion trial (scapular-plane abduction). Each trajectory represents an individual patient and is labeled consistently with Table 3. Note that patient 5 (P5) had very little glenohumeral motion at 3 months postoperatively and their change in bead-to-insertion distance very nearly overlaps that of Patient 9 (P9) in (A). Note also that the data trajectories are the same in (A) and (B), except that in (B), they are collapsed along the y-axis, such that the magnitude is 0 for the first frame of the motion trial. This allows for the change in repair tissue length (A) over time and (A, B) during scapular-plane abduction to be appreciated. In either (A) or (B), it is evident that the bead-to-insertion distance trajectories during scapular-plane abduction consistently took 1 of 2 forms. In 6 of 9 patients, the bead-to-insertion distance initially increased by a mean of 2.0 ± 0.6 mm, with maximum elongation occurring at a mean glenohumeral elevation angle of 41.5° ± 14.3°. After this point, the bead-to-insertion distance decreased until the end of motion. In the remaining 3 patients, the bead-to-insertion distance immediately decreased during the motion trial. Given that it is expected that the supraspinatus tendon should lengthen in response to muscle contraction, the patients were separated into 2 subgroups: functionally sufficient (n = 6) and functionally insufficient (n = 3). P, patient.
Differences in pain and function between the 2 patient subgroups (ie, functionally sufficient and functionally insufficient) are provided descriptively in Table 4 because of small and unbalanced sample sizes. Static retraction of repair tissue was descriptively higher in the functionally insufficient subgroup (16.4 ± 14.1 mm) versus the functionally sufficient subgroup (6.8 ± 4.0 mm). Similarly, maximum dynamic elongation of repair tissue during shoulder abduction in the functionally insufficient subgroup (0.3 ± 0.3 mm) was descriptively less than in the functionally sufficient subgroup (2.0 ± 0.6 mm). Interestingly, compared with the functionally sufficient subgroup, the functionally insufficient subgroup tended to have better pain and WORC scores but worse objective measures of function (ie, active ROM and normalized strength) (Table 4). Videos illustrating glenohumeral and bead kinematics in 3 patients are available in the Supplemental Material, and still images from these videos are presented in Figure 5.
Repair Tissue Mechanics and Pain and Function Outcomes in Functionally Sufficient and Functionally Insufficient Patients a

a Data are presented as mean ± SD (range). ROM, range of motion; VAS, visual analog scale; WORC, Western Ontario Rotator Cuff Index.
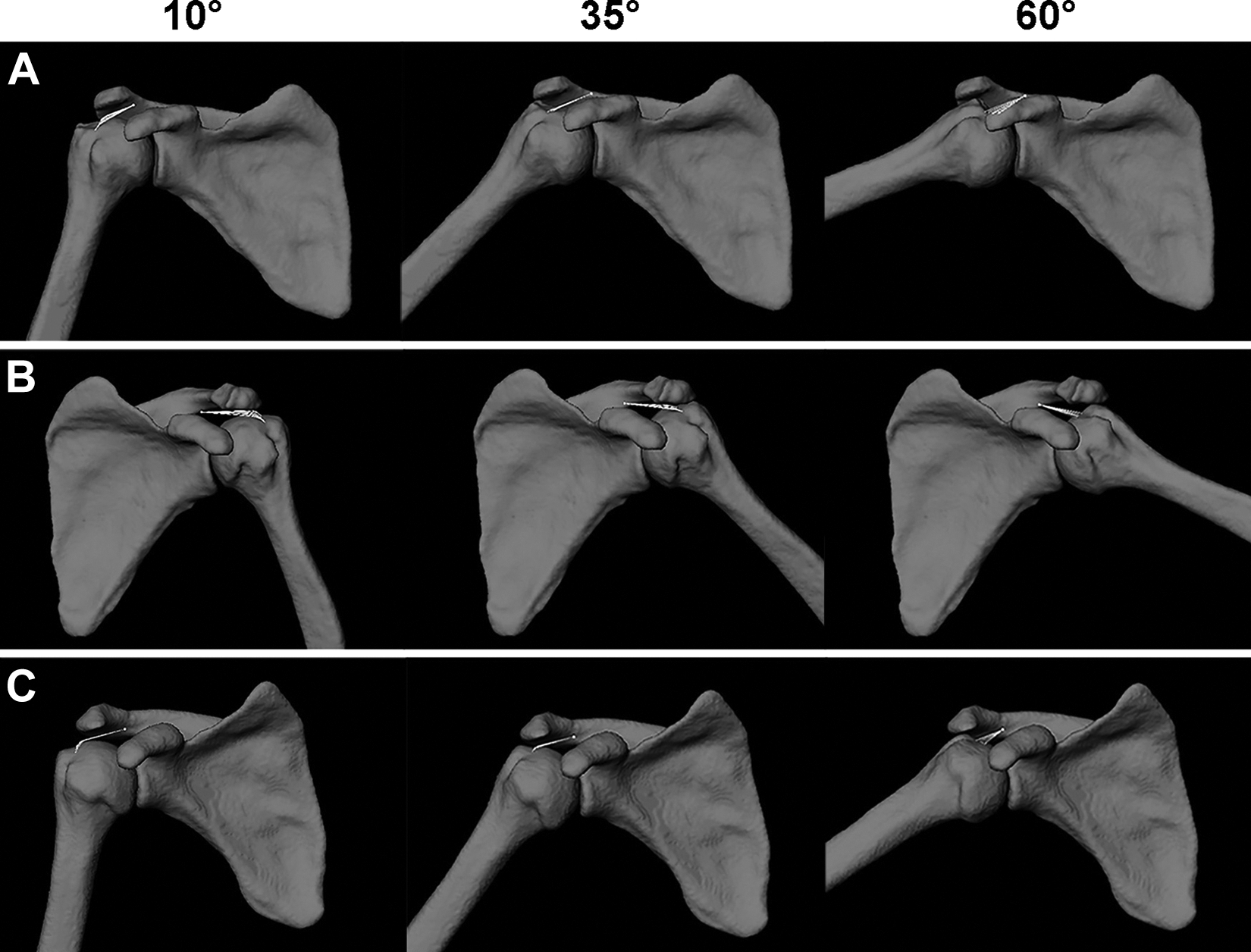
Still images from the supplementary videos illustrating glenohumeral and bead kinematics during a single trial of scapular-plane abduction for (A) patient 3, who was classified into the functionally sufficient subgroup; (B) patient 2, who was classified into the functionally insufficient subgroup because of a recurrent defect (ie, retear) on magnetic resonance imaging (MRI); and (C) patient 8, who was classified into the functionally insufficient subgroup but had an intact repair construct on MRI. Images are shown at 10°, 35°, and 60° of glenohumeral elevation. Note that the bead displaces medially during the dynamic trial in patient 3 (A) which is expected with supraspinatus muscle contraction when the muscle-tendon unit is structurally intact. However, in patients 2 and 8 (B and C, respectively), the absence of substantial medial bead displacement likely explains the steady decrease in the bead-to-insertion distance during the motion trial (Figure 4) as the humeral tendon insertion gradually approximates the bead.
Discussion
There are 3 key findings from this study. First, static retraction of repair tissue occurred in all patients, albeit to varying degrees, which supported our hypothesis (Table 3). Second, dynamic elongation of repair tissue during shoulder abduction in each patient exhibited 1 of 2 distinct patterns (Figure 4), which may have important implications for understanding postoperative shoulder function and potentially help reconcile the disconnect between structural and functional outcomes after rotator cuff repair. Third, significant improvements in strength or ROM were not detected between preoperatively and 3 months postoperatively, despite patient-reported improvements in pain and function (Table 2).
Previous studies documenting the effects of rotator cuff repair have often used postoperative imaging to compare the outcomes of patients whose repair construct is structurally intact versus those whose repair construct is structurally retorn. # When functional outcomes (strength, ROM, and/or patient-reported outcomes) are consistent with imaging-based structural outcomes, then the surgical procedure can be easily characterized as a successful repair or failed repair. However, it is not uncommon for a disconnect to exist between structural and functional outcomes. For example, McElvany and colleagues 38 reviewed 77 studies that compared the clinical results for intact and failed repair and concluded that patient-reported outcomes generally improve, regardless of whether the repair construct remains intact. Furthermore, McElvany et al 38 reported that “there was no consistent relationship between the integrity of the repair and the clinical outcome” (p. 495). Although it remains difficult to fully reconcile the disconnect between structural and functional outcomes after rotator cuff repair, we believe that the findings from this study provide preliminary evidence to support a mechanistic understanding for this disconnect.
The finding of static retraction of repair tissue occurring in all patients after rotator cuff repair, even in those with an apparently intact repair site on MRI, has many potential clinical implications. From the current study, it is difficult to distinguish the reason for the increased bead-to-insertion distance (ie, static retraction) over time. In patients with presumably intact repair constructs based on MRI findings, an increased bead-to-insertion distance may suggest that repair tissue has retracted and/or elongated but remains grossly intact. On the other hand, the discrepancy between these findings and MRI results may call into question the sensitivity of MRI for detecting recurrent defects. For example, it is possible that some repair constructs failed but that the defects were filled in with scar tissue, confusing the interpretation of repair integrity on MRI. Importantly, this possibility may help explain the disconnect between presumed repair integrity and functional outcomes often reported in the literature. 38 More research is needed to investigate these possibilities to improve the detection of recurrent tears.
Despite uncertainty about the physiological reason for the increased bead-to-insertion distance over time, the findings are in excellent agreement with a previous study of 14 patients by McCarron and colleagues. 37 Specifically, McCarron et al 37 reported an average static retraction of 12.4 mm (range, 3.5-20.7 mm) at 3 months after surgery, whereas the current study reported a mean static retraction of 10.0 mm (range, 1.5-29.9 mm) at the same time point. McCarron and colleagues 37 reported that repair tissue retraction increased from 12.4 mm at 3 months after surgery to 16.1 mm at 1 year after surgery, suggesting that most repair tissue retraction occurs during the critical 3-month healing period after rotator cuff repair. It is possible that repair tension may influence the extent to which repair tissue elongation occurs, and more research is needed to investigate this possibility. Interestingly, repair tissue retraction has also been observed after biceps tendon repair 36 and Achilles tendon repair. 44 These findings suggest that repair tissue retraction may occur after all tendon repair procedures and that any corresponding functional deficits, along with clinical considerations designed to minimize repair tissue retraction, warrant additional investigation.
Although static retraction provides important information regarding repair tissue’s potential structural integrity, measures of dynamic elongation may be able to provide additional information regarding its function. The most common pattern of dynamic elongation of repair tissue observed in this study involved an initial elongation of approximately 1 to 3 mm over the first 30° to 50° of glenohumeral abduction, followed by a steady decrease in the bead-to-insertion distance as the abduction angle increased (Figure 4). The initial elongation of repair tissue is likely caused by muscle contraction as the supraspinatus muscle contributes to shoulder abduction and dynamic glenohumeral joint stability. This interpretation is supported by previous electromyography studies reporting that the supraspinatus muscle is active primarily over the first 30° to 40° of humerothoracic abduction. 1 The initial decrease in the bead-to-insertion distance that occurs with increasing abduction is likely because of elastic recoil of the supraspinatus tendon, an important function of tendons in facilitating joint motion. 3 At higher abduction angles, the decrease in the bead-to-insertion distance is likely because of the combination of a decrease in supraspinatus muscle contraction, which results in less medial bead displacement, and the supraspinatus tendon insertion coming into closer proximity to the bead as the humerus continues to abduct.
In 3 of 9 patients, repair tissue did not elongate, but rather, the bead-to-insertion distance underwent an immediate and marked decrease during scapular-plane abduction (Figure 4). Interestingly, this pattern of decreasing the bead-to-insertion distance occurred in 1 patient with a recurrent tear (patient 2 in Figure 4) and 2 patients with an intact repair site (patient 1 and patient 8 in Figure 4). These data suggest that it is possible for the supraspinatus tendon in patients with an intact repair site to function mechanically in a way that is consistent with a retorn supraspinatus tendon. There are several potential explanations for this phenomenon. First, it is possible that excessive static retraction of repair tissue, which can be interpreted as lengthening of the tendon, results in a corresponding decrease in muscle length because the origin and proximal and distal insertions of the muscle-tendon unit are unchanged. A muscle’s force-generating capacity is influenced by its force-length relationship 23 ; therefore, it is unlikely that a shortened supraspinatus muscle will be capable of generating the same maximum force as it would in the native (ie, unshortened) condition. Second, the ability to transmit muscle forces through a lengthened and presumably less stiff tendon may be compromised when there is substantial static retraction of repair tissue. Third, supraspinatus neuromuscular impairments that may occur in response to a chronic tendon tear may not be adequately restored by surgical repair and/or addressed by current postoperative rehabilitation strategies. Lastly, it is possible that postoperative MRI failed to detect a recurrent tear in these 2 patients, despite being assessed independently by 2 blinded radiologists. Regardless of why an intact supraspinatus tendon would function mechanically like a retorn supraspinatus tendon, it is not surprising that these 3 patients had descriptively lower strength at 3 months after surgery compared with the 6 patients who were classified into the functionally sufficient subgroup (Table 4), because selective nerve block studies have shown that the supraspinatus and infraspinatus together account for 25% to 75% of overall shoulder strength. 21,32 While these data provide initial insight into the disconnect between structural and functional outcomes, further research is necessary to fully understand the complex relationships between repair integrity, supraspinatus function, and shoulder function after rotator cuff repair.
The finding of repair tissue retraction occurring in all patients may have implications for postoperative rehabilitation. Specifically, if increased repair tissue retraction is found to be associated with impaired long-term shoulder function (eg, strength, ROM, and dynamic joint stability), then this may suggest that a more conservative approach is warranted regarding the duration of immobilization, physical therapy exercise prescription, and gradual resumption of shoulder activities within the critical 3-month healing period after surgical repair. However, there is currently little understanding of how the volume and/or intensity of shoulder activities (ie, rehabilitation and activities of daily living) influence repair integrity and clinical outcomes after rotator cuff repair. The nearly 20-fold range of static retraction values across patients (range, 1.5-29.9 mm) further emphasizes the limited understanding of how mechanical loading through postoperative activities influences repair tissue mechanics. It is likely that repair tissue retraction is also influenced by the inherent physical properties of the muscle-tendon unit before surgery and that postoperative activity levels need to be more carefully calibrated to muscle-tendon properties to minimize repair tissue retraction and optimize shoulder function.
Limitations
This study has limitations to consider when interpreting the results. The primary limitation is the small sample size, which occurred because of 2 methodological challenges. Recruiting eligible and willing participants proved to be a major challenge in part because of the invasive nature of the study (Figure 1). Additionally, we chose to discontinue bead implantation as a precaution after potential displacement in 3 patients, as previously described. Similar issues with bead displacement were reported by McCarron et al 37 and highlight the challenges associated with assessing in vivo tendon deformation. While the approach used to identify patients with excessive bead displacement is evidence based, it is possible that subtler bead displacement occurred in the remaining 9 patients and could explain some of the high between-participant variability. In these patients, however, the primary direction of bead displacement occurred medially, which is consistent with the direction of clinical tendon retraction.
The invasive nature of the study also resulted in slow recruitment. Consequently, patients from multiple surgeons were recruited in an effort to improve enrollment. However, this introduced the potential for surgeon experience and repair methodology to confound the results. Although surgeons attempted to implant the tantalum bead in a consistent way, it is possible that inconsistencies between surgeons may have influenced bead displacement data. Furthermore, the small sample size also did not allow for the assessment of potential subgroups based on tear configuration and important surgical factors such as full anatomic tear reduction, both of which could have influenced bead displacement over time.
Another limitation of this study is the short follow-up period (ie, 3 months after surgery). However, previous studies that have conducted MRI or ultrasonography longitudinally after rotator cuff repair report that 42% to 78% of retears occur within the first 3 months after surgery, 4,29,40 as does the majority of repair tissue retraction. 37 Therefore, the results of the study cover this critical period in a patient’s postoperative recovery. Furthermore, the relatively short follow-up period may have influenced the ability to detect improvements in shoulder strength, given that postoperative rehabilitation protocols do not allow for resisted exercises until approximately 10 to 12 weeks after surgery. We plan to assess longer-term outcomes in future work.
Conclusion
Repair tissue elongation (static retraction and maximum dynamic elongation) appeared to be a common and significant finding at 3 months after arthroscopic rotator cuff repair. Dynamic elongation of repair tissue during scapular-plane abduction exhibited 1 of 2 distinct patterns, which may suggest different patterns of supraspinatus mechanical and neuromuscular function. Taken together, the findings of this study lend insight that may help to reconcile the disconnect between structural and functional outcomes after rotator cuff repair.
Supplemental material for this article is available at: http://journals.sagepub.com/doi/suppl/10.1177/23259671221084294.
Footnotes
Acknowledgment
The authors thank the participants who volunteered for this study, as well as Steven B. Soliman, DO, RMSK, for interpreting the MRI scans.
Final revision submitted November 29, 2021; accepted December 23, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research reported in this article was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health (award No. R01AR051912). V.M. has received education payments from Arthrex, consulting fees from Pacira Pharmaceuticals, and hospitality payments from Smith & Nephew and Stryker. E.C.M. has received education payments from Arthrex, consulting fees from Endo Pharmaceuticals and Smith & Nephew, and speaking fees from Smith & Nephew. S.M. has received education payments from Arthrex; consulting fees from DePuy/Medical Device Business Services, Exactech, and FX Shoulder USA; and hospitality payments from Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Henry Ford Health System (No. 12146).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
