Abstract
Objectives:
Anterior cruciate ligament (ACL) injury is one of the leading orthopedic injuries in young individuals and even with surgical repair it is reported that around 50% of patients develop post-traumatic osteoarthritis (PTOA). Although ACL reconstruction improves anterior-posterior knee joint stability and cartilage contact biomechanics, some studies have shown persistent abnormalities in the tibiofemoral kinematics. Extensive rehabilitation work is often required following both ACL tear and ACL reconstruction surgery. New bone formation (osteophyte) and synovial inflammation (synovitis) are often associated with the progression and severity of OA post-injury and surgery, however the effect of mechanical loading stimulus in musculoskeletal repair remains relatively unexplored. The role played by resident synovial stromal cells in the activation of their local inflammatory and regenerative programming is poorly understood. Work in our laboratory has shown that in vivo imaging of activated fibroblasts using 68Ga-FAP radiotracer may be a valuable tool to follow the progression of OA and the bone-tendon interface healing. This study aims to explore the therapeutic effect of exercise following ACL reconstruction on the progression or mitigation of PTOA following an ACL injury, evaluating the mechanism and contribution of synovial fibroblast activation using microPET/CT imaging.
Methods:
All study procedures were approved by Weill Cornell IACUC (#2019-0034). Thirty, 12 week old male C57BL/6 mice underwent a non-invasive ACL rupture induced by a single axial compressive load of the tibia at a rate of 1mm/s on the right knee (
Results:
PET/CT scans demonstrated accumulation of 68Ga-FAP in the injured and surgical knees compared to the contralateral healthy control knees (
Conclusions:
The intimal layer of the native synovium is composed of 2 main cell types: macrophages and fibroblast-like cells. Recent studies have shown a correlation between activated macrophages and their association with OA severity and joint symptoms; however, the cross-talk between these 2 types of cells and their contribution to PTOA following ACL injury and reconstruction surgery is poorly understood. Our study identified time-dependent changes in FAP activity following ACL reconstruction and mechanical loading stimulus that could be correlated to PTOA development. Our data demonstrates that ACL reconstruction followed by a low intensity exercise regimen leads to a decrease of FAP activity in the ACL injured knee. We also found that the ACL reconstruction groups had less degenerative changes in the posterior medial tibial plateau, which may be due to an improvement of anterior-posterior joint stability, and the exercise group showed a greater amount of bone formed at the tendon-bone interface of the tibia suggesting a superior graft healing process. Immunochemistry for FAP-α is currently being analyzed to confirm the spatial localization of FAP expression.

Study design, All animals underwent a non-invasive ACL rupture and were allocated into 4 treatment groups: non-treated (Gl); non-treated with sham surgery (G2); treated with ACL reconstruction (G3); treated with. ACL reconstruction and treadmill exercise.
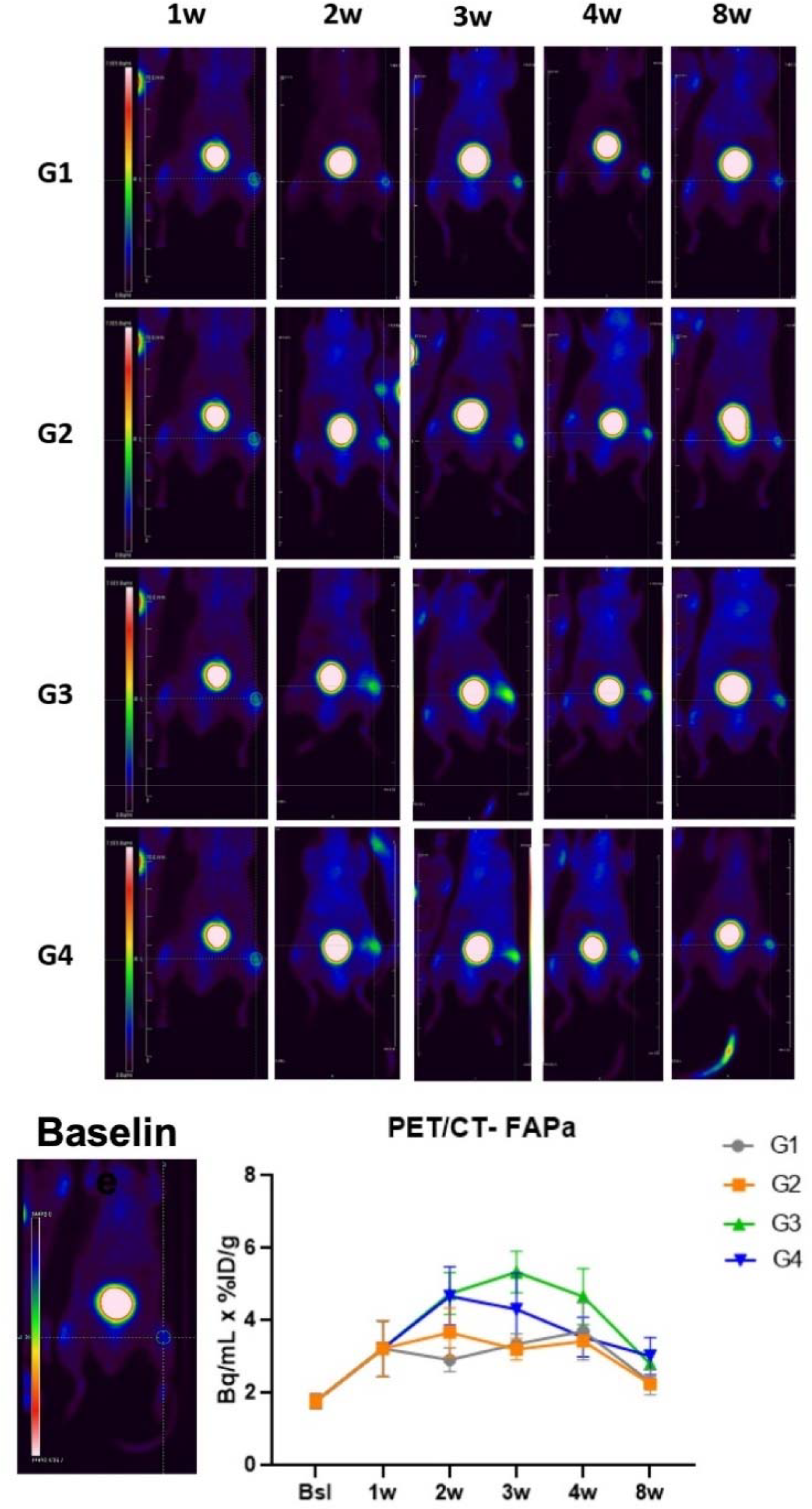
PET/CT representative coronal images of the 4 different groups and quantitative results for 68Ga-FAPI uptake over time.

microCT analysis demonstrating the surface morphology of the mouse knee joint showing the marked structural changes in the posterior medial compartment leading to erosion of the tibial plateau (A, yellow circle) and the amount of new bone formed al the tendon-bone interface of the tibia in groups 3 and 4 (B).
