Abstract
Background:
The impact of preoperative opioid use on outcomes after shoulder surgery is unknown.
Purpose/Hypothesis:
To examine the role of preoperative opioid use on outcomes in patients after shoulder surgery. We hypothesized that preoperative opioid use in shoulder surgery will result in increased postoperative pain and functional deficits when compared with nonuse.
Study Design:
Systematic review; Level of evidence, 3.
Methods:
A systematic review was performed using the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Included were all English-language studies comparing clinical outcomes of shoulder surgery in patients who used opioids preoperatively (opioid group) as well as patients who did not (nonopioid group) with a minimum follow-up of 1 year. Outcomes included range of motion, American Shoulder and Elbow Surgeons score, Constant-Murley score, and visual analog scale for pain. Study quality was evaluated with the Modified Coleman Methodology Score and the MINORS score (Methodological Index for Non-randomized Studies).
Results:
Included were 5 studies (level 2, n = 1; level 3, n = 4): Two studies were on total shoulder arthroplasty, 2 on reverse total shoulder arthroplasty, 1 on both, and 1 on arthroscopic rotator cuff repair. There were 827 patients overall: 290 in the opioid group (age, 63.2 ± 4.0 years [mean ± SD]; follow-up, 38.9 ± 7.5 months) and 537 in the nonopioid group (age, 66.0 ± 4.7 years; follow-up, 39.5 ± 8.1 months). The opioid group demonstrated significantly worse pre- and postoperative visual analog scale and Constant-Murley score pain scores as compared with the nonopioid group. Mean American Shoulder and Elbow Surgeons scores were significantly lower in the opioid group at pre- and postoperative time points as compared with the nonopioid group (P < .05 for all). However, both groups experienced similar improvement in outcomes pre- to postoperatively. One study showed that the opioid group consumed significantly more opioids postoperatively than the nonopioid group and for a longer duration (P < .05). The overall mean Modified Coleman Methodology Score and MINORS score were 64.2 ± 14 and 15.8 ± 1.0, respectively.
Conclusion:
Opioid use prior to various shoulder surgical procedures negatively affected postoperative pain and functionality. Although the opioid group showed significantly worse scores postoperatively, the groups experienced similar improvements.
The opioid crisis continues to complicate the pre- and postoperative management of patients within the orthopaedic community. 20 According to the US Centers for Disease Control and Prevention, 400,000 people died from an overdose involving any opioid, prescription or illicit, between 1999 and 2017. Orthopaedic surgeons are the third-highest prescribers of opioids in the United States. 34 For example, in a large prospective study, postoperative opioids were being prescribed approximately 3 times more than needed after upper extremity procedures such as shoulder surgery. 13 Furthermore, a similar study found that 30 opioid pills, which is the number most frequently prescribed after upper extremity surgery, appear to be excessive in this context and may become a source of diversion. 23 However, with increased awareness, guideline changes, and patient education, decreases in opioid prescribing are occurring, including within the orthopaedic community. 13,27
Although preoperative opioid use has been studied in the context of total knee and hip arthroplasties, no systematic review has been conducted to compare patient-reported outcomes (PROs) in patients undergoing shoulder surgery with or without preoperative opioid consumption. The purpose of this study was to compare postoperative outcomes of patients after shoulder surgery based on preoperative opioid consumption. We hypothesized that the effects of preoperative opioid use in shoulder surgery will result in increased postoperative pain and functional deficits when compared with patients who have not preoperatively used opioids.
Methods
This systematic review was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and checklist. Two independent reviewers (D.M.J.D and J.-R.S.) searched PubMed, Embase, and the Cochrane Library for articles published through March 30, 2020. The following search phrase was used: preoperative AND opioid AND shoulder. A total of 216 studies were reviewed by title and/or abstract to determine eligibility based on the following inclusion criteria: studies reporting pre- and postoperative clinical outcomes of patients who underwent various shoulder surgical procedures, studies reporting pre- and postoperative PRO scores, studies comparing patients who took any opioids preoperatively and those who did not, minimum 1-year follow-up, English-language literature, and retrospective and prospective studies of evidence levels 1 to 4. Exclusion criteria included cadaveric or animal studies, nonclinical studies, and case reports. Data extraction was performed independently (D.M.J.D.), and when studies were comparative, only data from the population that met the aforementioned criteria were extracted. Funding and third-party involvement were not required in this analysis.
Assessment of Study Quality
The quality of study methodology was evaluated using the Modified Coleman Methodology Score (MCMS) based on a scaled potential score ranging from 0 to 100. Scores ranging from 85 to 100 are excellent, 70 to 84 good, 55 to 69 fair, and <55 poor. 6
Risk of bias was assessed using the Methodological Index for Non-randomized Studies (MINORS score), which incorporates 8 items to assess overall bias and 12 items in comparative studies. The MINORS score has scaled potential scores, ranging from 0 to 16 for noncomparative studies and 0 to 24 for comparative studies. Each item is scored 0 (not reported), 1 (reported but inadequately), or 2 (reported adequately). 26
Reporting Outcomes
The following outcomes were assessed: age, sex, follow-up, range of motion (ROM), body mass index (BMI), shoulder surgery type, preoperative opioid use definition, opioid morphine equivalents, complication rates, comorbidities, and PRO scores. ROM was based on forward flexion, external rotation at the side and at 90° abduction, internal rotation at the side and in abduction, and abduction. PROs included the Constant-Murley score (CMS), 7 visual analog scale (VAS) for pain, 4 Simple Shoulder Test, 15 American Shoulder and Elbow Surgeons (ASES) shoulder score, 22 Western Ontario Osteoarthritis of the Shoulder (WOOS), 16 and the Single Assessment Numeric Evaluation. 33 ROM and PROs were evaluated both pre- and postoperatively. Opioid consumptions are converted and reported as morphine equivalents.
Statistical Analysis
The heterogeneity of the overall patient population prevented pooling of outcome data and meta-analysis calculations. Therefore, descriptive statistics are presented. The mean values and accompanying standard deviations are reported for pain scores, functionality scores, and ROM. Overall improvement is indicated between the pre- and postoperative time points. Values for Thompson et al 28 are presented as medians, following the format of the authors.
Results
Five studies 5,17,18,28,32 (level 3, n = 4; level 2, n = 1) published between 2015 and 2019 were included in this study (Figure 1).
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Assessment of Study Quality
Table 1 shows the results of the MCMS and MINORS scores. The MCMS from the 5 studies (mean, 64.2) indicated fair methodology. The mean MINORS score for comparative studies was 15.8.
Studies: MCMS and MINORS Scores a

a MCMS, Modified Coleman Methodology Score; MINORS, Methodological Index for Non-randomized Studies; NR, not reported.
Patient Demographics
A total of 827 patients were included in this systematic review: 290 (35%) preoperative opioid users and 537 (65%) nonusers. Of the opioid group, 128 (43%) were male, and of the nonopioid group, 311 (59%) were male. Patient age at the time of the surgery ranged from 58.6 to 68.5 years for the opioid group and 58.4 to 71.4 for the nonopioid group. The mean follow-up ranged from 34.0 to 47.2 months for the opioid group and from 31.5 to 47.2 months for the nonopioid group (Table 2). All studies analyzed BMI, but only 1 study 18 reported a statistical difference (P = .01) between the opioid users and those who did not use opiods. Cheah et al 5 reported BMI as >30 and <30. There were 84 patients in the opioid group versus 73 in the nonopioid group with a BMI >30.
Patient Demographics of the Included Studies a
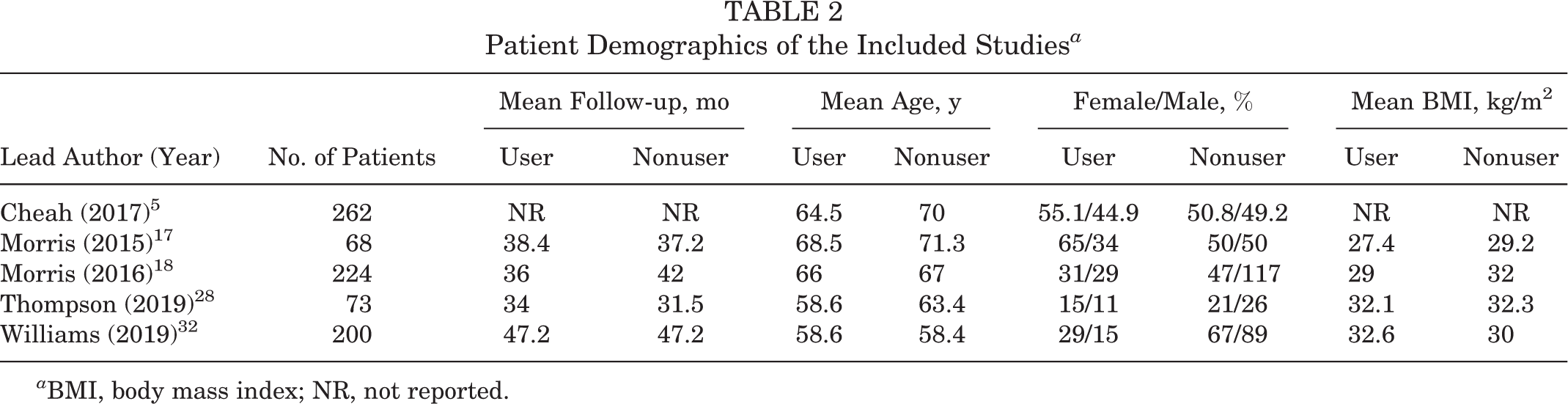
a BMI, body mass index; NR, not reported.
Four studies 17,18,28,32 examined comorbidities such as smoking status, chronic back pain, depression, diabetes, and heart disease (Table 3). Williams et al 32 and Morris et al 18 found significantly higher frequencies of back pain (P = .007 and P = .004, respectively) and depression (P < .04 and P = .04) in the opioid group versus the nonopioid group. However, 3 of these studies 17,18,28 showed no statistically significant differences between groups in regard to age, sex, follow-up duration, smoking, diabetes, and heart disease.
Patient Comorbidities of the Included Studies a

a Values are reported as No. (%) of patients. NR, not reported.
Shoulder Diagnoses and Pathologies
Two studies 18,28 reported on total shoulder arthroplasty (TSA), 1 study 17 on reverse TSA (RTSA), and 1 study 5 on both these procedures, with an additional study 32 on arthroscopic rotator cuff repair. Study summaries and opioid user definitions are presented in Table 4.
Studies: Summaries and Opioid User Definitions a

a RTSA, reverse total shoulder arthroplasty; TSA, total shoulder arthroplasty.
b All studies were retrospective reviews.
Surgical Indications
Total Shoulder Arthroplasty
Morris et al 18 reported that all 224 patients underwent TSA for the treatment of primary glenohumeral arthritis. A total of 22 (9.8%) patients had revision surgery or an intra- or postoperative complication.
Thompson et al 28 stated that all 73 patients underwent TSA for primary glenohumeral osteoarthritis. Initially, 180 anatomic TSAs were eligible, but 65 were excluded for diagnoses other than primary glenohumeral osteoarthritis, such as inflammatory arthritis, revision arthroplasty, posttraumatic arthritis, and osteonecrosis. Of the 115 patients, 73 (63.5%) met the 2-year follow-up inclusion criteria.
Cheah et al 5 did not report the indication for TSA or RTSA.
Reverse Total Shoulder Arthroplasty
Morris et al 17 did not state the indications for RTSA surgery, although exclusion criteria included intra- or postoperative complications: dislocation (n = 3), infection (n = 2), intraoperative humeral fracture (n = 2), acromial stress fracture (n = 1), and scapular spine stress fracture (n = 1).
Arthroscopic Rotator Cuff Repair
Williams et al 32 reported that 200 patients underwent arthroscopic rotator cuff repair for full- or partial-thickness tears of the supraspinatus tendon. Exclusion criteria included patients <18 years old, revision cases, history of arthroscopic shoulder surgery, and patients without pre- and postoperative outcomes questionnaires from the study sample.
Surgical Technique and Prostheses
Total Shoulder Arthroplasty
Morris et al 18 reported on the following anatomic TSA systems: Aequalis, Aequalis Ascend, and Aequalis Ascend Flex (Tornier). All followed standard surgical techniques and postoperative rehabilitation protocols. 8,9,14
Thompson et al 28 noted that the surgical procedures were performed with the patient in the beach-chair position with a deltopectoral approach and that muscular paralysis was used. The subscapularis was managed with tenotomy and transosseous repair. Humeral stems were noncemented, and glenoid components used a central post with cementing of the peripheral pegs.
Cheah et al 5 did not include details of the surgical techniques.
Reverse Total Shoulder Arthroplasty
Morris et al 17 reported that the Aequalis RTSA system was used for all patients during the study period. The RTSA technique used during the study period was well-described, and a standardized postoperative rehabilitation protocol was followed. 9,14,29
Arthroscopic Rotator Cuff Repair
Details of surgical techniques were not indicated by Williams et al. 32
Clinical Outcomes
Pain Scores
Three studies 5,28,32 examined pain using the VAS (Table 5). Cheah et al 5 compared VAS scores of nonopioid users, short-acting opioid users, and long-acting opioid users preoperatively and at 4 postoperative time points (0, 1, 2, and 3 days). Short-acting opioids include oral hydrocodone, oxycodone, hydromorphone, and morphine. Long-acting opioids include extended-release morphine, extended release oxycodone, methadone, and fentanyl patch (Table 4). The authors stated that preoperatively, the long-acting opioid users had higher VAS scores than either the short-acting or the nonuser group, although this was not significantly different (P = .1). Notably, however, at postoperative day 1, the long-acting opioid users did have significantly higher VAS scores (6.0 ± 1.5) as compared with the short-acting users (4.9 ± 1.9) and the nonuser group (4.0 ± 1.8; P < .001). VAS scores were not reported beyond that of 3 consecutive postoperative days.
Pain Scores a

a Values are expressed as mean ± SD, unless noted otherwise. ASES, American Shoulder and Elbow Surgeons; CMS, Constant-Murley score; NR, not reported; VAS, visual analog scale.
b Pre- vs postoperative change between groups.
c Long-acting opioid user.
d Postoperative day 3 (SD not reported).
e Median values.
Thompson et al 28 found that the opioid user group had significantly worse median VAS scores preoperatively (P = .02) and postoperatively (P = .001) as compared with the nonopioid users. Similarly, Williams et al 32 found a trend toward higher VAS scores at pre- and postoperative time points in the opioid user group; however, these differences were not statistically significant (P = .3). Both studies found no significant differences in VAS score improvement pre- to postoperatively between groups (P > .05 for both).
Two studies 17,18 examined pain using CMS and the ASES score (Table 5), finding that the opioid group had significantly lower preoperative CMS (P < .05) and ASES (P < .01) outcomes as compared with the nonopioid group. Only Morris et al 18 found that the opioid group had significantly lower postoperative CMS (P = .01) and ASES (P = .02) values versus the nonopioid group. However, both studies stated that there were no significant differences in CMS or ASES score improvement pre- to postoperatively between groups (P > .05 for both).
Functional Scores
Table 6 presents the functional scores. Cheah et al 5 studied TSA and RTSA procedures. Pre- to postoperatively, the opioid and nonopioid groups showed significant improvements in ASES scores after TSA procedures (P < .003 and P = .0005, respectively). Likewise, pre- to postoperatively, the opioid and nonopioid groups showed significant improvements in ASES scores after RTSA procedures (P < .0001 for both).
Functional Scores a

a Values are expressed as mean ± SD. ASES, American Shoulder and Elbow Surgeons; CMS, Constant-Murley score; RTSA, reverse total shoulder arthroplasty; SANE, Single Assessment Numeric Evaluation; SST, Simple Shoulder Test; TSA, total shoulder arthroplasty; WOOS, Western Ontario Osteoarthritis of the Shoulder.
b Pre- vs postoperative change between groups.
c Median values.
Two studies 17,18 reported CMS, ASES, WOOS, and Single Assessment Numeric Evaluation scores. Postoperative ASES and WOOS scores were significantly improved for both groups (P < .001); however, in the 2 studies, the opioid group had significantly lower preoperative ASES scores (P < .004) and WOOS index (P < .008) than the nonopioid group. The studies similarly showed that the opioid group had significantly lower postoperative CMS (P < .04), ASES scores (P < .01), and WOOS indices (P < .02) when compared with the nonopioid group.
All 5 studies 5,17,18,28,32 commented on the difference in overall ASES score improvement between groups (Table 6). ASES values from Cheah et al 5 were pooled from 59 patients who were followed up for at least 2 years postoperatively. Thompson et al 28 conducted the only study to note a significant difference (P = .01) in overall improvement of median ASES scores, showing that the opioid group had significantly inferior results than the nonopioid group.
Furthermore, all 3 studies 17,18,32 that included CMS outcomes and the 2 studies 17,18 with WOOS indices found no significant differences in overall improvement between groups.
Range of Motion
Four studies 17,18,28,32 measured ROM (Table 7). Morris et al 18 reported that the nonopioid group had significantly greater preoperative forward flexion (P = .005) and abduction (P = .03); however, differences in external rotation were insignificant (P = .6).
Range of Motion a

a Values are reported in degrees as mean ± SD. ROM, range of motion.
b Pre- vs postoperative change between groups.
c Median values.
Thompson et al 28 also found significant improvements in ROM for the opioid group (internal rotation, P = .001; external rotation, P = .004; forward flexion, P = .001) and the nonopioid group (internal rotation, P = .002; external rotation, P = .001; forward flexion, P = .001) from the pre- to postoperative period.
Four studies 17,18,28,32 commented on the difference in overall ROM improvement between groups. All studies stated that there were no significant differences in improvement between groups for forward flexion, external rotation, and abduction (P > .05 for all).
Opioid Consumption
Of the 5 studies, 2 indicated the amount of opioid consumption, 5,32 both in mean morphine equivalents.
Cheah et al 5 found a significant difference in the total mean morphine equivalents consumed as well as the mean morphine equivalents consumed on postoperative days 0, 1, and 2 (P < .001 for all) between groups. The authors stated that the short- and long-acting opioid users consumed significantly higher total amounts than the nonopioid group (149% and 311% more, respectively; P < .001). The total morphine equivalents consumed for the nonopioid group and the short- and long-acting opioid users were 66.9, 111.4, and 208.3, respectively.
Williams et al 32 reported the median duration of opioid therapy, which is the length of time from surgery to the last filled prescription. For the nonopioid group, the median duration of opioid therapy was 1 week (range, 0.0-206.6 weeks), which was shorter than that for the opioid group (12.8 weeks; range, 0.0-55.7 weeks) and was significantly different (P < .05). The authors also reported that the opioid group received 1.91 times (95% CI, 1.31-278) more opioids over a postoperative course of treatment that was 2.73 times (95% CI, 1.62-4.59) longer than the nonopioid group.
Complications, Revisions, and Failures
Two of 5 studies 5,28 compared complication rates between the groups. Cheah et al 5 reported a 7.2% complication rate in the opioid group as compared with a 7.3% complication rate in the nonopioid group (P > .99). Additionally, the reoperation rate was 2.2% in the opioid group and 0.8% in the nonopioid group (P = .6).
Thompson et al 28 reported a complication rate of 14% in the opioid group and 2% in the nonopioid group (P = .05).
Discussion
Preoperative opioid use has been studied in the context of total knee and hip arthroplasties, and patients who took opioids before either procedure experienced worse overall pain and functional benefits than patients who did not. 10,21 Similarly, our study showed that preoperative opioid users had lower absolute functionality scores and ROM as well as higher pain scores after shoulder surgery. Differences in improvement between opioid and nonopioid groups were not statistically significant, possibly because of the heterogeneity of the studies.
Although 2 of our studies 5,28 contradicted each other with respect to preoperative opioid use and increased complication rate, the current literature does indicate that preoperative opioid use may increase complication rates. Sing et al 25 showed that preoperative opioid use in total joint arthroplasties led to longer hospital stays and discharge to facility and increased incidence of perioperative complications within 30 days. Ben-Ari et al 1 also showed that patients who underwent revision total knee arthroplasty within 1 year were more likely to have taken opioids preoperatively. Furthermore, Blevins et al 3 noted that preoperative opioid users had a 35% higher chance of surgical site infection and a 44% higher chance of surgical revision.
Last, the results of our study demonstrated that preoperative opioid users also consumed significantly higher amounts of opioids after shoulder surgery and required longer opioid therapy. As previously mentioned, postoperative opioids are being prescribed at rates approximately 3 times more than needed after shoulder surgery. 13,23 Additionally, unused and undisposed opioids are sources of diversion and contribute to their nonmedical usage and possibly death from abuse. 2,13
Opioid tolerance can arise in the context of overprescribing and extended therapy. Tolerance is the result of the mechanisms of opioid-induced hyperalgesia (OIH) as well as its effects at the mu opioid receptor. The mechanisms of OIH may be the reason behind opioid tolerance and why some patients require prolonged opioid therapy. Furthermore, mu opioid receptors express high levels of genetic variability, suggesting that patients must be analyzed and counseled on the effects of opioids on a case-by-case basis. Sex may also be a factor in the differences seen with OIH. 24 Although addiction happens to a small percentage of opioid users, superimposing factors such as prolonged therapy, repeated administration, and increasingly higher dosage can precipitate the insidious effects of addiction. 30 Keeping these factors in mind when prescribing opioids is vital for patient safety and satisfaction.
Recent studies that did not meet inclusion criteria for this review analyzed the effect of preoperative opioid use on increased opioid consumption over extended postoperative periods. In 2 studies, Khazi et al 11,12 examined this effect in patients undergoing TSA, RTSA, and shoulder stabilization surgery. The incidence of opioid use at 1 year postoperatively was significantly higher in patients who used opioids 3 months before surgery as compared with those who did not (P < .0001 in both studies). Both studies also identified several factors, including fibromyalgia, obesity, and age <65 years, that conferred increased risk of prolonged postoperative opioid use. Furthermore, Nicholson et al 19 constructed a model to preoperatively predict prolonged opioid use after shoulder surgery. The study identified 10 risk factors that were significantly associated with prolonged opioid use, of which preoperative opioid use and insurance type were the strongest.
Some progress has been made toward thwarting the opioid crisis, especially within the orthopaedic community. With preoperative education, significant decreases in the number of narcotic pills necessary to prescribe have been shown. Syed et al 27 placed patients into a randomized group that received formal education detailing recommended postoperative opioid usage, adverse effects, dependence, and addiction via a 2-minute narrated video and handout outlining the risks of narcotic overuse and abuse. This particularly affected preoperative opioid users because these patients were 6.8 times more likely to stop narcotics at their 3-month follow-up visit as compared with the control group (odds ratio, 6.80; 95% CI, 1.57-29.43; P = .008). On a different front, the importance of multimodal pain control models, best-practice prescribing strategies, and consensus protocols by providers has shown promise in abating the opioid crisis. 35 Welton et al 31 characterized the current pain management strategies used peri- and postoperatively among orthopaedic shoulder surgeons. Peri- and postoperatively, >90% of survey responders reported using a standard pain management regimen. After TSA, short-acting narcotics were prescribed by 93.6% of surgeons, but long-acting narcotics were prescribed by only 13.6%. Also, 44.2% of surgeons stated that they treated their patients’ pain for 12 weeks before providing a referral to a pain-management specialist, while 31.5% did so for 6 weeks. With all of this in mind, future efforts focused on exploring patient education and proper drug disposal while creating standardized educational protocols will be beneficial. Furthermore, because most shoulder surgeons already utilize standard pain-management regimens, addressing the efficacy of these protocols and exploring improvements can be important parts of an effective strategy to combat the opioid crisis.
Limitations
The strengths of this study include a comprehensive systematic review performed by 2 independent reviewers. The limitations should also be noted. All of our studies are retrospective level 3 and level 2, which means a risk of selection bias. However, the quality of study methodology is fair, as measured by the MCMS. Additionally, 3 studies 5,17,18 reporting means did not perform tests for normality of their continuous data; thus, the inferences drawn may not be reliable. All of the studies varied in surgical technique, reported outcomes, and measured complications. Important to note is the variance in comorbidities presented in the patient demographics. Moreover, chronic back pain and depression were statistically significant comorbidities associated with preoperative opioid use according to Williams et al 32 and Morris et al. 18 In future studies, controlling for these variables will be important in exploring the effect of these commonly chronic conditions on preoperative opioid use.
Conclusion
The available data suggest that preoperative opioid use before various shoulder surgical procedures negatively affects postoperative pain and functionality. Although the opioid group showed significantly lower scores, the groups experienced similar improvements. Preoperative opioid use may also lead to greater and prolonged postoperative opioid requirement.
Footnotes
Final revision submitted October 21, 2020; accepted November 24, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: E.C.M. has received research support from Arthrex, Biomet, Breg, Mitek, Ossur, Smith & Nephew, and Stryker; consulting fees from Biomet, DePuy/Medical Device Business Services, and Zimmer Biomet; speaking fees from Arthrex; and royalties from Elsevier and Zimmer Biomet. A.J.S. has received education payments from Gemini Mountain Medical and consulting fees from Medacta. R.M.F. has received grant support from Arthrex; education payments from Arthrex, Medwest, and Smith & Nephew; and speaking fees from Arthrex. J.T.B. has received research support from Biomet; consulting fees from DJO, Smith & Nephew, and Encore Medical; and royalties from Shukla Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
