Abstract
Background:
Little is known about sex-based differences in anterior cruciate ligament (ACL) tissue quality in vivo or the association of ACL size (ie, volume) and tissue quality (ie, normalized signal intensity on magnetic resonance imaging [MRI]) with knee anatomy.
Hypothesis:
We hypothesized that (1) women have smaller ACLs and greater ACL normalized signal intensity compared with men, and (2) ACL size and normalized signal intensity are associated with age, activity levels, body mass index (BMI), bicondylar width, intercondylar notch width, and posterior slope of the lateral tibial plateau.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
Knee MRI scans of 108 unique ACL-intact knees (19.7 ± 5.5 years, 62 women) were used to quantify the ACL signal intensity (normalized to cortical bone), ligament volume, mean cross-sectional area, and length. Independent t tests were used to compare the MRI-based ACL parameters between sexes. Univariate and multivariate linear regression analyses were used to investigate the associations between normalized signal intensity and size with age, activity levels, BMI, bicondylar width, notch width, and posterior slope of the lateral tibial plateau.
Results:
Compared with men, women had significantly smaller mean ACL volume (men vs women: 2028 ± 472 vs 1591 ± 405 mm3), cross-sectional area (49.4 ± 9.6 vs 41.5 ± 8.6 mm2), and length (40.8 ± 2.8 vs 38.1 ± 3.1 mm) (P < .001 for all), even after adjusting for BMI and bicondylar width. There was no difference in MRI signal intensity between men and women (1.15 ± 0.24 vs 1.12 ± 0.24, respectively; P = .555). BMI, bicondylar width, and intercondylar notch width were independently associated with a larger ACL (R 2 > 0.16, P < .001). Younger age and steeper lateral tibial slope were independently associated with shorter ACL length (R 2 > 0.03, P < .04). The combination of BMI and bicondylar width was predictive of ACL volume and mean cross-sectional area (R 2 < 0.3). The combination of BMI, bicondylar width, and lateral tibial slope was predictive of ACL length (R 2 = 0.39). Neither quantified patient characteristics nor anatomic variables were associated with signal intensity.
Conclusion:
Men had larger ACLs compared with women even after adjusting for BMI and knee size (bicondylar width). No sex difference was observed in signal intensity, suggesting no difference in tissue quality. The association of the intercondylar notch width and lateral tibial slope with ACL size suggests that the influence of these anatomic features on ACL injury risk may be partially explained by their effect on ACL size.
Registration:
NCT02292004 and NCT02664545 (ClinicalTrials.gov identifier).
It is well established that female athletes have higher anterior cruciate ligament (ACL) tear rates than their male counterparts. 2,3,24 Causes of such disparity are likely complex and remain largely a topic of debate. It has been hypothesized that the sex bias in knee injury rates may be due to differences in neuromuscular control, ligamentous laxity, and/or anthropometric parameters such as intercondylar notch size and ACL size. 29,48 To identify specific risk factors for ACL injury, particularly ones that may contain a sex bias, a large body of literature describing the native structure and function of the ACL within both sexes has been published. Included in this has been a focus on geometric parameters including the ACL length and width, 17 cross-sectional area 18 and volume, 61 and the impact of these factors on future injury to the ACL. These geometric properties have been shown to display variation by sex. 13,28 Chandrashekar et al 13 reported that women had smaller absolute ACL volume and cross-sectional area than men, and Anderson et al 1 found that ACL width and cross-sectional area were smaller in female high school basketball athletes compared with their male counterparts.
Signal intensity—a measure of ligamentous uniformity and structural integrity on magnetic resonance imaging (MRI)—is increasingly utilized in clinical studies to assess the maturity of ACL grafts after ACL reconstruction in humans 22,57 and more recently to evaluate ligament quality after biologically enhanced ACL repair techniques. 35,45 MRI-based ligament signal intensity has been used as a surrogate for tissue quality (eg, collagen organization and tensile strength), with lower signal (ie, a darker appearance) indicative of a more organized and mature ligament. 4 –6,9 –11,21 In contrast to studied sexual dimorphisms in ACL size, to our knowledge, there has been no study published that looked specifically at signal intensity of intact native ACL and the presence of any baseline differences between men and women. A better understanding of sex-based differences in ACL tissue quality may help better explain the higher prevalence of ACL injury in women.
The objectives of this study were to use MRI and 3-dimensional image analysis to determine whether there are anatomic and signal intensity differences between sexes, and to establish whether the sex differences between ACL size and signal intensity are related to age, activity levels, body size, knee size, and knee anatomy. We hypothesized that (1) women have smaller ACLs and greater normalized signal intensity of the ACL compared with men, and (2) the ACL size and normalized signal intensity are associated with age, activity levels, body mass index (BMI), bicondylar width (a measure of knee size), intercondylar notch width, and posterior slope of the lateral tibial plateau, which are thought to be major risk factors for ACL injuries. 7,61
Methods
Study Patients
The study protocol received institutional review board approval. A total of 108 participants (19.7 ± 5.5 years, 62 women) across 2 clinical trials (NCT02292004 and NCT02664545) investigating a novel biologic device for acute ACL tears 43,44 underwent contralateral (uninjured) knee MRI between 12 and 24 months postoperatively as part of the trial. Patients were excluded from this analysis if they had previously undergone ACL surgery on the contralateral (uninjured) side. The intact status of the contralateral ACLs was confirmed by a board-certified musculoskeletal radiologist (K.E.). Data regarding the BMI of the patients were also extracted at the time of imaging.
Imaging Outcomes
A 3.0-T scanner (Tim Trio; Siemens) and a 15-channel knee coil (Siemens) were used to obtain a 3-dimensional Constructive Interference in Steady State (CISS) sequence (repetition time/echo time, 14/7 ms; flip angle, 35°; 16-cm field of view; slice × frequency × phase, 100 × 384 × 284). Contralateral intact ACLs were manually segmented from the sagittal CISS image stacks in Mimics (version 17.0; Materialize) to measure the ligament volume and length, which were then used to calculate the mean cross-sectional area (volume/length). 34,45 The ligament signal intensity was also calculated as the average grayscale value across the ligament voxels for each patient normalized to the patient-specific grayscale value of the femoral cortical bone. 34,45 The bicondylar width, intercondylar notch width, and posterior slope of the lateral tibial plateau were measured from the sagittal and coronal views using MR image viewing software (Osirix Viewer version 8.5; Pixmeo SARL). The width of the femoral notch was measured in the coronal plane, parallel to a line along the most inferior aspects of the femoral condyles. 47 The measurement was done at the middle of the ACL attachment. 61 The same coronal slice was also used to measure bicondylar width as a surrogate for overall knee size, as commonly used in ACL literature, in contrast to tibial plateau width, which has been used in the total knee arthroplasty field. The posterior slope of the lateral tibial plateau was measured using the techniques described by Hudek et al. 32 Briefly, a central sagittal slice, in which the tibial attachment of the posterior cruciate ligament, the intercondylar eminence, and the anterior and posterior tibial cortices appeared in a concave shape, was selected. Then, 1 cranial circle (touching the anterior, posterior, and cranial tibial cortex) and 1 caudal circle (touching the anterior and posterior cortex) were fitted to the tibial head. The line connecting the center of the 2 circles was defined as the longitudinal tibial axis. 32 This technique has been shown to be highly repeatable and independent of proximal tibial length covered in the MRI scan. 42 The lateral tibial slope was measured in a sagittal slice at the center of the lateral tibial plateau as the angle between a line that joined the peak points on the anterior and posterior bony rims of the plateau and a line perpendicular to the longitudinal axis of the tibia. 25 All the anatomic measurements and ACL segmentation techniques are shown in Figure 1.
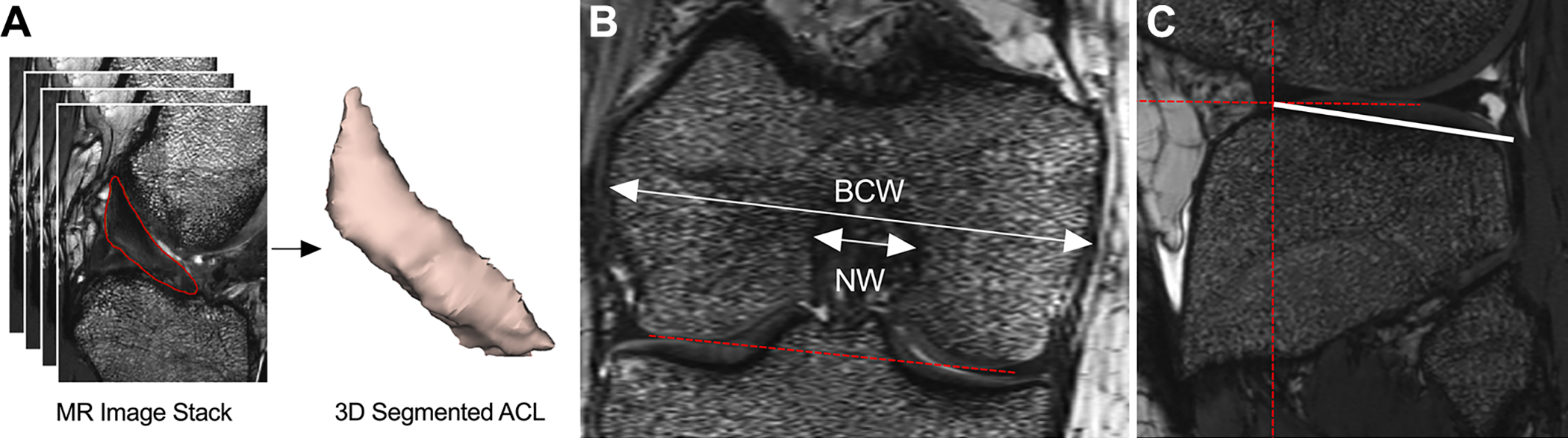
(A) Three-dimensional segmentation of the anterior cruciate ligament (ACL) from magnetic resonance (MR) image stacks, and measurement techniques used to quantify (B) bicondylar and notch widths (BCW and NW) and (C) posterior slope of the lateral tibial plateau. The dashed red lines indicate the line passing through the inferior aspects of the femoral condyles (B) and the reference lines to measure the posterior slope of the lateral tibial plateau (C).
The segmentations and measurements were performed by an experienced examiner (A.M.K.) with more than 5 years of experience in 3-dimensional quantitative analysis of the ACL from MRI. To assess the reliability of the measurements, a randomly selected subset of 25 patients were reanalyzed later by the same examiner and a second independent examiner (D.M.), both of whom were blinded to the patients’ identity. Variance estimates were then used to compute the intraclass correlation coefficients (ICCs) for within- and between-examiner reliablity. 19
Statistical Analysis
Two-sample t tests were used to compare the ACL parameters between men and women. Univariate linear regression analyses were performed to assess the associations between age, BMI, preoperative Marx Activity Score (0-16, with higher scores indicating higher cutting and pivoting activity levels), 12 bicondylar width, intercondylar notch width, and lateral tibial slope to quantified features of the ACL: ACL signal intensity, length, mean cross-sectional area, and volume. Additionally, stepwise regression analyses were used to identify the best predictive models for each ACL imaging outcome. For the stepwise procedure, all patient and anatomic variables (ie, sex, age, BMI, bicondylar and notch width, and tibial slope) were considered candidates for inclusion in the model. Data are presented as mean ± standard deviation. Two-sided P values are reported and were considered significant when P < .05.
RESULTS
Representative images of ACLs for 1 female and 1 male patient are shown in Figure 2. Compared with men, women had a smaller ACL volume (2028 ± 472 vs 1591 ± 405 mm3, respectively; P < .001), mean cross-sectional area (49.4 ± 9.6 vs 41.5 ± 8.6 mm2, respectively; P < .001), and length (40.8 ± 2.8 vs 38.1 ± 3.1 mm3, respectively; P < .001) (Figure 3). Compared with men, women had a smaller ACL cross-sectional area even after normalizing to BMI (P = .002) or bicondylar width (P = .015). However, there were no differences in ACL volume after normalizing to intercondylar notch width (P = .392). There was no difference in ACL normalized signal intensity between men and women (1.15 ± 0.24 vs 1.12 ± 0.24, respectively; P = .555) (Figure 3).

Representative images of the anterior cruciate ligament for a (A) female and a (B) male patient.

Sex-based differences in anterior cruciate ligament (ACL) size (volume, mean cross-sectional area, and length) and normalized signal intensity. Bars are median and P values are 2-sided.
Older age was independently associated with longer ACL (R 2 = 0.04, P = .038), while there were no significant associations between age and other quantified ACL metrics (Figure 4). Higher Marx Activity Score was not significantly associated with either ACL size (volume, mean cross-sectional area, and length) or normalized signal intensity (R 2 < 0.3, P > .1) (Figure 5). Higher BMI was independently associated with a larger ACL (volume, mean cross-sectional area, and length) (R 2 > .1, P < .01). There was no significant association between normalized signal intensity and BMI (Figure 6). A wider femoral condyle was independently associated with a larger ACL (R 2 > .1, P < .001). There was no significant association between normalized signal intensity and bicondylar width (Figure 7). A wider intercondylar notch was independently associated with a larger ACL (R 2 > .1, P < .01). There was no significant association between normalized signal intensity and intercondylar notch width (Figure 8). Steeper lateral tibial slope was associated with a shorter ACL (R 2 = .07, P = .012). There were no significant associations between normalized signal intensity, cross-sectional area, or volume and lateral tibial slope (Figure 9).

Univariate regression analysis demonstrating the linear relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with age. Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.
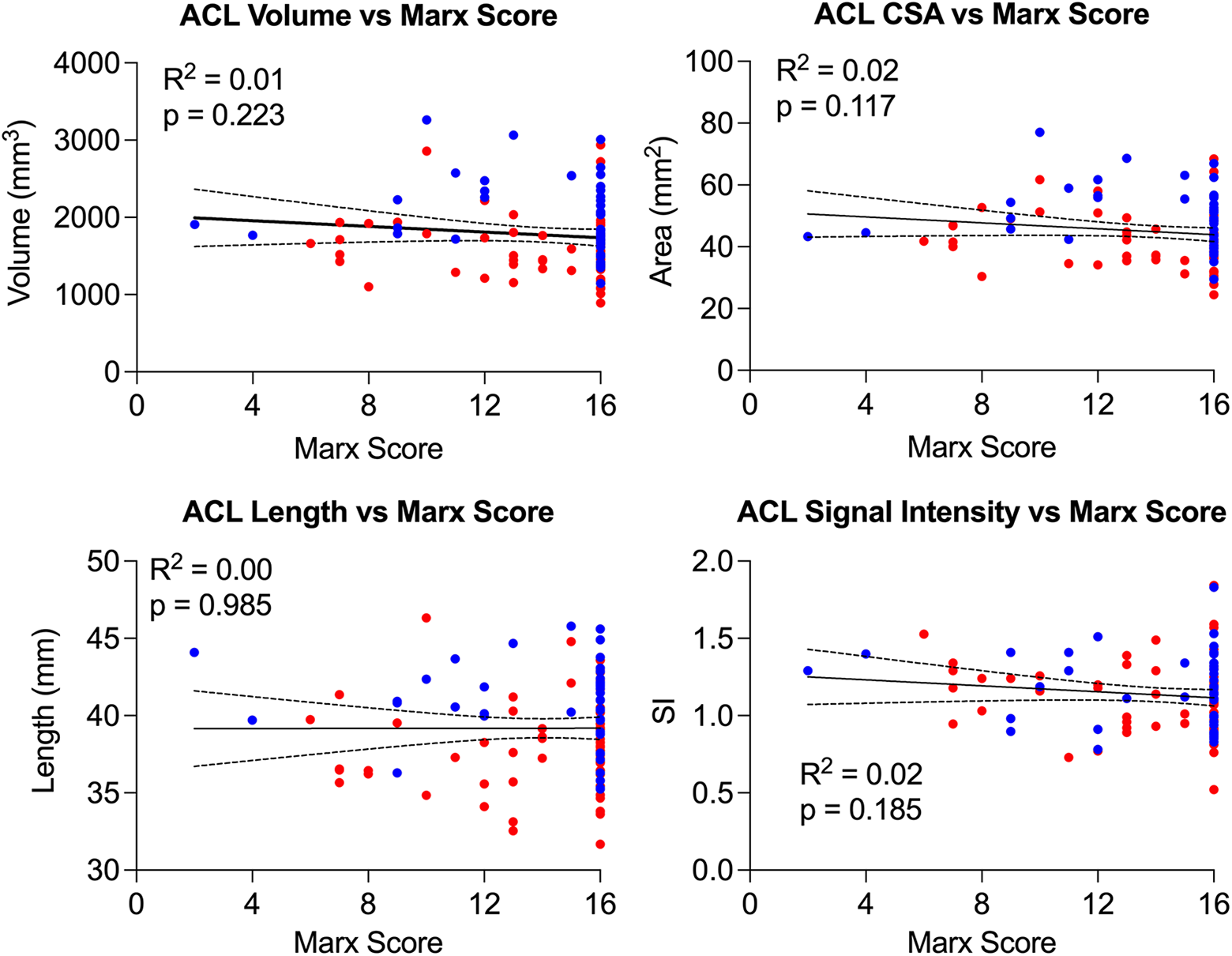
Univariate regression analysis demonstrating the linear relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with Marx Activity Score. Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.
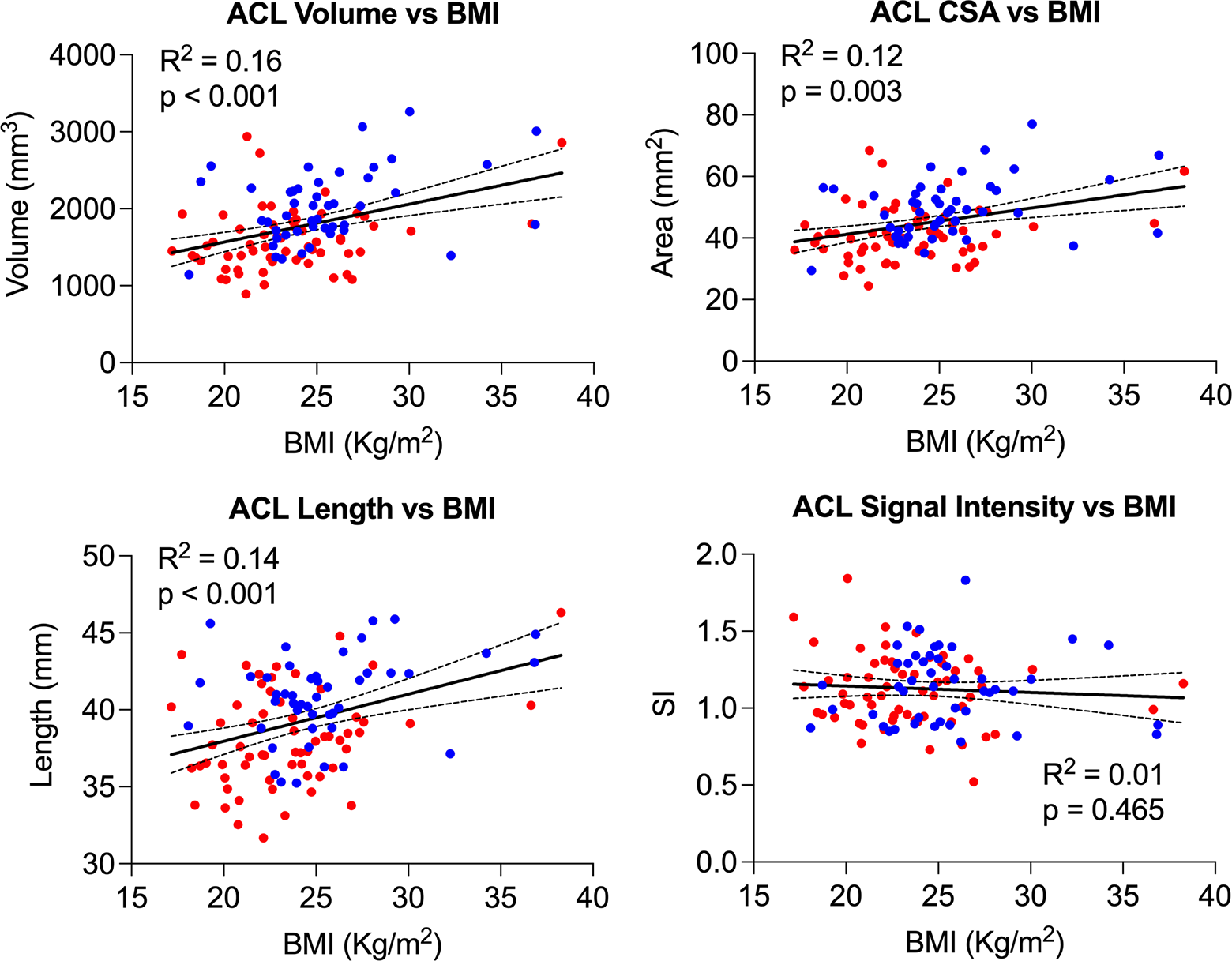
Univariate regression analysis demonstrating the linear relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with body mass index (BMI). Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.

Univariate regression analysis demonstrating the linear relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with bicondylar width (BCW). Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.

Univariate regression analysis demonstrating the linear relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with intercondylar notch width (NW). Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.
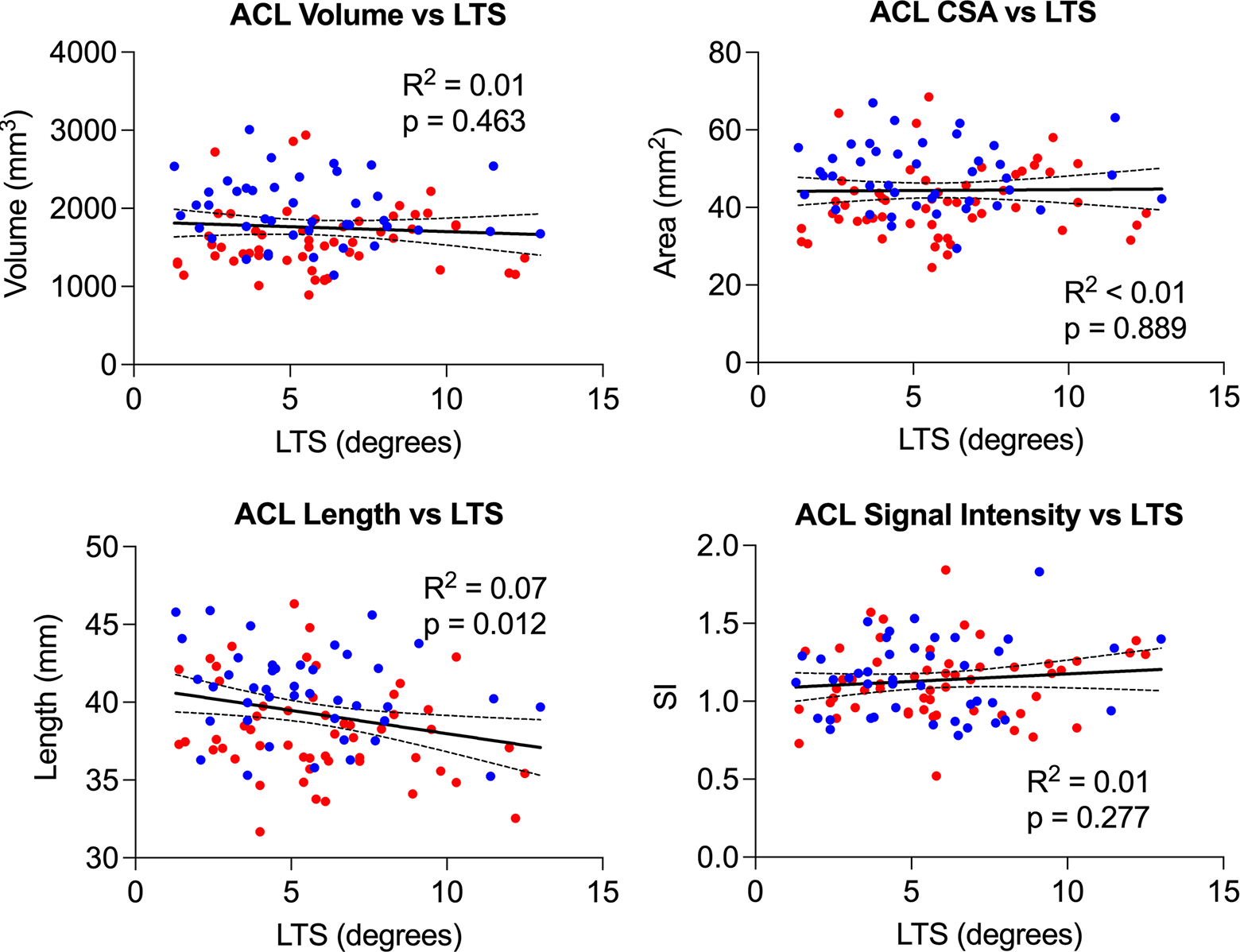
Univariate regression analysis demonstrating the inverse relationship of anterior cruciate ligament (ACL) size (volume, mean cross-sectional area [CSA], and length) and signal intensity with steeper lateral tibial slope (LTS). Red dots represent women and blue dots men. The regression line (solid line) and corresponding 95%CI (dashed lines) are shown for each univariate regression analysis.
The results of the stepwise regression models for each ACL imaging outcome are presented in Table 1. The derived prediction model for ACL volume included only the BMI and bicondylar width, which explained 29% variability in ACL volume. The model predicting the ACL mean cross-sectional area included BMI and bicondylar width, which explained the 19% variability in ACL mean cross-sectional area. The combination of BMI, bicondylar width, and lateral tibial slope produced the predictive model of ACL length score and explained the 39% variability in this outcome. None of the patient and anatomic variables were able to predict ACL normalized signal intensity.
Stepwise Regression Analysis Indicating Significant Predictors of ACL Imaging Outcomes in a Multivariate Model a

a Only the significant predictors in the final models are shown. ACL, anterior cruciate ligament; BCW, bicondylar width; BMI, body mass index; CSA, cross-sectional area; LTS, lateral tibial slope.
Strong ICC values were observed within and between examiners for the quantified imaging outcomes (Table 2).
ICCs for Reliability of Imaging Measurements a

a ACL, anterior cruciate ligament; ICC, intraclass correlation coefficient.
Discussion
Consistent with previous literature, 1,13 men had larger ACLs compared with women in all measured parameters: volume, mean cross-sectional area, and length. Interestingly, these sex-based differences remained significant even after adjusting for body size (BMI) or knee size (bicondylar width) while there were no sex differences in ACL size after normalizing to intercondylar notch width. No sex difference, however, was observed in normalized signal intensity of the intact ACLs. Furthermore, we observed strong associations between BMI, bicondylar width, and intercondylar notch width with ACL size: volume, cross-sectional area, and length. We also saw inverse associations between younger age and steeper lateral tibial slope with ACL length. However, neither of the quantified patient and anatomic parameters (age, activity levels, BMI, bicondylar width, intercondylar notch width, lateral tibial slope) were associated with the normalized signal intensity of the ACL.
The significant sex differences in ACL injury risk have motivated numerous efforts to identify risk factors responsible for this discrepancy. While several studies 13,28,30 have been focused on sex differences in ACL morphology, little is known on sex differences in ACL structural properties. A normalized signal intensity of ACL tissue from MRI has been reported to be a useful method to evaluate ACL grafts and predict important clinical outcomes after ACL reconstruction. 38,39,50,51,56 It has also recently been shown to be useful in assessing ACL healing after enhanced ACL restoration, 34,45 in which a bridging collagen scaffold is used to promote native healing of the torn ACL. Normalized signal intensity has also been shown to be negatively correlated with experimentally measured structural and histological properties of the repaired or reconstructed ACL in a porcine model. 9,49 A higher normalized signal intensity is indicative of a less organized and lower-quality ACL. 60 While MRI is commonly used to determine the extent of ACL injury, these studies (cited in this paragraph) highlight the utility of MR-based signal intensity assessment to evaluate ACL quality, which could be used for injury risk assessment. MRI also enables detailed analysis of morphological features that have been repeatedly linked to higher risk of injury 7,26,61 and reinjury 16,37,52,59 as well as inferior outcomes after ACL reconstruction. 35
Consistent with prior studies, 1,13 we showed that female athletes have smaller ACLs compared with their male counterparts. Interestingly, in the current study, no sex difference was observed in normalized signal intensity of intact, uninjured ACLs, suggesting that despite their overall smaller size, ACLs in women have similar baseline tissue quality (assessed on MRI) to those seen in men. While sex differences in hormonal levels (eg, estrogen) have been shown to influence ACL tensile strength in small preclinical models, 36,55 their clinical impact on intact ACL tissue is unknown. We therefore surmise that baseline ligament quality plays little role in the ACL injury risk discrepancy between men and women, specifically in relation to macroscopic structural integrity of the tissue. Previous literature has shown that there may be an ultrastructural difference in ACL tissue between men and women. Hashemi et al 27 reported that the average number of collagen fibrils per unit area was significantly larger in men. Therefore, despite this difference in density of collagen of ACL tissue between sexes, likely accounting for the overall size difference, the quality and organization of the tissue reported via MR signal intensity remains similar. While ACL size has been shown to be significantly changed by age, 30 we did not see any correlations between ACL volume and cross-sectional area with age. This was primarily because of the age distribution of the studied patient cohort (Appendix Figure A1), which included age range corresponding to minimal ACL growth. 30
Previous studies 1,13,14,20 have shown little consistency in accounting for bony geometry and body size, which may have influenced overall ACL size. Anderson et al 1 found that ACL cross-sectional area was smaller in women when accounting for body weight, while Jamison et al 33 reported body height was the only predictor of ACL volume within a multifactorial model, which did not factor in body weight or any other anatomic features. Our findings of associations between BMI and bicondylar width with ACL size in both univariate and multivariate settings confirm those prior reports. The finding of the current study that sex differences remain even after adjusting for BMI and bicondylar width suggests that ACL size differences between men and women cannot simply be explained by differences in body/joint size scaling. Instead, it may be a more complex interplay between the intercondylar notch and subsequent ACL development. Consistent with this, we also observed a strong association between intercondylar notch size and overall ACL size. One potential conclusion is that smaller notch size increases ACL injury risk secondary to a smaller ACL. This conclusion is supported by previous reports 17,53,54 of smaller intercondylar notch widths in men and women with torn ACLs and correlations between ACL size and notch width. A recent meta-analysis by Li et al 40 demonstrated that notch width measured via MRI was significantly smaller in patients with an ACL injury. Other studies have reported ACL volume to be an independent predictor; Whitney et al 61 illustrated a smaller ACL volume as an independent predictor of ACL injury, while Chaudhari et al 15 demonstrated that patients with an ACL injury had smaller contralateral ACL volume than controls. This difference remains clinically important as smaller ACLs have reduced resistance to deformation and lower tensile failure loads, causing them to be at a higher risk of tear with greater external forces. 13,27 Furthermore, notchplasty has been used during ACL reconstruction to prevent graft impingement, particularly with hamstring quadruple-bundle autograft, given its large diameter. 31,58 In a recent study, notchplasty was shown to enhance the size of the repaired ACL after bridge-enhanced ACL repair. 45 MRI measurements of the notchplasty size revealed that for every 1 mm of notchplasty performed, the cross-sectional area of the repaired ACL increased by 2.4 mm2 (approximately 4% of the ligament size). 45 This supports our finding of an association between intercondylar notch size and ACL size, which appears to be independent of other anatomic factors.
As a secondary finding within our study, the association between a steeper lateral tibial slope and a shorter ACL demonstrates another complex interaction between 2 established ACL injury risk factors. Lateral tibial slope has gained significant attention in its relation to ACL biomechanics and injury risk. Exact values vary depending on measurement technique and type of imaging used for analysis, but women have been shown to have a steeper lateral tibial slope compared with men. 26 A steeper lateral tibial slope has also been linked to increased risk of ACL injury 7,23,41,46,59 and inferior outcomes after ACL reconstruction, including graft failure. 16,37,52,59 The observed correlation found in our study between a steeper lateral tibial slope and a shorter ACL suggests that at least part of the higher risk profile of patients with a steep lateral tibial slope may be explained by its association with a smaller ACL. The growing consideration for tibial osteotomies to correct such anatomic variants during ACL reconstruction may need to be considered cautiously in patients known to have shorter ACLs, as this invasive bony realignment may not be clinically and/or functionally necessary for a subset of patients whose risk profile is more significantly related to the smaller size of their ACL and not their tibial anatomy.
There are a few limitations to this study to consider. While previous studies have established strong associations between normalized signal intensity of the repaired ACL or reconstructed graft with their mechanical and histological properties, 9,49 such relationships have not been directly established for native ACL tissue. However, considering the similarities between the repaired ACL and the native intact ACL, we expect to see similar associations between intact ACL MRI signal and its structural properties. It is difficult to directly compare signal intensity values across institutions given the dependence on specific hardware for standardization. To address this, we used the same imaging parameters and MRI machine across all study patients (3T Trio; Seimens). In addition, we normalized the ACL signal intensity values to those of the individuals’ specific cortical bone to minimize concerns of variability between scan sessions. 8 Further research is underway to identify and optimize relaxometry methods that may be less reliant on specific hardware, providing greater consistency of MRI measurement across different machines. 4,6,10,11 The MR signal intensity analysis is based on a single time point and is therefore unable to capture potential changes over time. We also do not have direct data on smaller ACL size leading to inferior structural properties, in particular after adjusting for confounders (eg, BMI). Patients included in this study all sustained a unilateral ACL tear. Further biomechanical studies in patients without any ACL injuries are required to assess these observations in patients at lower risk of ACL injury. Finally, similar to any imaging measurements, there are inherent measurement errors. We have tried to minimize these errors by using robust and widely used measurement techniques. We also conducted a reliability assessment that showed strong measurement reliability across all the reported outcomes.
Conclusion
We have shown in this study that at baseline, intact ACL tissue normalized MR signal intensity is the same in men and women. However, ACLs in women are smaller than those of men, primarily because of the smaller knee and intercondylar notch sizes. Collectively, these findings support the smaller ACLs in women as one of the ACL-related contributing factors leading to higher risk of ACL injuries among women. Our study additionally adds evidence regarding the complex relationships between key anatomic features of the knee and ACL size, which may explain why these features have previously been identified as risk factors for ACL injury.
AUTHORS
BEAR Trial Team: Benedikt Proffen, MD; Nicholas Sant, BS; Gabriela Portilla, BA; Ryan Sanborn, BA; Christina Freiberger, BS; Rachael Henderson, BS; Kirsten Ecklund, MD; Yi-Meng Yen, MD, PhD; Dennis Kramer, MD; and Lyle Micheli, MD. All authors are affiliated with the Department of Orthopaedic Surgery, Boston Children’s Hospital, Boston, Massachusetts, USA.
Footnotes
Acknowledgment
We acknowledge the significant contributions of the clinical trial team, including Bethany Trainor. We also acknowledge the contributions of our medical safety monitoring team, Joseph DeAngelis, Peter Nigrovic, and Carolyn Hettrich; our data monitors, Maggie Malsch, Meghan Fitzgerald, and Erica Denhoff; and the clinical care team for the trial patients, Kathryn Ackerman, Alyssa Aguiar, Judd Allen, Michael Beasley, Jennifer Beck, Dennis Borg, Jeff Brodeur, Stephanie Burgess, Melissa Christino, Sarah Collins, Gianmichel Corrado, Sara Carpenito, Corey Dawkins, Pierre D’Hemecourt, Jon Ferguson, Michele Flannery, Casey Gavin, Ellen Geminiani, Stacey Gigante, Annie Griffin, Emily Hanson, Elspeth Hart, Jackie Hastings, Pamela Horne-Goffigan, Christine Gonzalez, Meghan Keating, Elizabeth KillKelly, Elizabeth Kramer, Pamela Lang, Hayley Lough, Chaimae Martin, Michael McClincy, William Meehan, Ariana Moccia, Jen Morse, Mariah Mullen, Stacey Murphy, Emily Niu, Michael O’Brien, Nikolas Paschos, Katrina Plavetsky, Bridget Quinn, Shannon Savage, Edward Schleyer, Benjamin Shore, Cynthia Stein, Andrea Stracciolini, Dai Sugimoto, Dylan Taylor, Ashleigh Thorogood, Kevin Wenner, Brianna Quintiliani, and Natasha Trentacosta. We thank the perioperative and operating room staff and the members of the Department of Anesthesia, who were extremely helpful in developing the perioperative and intraoperative protocols. We also acknowledge the efforts of other scaffold manufacturing team members, including Gabe Perrone, Gordon Roberts, Doris Peterkin, and Jakob Sieker. We are grateful for the study design guidance provided by the Division of Orthopedic Devices at the Center for Devices and Radiological Health at the U.S. Food and Drug Administration under the guidance of Laurence Coyne and Mark Melkerson, particularly the efforts of Casey Hanley, Peter Hudson, Jemin Dedania, Pooja Panigrahi, and Neil Barkin. Lastly, we are grateful to the patients and their families who participated in this study; their willingness to participate in research that may help others in the future inspires all of us.
Final revision submitted August 27, 2021; accepted September 16, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study received funding support from the Translational Research Program at Boston Children’s Hospital, the Children’s Hospital Orthopaedic Surgery Foundation, the Children’s Hospital Sports Medicine Foundation, Boston Children’s Hospital Faculty Council, the Football Players Health Study at Harvard University (funded by a grant from the National Football League Players Association), and the National Institutes of Health and the National Institute of Arthritis and Musculoskeletal and Skin Diseases (grants R01-AR065462 and R01-AR056834). The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Medical School, Harvard University or its affiliated academic healthcare centers, the National Football League Players Association, Boston Children’s Hospital, or the National Institutes of Health. M.M.M. is a founder, paid consultant, and equity holder in Miach Orthopaedics, which was formed to work on upscaling production of the BEAR scaffold. M.M.M. has also received honoraria from the Musculoskeletal Transplant Foundation. B.C.F. is a paid associate editor for The American Journal of Sports Medicine and the spouse of M.M.M. with the inherently same conflicts. A.M.K. is a paid consultant for Miach Orthopaedics. D.K. and Y.-M.Y. have received education payments from Kairos Surgical. B.P. has manufactured the scaffolds used in the trials at Boston Children’s Hospital and is a paid consultant and equity holder in Miach Orthopaedics. N.S. has manufactured scaffolds used in the trials at Boston Children’s Hospital and is a paid consultant for Miach Orthopaedics. M.M.M., A.M.K., B.P., and N.S. maintained a conflict-of-interest management plan that was approved by Boston Children’s Hospital and Harvard Medical School during the conduct of the trial, with oversight by both conflict-of-interest committees and the institutional review board of Boston Children’s Hospital. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Boston Children’s Hospital (protocol No. IRB-P00012985).
