Abstract
Background:
Although tranexamic acid (TXA) has been shown to reduce bleeding in joint replacement procedures, its effectiveness for anterior cruciate ligament reconstruction (ACLR) has not been widely reported.
Purpose:
To evaluate the effectiveness of TXA to reduce postoperative hemarthrosis and improve clinical outcomes after ACLR.
Study Design:
Systematic review; Level of evidence, 2.
Methods:
A systematic review of the literature following the PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) was performed; literature retrieval was carried out using the MEDLINE, Embase, and Cochrane Library electronic databases. The inclusion criteria were comparative studies in English that reported the administration of intravenous or intra-articular TXA versus other modalities or placebo in patients undergoing ACLR.
Results:
Six studies comprising 418 patients who were treated with TXA were included. Heterogeneity among studies did not allow for the pooling of data. Five studies showed decreased drainage volume in the first 24 or 48 hours postoperatively as compared with control (ACLR with no TXA). Four studies showed lower hemarthrosis grades and visual analog scale scores in TXA versus control in the early postoperative period, although this difference was not evident at 4 weeks postoperatively. No studies showed differences in infection, deep venous thrombosis, or adverse events between the TXA and control groups.
Conclusion:
The current best available evidence suggests that TXA administration at the time of ACLR results in decreased intra-articular bleeding (measured using a drainage system), hemarthrosis grade, and pain when compared with control.
Keywords
In 1988, Small 36 reported a low complication rate (1.68%) in arthroscopic surgery; however, 60.1% of these complications were hemarthrosis. Hemarthrosis can cause postoperative pain and swelling, reduce patient satisfaction, increase the risk of infection and cartilage toxicity, and delay recovery and resumption of exercise. 16,17,32 For severe joint hemarthrosis, knee arthrocentesis needs to be performed for the alleviation of pain and capsular tension and to reduce joint effusion. Notwithstanding all these benefits, arthrocentesis is an invasive procedure, which can also bring pain and carries a small risk of septic arthritis. 29
To alleviate the effect of hemarthrosis, clinicians have been testing various ways to control postoperative bleeding. Of all the possible methods, tourniquet has been used since it was first proposed in 1904, for it can notably improve the surgical visual field, reduce intraoperative bleeding, and shorten the time of operation. 39 However, Nakayama and Yoshiya 30 found that using a tourniquet can increase the drainage volume of the articular cavity as compared with not using a tourniquet. The necessity of a postoperative drainage system also remains controversial. Several randomized controlled trials (RCTs) 9,25 on anterior cruciate ligament reconstruction (ACLR) and the systematic review conducted by Clifton et al 7 concluded that using a drain after ACLR does not reduce postoperative bleeding. Tatari et al 37 came to the same conclusion after studying the effect of a drainage system after knee arthroscopic surgery; that is, using a drain did not mitigate the risk of hemarthrosis.
Tranexamic acid (TXA) is a kind of synthetic analog of lysine, which was developed in 1957, with an antifibrinolysis effect. 31 It acts by reversibly blocking the lysine binding sites of plasminogen, thus preventing plasmin activation and, in consequence, affecting the formation of the fibrin degradation product. 22 TXA has been widely used for hemostasis during perioperative periods, such as cardiac surgery, liver transplantation surgery, and major orthopaedic procedures. 10,18 RCTs have revealed that intravenous or regional injection of TXA during knee arthroplasty could greatly reduce the occurrence rate of bleeding and anemia during the perioperative period and lower blood transfusion needs. 1,2,15,34,40 In a systematic evaluation, Alshryda et al 2 indicated that the administration of TXA during the perioperative period of knee arthroplasty can cut down the amount of bleeding by 245 mL.
After performing a comparison of bleeding amount after ACLR between patients who received TXA and those who did not, Karaaslan et al 19,20 concluded that injecting TXA could considerably reduce postoperative bleeding, decrease postoperative pain, and enhance early postoperative outcomes. Some studies 26,33,38 have raised concerns, declaring that it might influence the safety of cartilage, since a high concentration of TXA administered intra-articularly has been proven to have toxic effects on human and animal chondrocytes in vitro. However, Parker et al 33 demonstrated that the toxic effects were dose dependent and that keeping the TXA concentration between 10 and 20 mg/mL would ensure safe application. Furthermore, there is a lack of toxicity to chondrocytes in vitro when TXA is given at normal doses intra-articularly at the time of surgery. 4,13
The current study systematically evaluated the published literature on the administration of TXA during ACLR, as well as the efficacy and safety of TXA, and analyzed whether it can reduce intra-articular bleeding and improve clinical results in the early postoperative period.
Methods
Literature Search
This review was designed and performed according to the PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) and AMSTAR guidelines (assessing the methodological quality of systematic reviews). 28 The online MEDLINE, Embase, and Cochrane Library databases were searched for all English-language studies to May 1, 2021. Two independent reviewers (Y.J. and Y.S.) searched each database using the following strategy: (“anterior cruciate ligament” OR ACL) AND (“tranexamic acid” OR TXA). A manual search for references of included articles was also performed to ensure that no eligible studies were missed.
Inclusion and Exclusion Criteria
The inclusion criteria were as follows: (1) RCT or prospective comparative study that evaluated the effect of TXA on ACLR; (2) studies that reported estimated blood loss, hemarthrosis grade, visual analog scale (VAS) score, or other clinical outcome; and (3) studies published in English. The exclusion criteria were (1) retrospective comparison studies, review articles, and basic science articles and (2) studies that did not report required data of interest.
Assessment of Risk of Bias
The risk of bias was assessed for RCTs using the Cochrane Collaboration Risk of Bias Tool, 14 which consists of the following items: random sequence generation, allocation concealment, blinding of patients and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. Each study was rated as having low, unclear, or high risk of bias for each item. The risk-of-bias summary was generated using Cochrane Review Manager 5.3 (Cochrane Collaboration, Nordic Cochrane Centre).
Data Collection
The following basic data were collected from each study by 2 independent reviewers (Y.J. and Y.S.): first author, publication year, study design, level of evidence, sample size and mean age of each group, and follow-up period. The following characteristics of administration were also collected: the dosage, route, and timing of TXA and the control group. The heterogeneity of administration protocols among the studies did not allow for pooling of data, and the clinical outcomes are summarized in a narrative fashion: blood loss, hemarthrosis grade, VAS, range of motion (ROM), functional outcomes, and complications.
Results
Literature Search and Study Details
A PRISMA flow diagram to identify studies for inclusion is illustrated in Figure 1. A total of 72 studies were identified from the initial search, and 42 duplicates were removed. After application of inclusion and exclusion criteria, only 6 comparative studies were identified in this systematic review: 4 RCTs, 11,20,21,24 1 prospective cohort study, 6 and 1 abstract. 19 The abstract that was included was of a prospective randomized study that documented the TXA administration protocol and drain output volume. 19 Although this abstract had a similar authorship to 1 of the studies, 20 the described administration routes, dosage, and outcome measurements were different. Thus, both were included in this review.

Flow diagram of study selection. TXA, tranexamic acid.
The main characteristics of the 6 studies in the systematic review are presented in Table 1. A total of 775 patients were included: 418 in the TXA group and 357 in the control group. The administration protocols are listed in Table 2. The exclusion criteria of studies were mainly coagulation (thrombotic disease) or bleeding disorders, preoperative anticoagulation therapy, renal disorder or insufficiency, revision ACLR, multiple-ligament injury, history of knee surgery on the affected knee, and significant preoperative pain (VAS score >5). Chiang et al 6 did not describe the exclusion criteria of patients. Two studies 11,20 administered TXA via intravenous infusion; 2 studies, 6,21 via intra-articular injection; 1 study, 19 via both routes; and 1 study, 24 via either intravenous or intra-articular injection. Four studies 11,19,20,24 had a saline solution control group, while 2 studies 6,21 had no control group.
Characteristics of the Included Studies a

a HT, hamstring tendon; IA, intra-articular; IV, intravenous; LOE, level of evidence; NG, not given; PCS, prospective comparative study; RCT, randomized controlled trial; TXA, tranexamic acid.
Administration Protocols of Included Studies a

a IA, intra-articular; IKDC, International Knee Documentation Committee; IV, intravenous; ROM, range of motion; TXA, tranexamic acid; VAS, visual analog scale.
Risk-of-Bias Assessment
The risk of bias of the 4 RCTs 11,20,21,24 is shown in Figure 2. Two studies 20,24 did not clearly describe the procedures of allocation concealment; thus, they were rated as having an unclear risk of selection bias. One study 21 that did not cite the postoperative Lysholm score was rated as having a high risk of reporting bias. The follow-up period in 3 studies 6,20,24 was just 4 weeks, which might not be enough to allow examination of functional outcomes, ROM, and complications of TXA administration, with a high risk of other bias.

Risk of bias of the randomized controlled trials.
Estimated Blood Loss
Five studies 6,11,19,20,24 showed statistically significant decreases in drain output with TXA administration, as opposed to without it, within the first 24 or 48 hours. Only the study by Lee et al 21 did not show a significant difference between the groups (mean ± SD; TXA group, 467 ± 242 mL; control group, 558 ± 236 mL; P = .20), calculating blood loss based on hemoglobin (Hb) balance. It is worth mentioning that the blood loss reported by Lee et al was significantly higher than that reported in the other studies (Table 3). The Hb method can be affected by hemodilution from intravenous fluid administration, which may lead to a higher value of blood loss.
Study Results: Blood Loss, Hemarthrosis, and VAS Pain Scores a
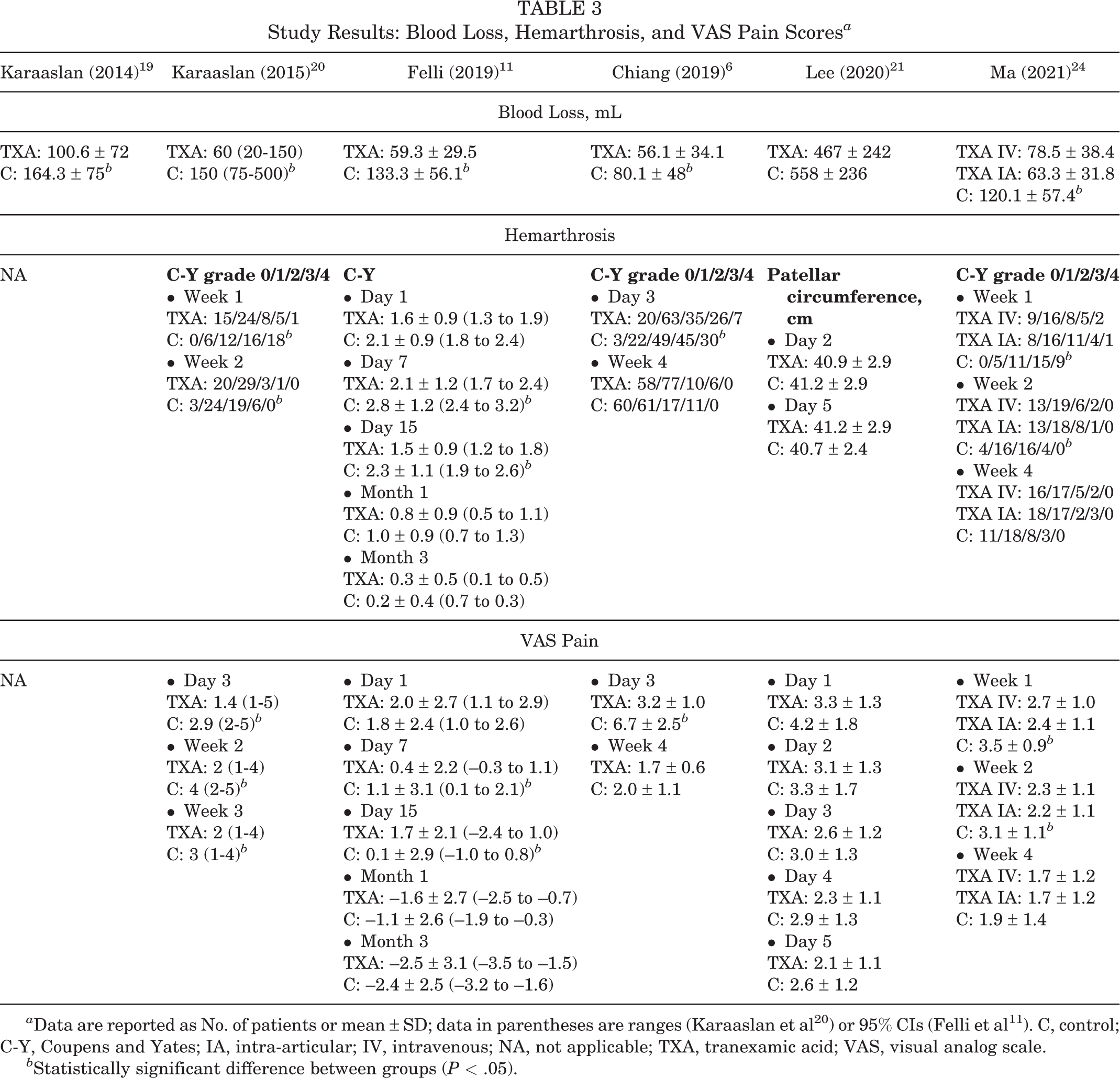
a Data are reported as No. of patients or mean ± SD; data in parentheses are ranges (Karaaslan et al 20 ) or 95% CIs (Felli et al 11 ). C, control; C-Y, Coupens and Yates; IA, intra-articular; IV, intravenous; NA, not applicable; TXA, tranexamic acid; VAS, visual analog scale.
b Statistically significant difference between groups (P < .05).
Hemarthrosis and Pain
Five studies 6,11,20,21,24 examined postoperative hemarthrosis (per the Coupens and Yates classification 8 ) and pain (per VAS). Four studies cited significantly lower scores in the early postoperative period (<4 weeks) for the TXA group than for the control group (week 2 in Karaaslan et al, 20 days 7 and 15 in Felli et al, 11 day 3 in Chiang et al, 6 and week 2 in Ma et al 24 ); however, the differences disappeared beyond 2 weeks postoperatively. One study 21 showed no difference in hemarthrosis grade when measuring patellar circumference and no difference in VAS pain scores between the TXA and control groups (Table 3).
ROM and Functional Outcomes
Karaaslan et al 20 reported a significantly larger ROM in the TXA group versus the control group on postoperative day 2 (107.36° ± 8.36° vs 103.65° ± 7.68°), and Felli et al 11 noted similar findings on postoperative day 7 (86.1° ± 11.9° vs 69.2° ± 27.1°) (Table 4). Chiang et al 6 and Lee et al 21 did not find a between-group difference in ROM on postoperative weeks 4 and 6, respectively. Regarding functional outcomes, Karaaslan et al 20 found a significant difference in Lysholm scores in favor of the TXA group in postoperative weeks 2 and 4. However, no between-group difference in Lysholm scores was noted by Felli et al or Ma et al 24 in postoperative week 4. Chiang et al did not find a significant difference in IKDC scores (International Knee Documentation Committee) between the groups.
Study Results: Range of Motion and Functional Outcomes a

a Data are reported as No. of patients or mean ± SD; data in parentheses are ranges (Karaaslan et al 20 ) or 95% CIs (Felli et al 11 ). C, control; IA, intra-articular; IKDC, International Knee Documentation Committee; IV, intravenous; NA, not applicable; TXA, tranexamic acid.
b Statistically significant difference between groups (P < .05).
Complications
No patients exhibited complications evoked via TXA administration, such as infection, deep venous thrombosis, or adverse events (Table 5).
Complications

Discussion
The current systematic review was performed to compare the effects of administering TXA in patients undergoing ACLR on reducing postoperative bleeding and on clinical outcomes. Of the 6 studies, 5 showed decreased drainage volume in the first 24 or 48 hours postoperatively in the TXA compared with control groups (ACLR with no TXA). 6,11,19,20,24 Four studies showed lower hemarthrosis grade and VAS score in TXA cases than controls in the early postoperative period, though this difference was not evident at 4 weeks postoperatively. 6,11,20,24 No studies showed differences in infection, deep venous thrombosis, or adverse events between the TXA and control groups, but the longest follow-up was only 6 weeks.
TXA has been widely adopted in orthopaedic surgery, such as joint replacement surgery and spinal surgery, for it can mitigate postoperative bleeding and swelling. As related reports have mainly focused on bone surgery, few have concentrated on the application of TXA in soft tissue surgery of the knee joint. Andrés-Cano et al 5 found that a 4.4% incidence of hemarthrosis may occur after ACLR. Of the 173 complications in 8741 knee procedures, the most common was hemarthrosis (60.1% of all complications). 36 Hemarthrosis can result in postoperative pain and swelling and delay recovery and the resumption of exercise; hence, it is necessary to study the hemostatic effect of TXA in ACLR, which belongs to soft tissue surgery of the knee joint. In view of some concerns on the toxic effects of a high concentration of TXA on human and animal chondrocytes in vitro, we need to study the safety of applying TXA in ACLR. This review discovered that intravenous and intra-articular administration of TXA in ACLR could significantly reduce postoperative intra-articular bleeding, as measured using a drainage system. In addition, no complications (eg, infection, deep venous thrombosis) or adverse events were caused by administration of TXA. However, the studies did not examine the potential toxic effect of TXA on the chondrocytes with intra-articular administration, as this would not become apparent until 6 to 12 months later.
The administration route of TXA is a hot topic of controversy. In a systematic review of joint replacement, Alshryda et al 3 concluded that regional intra-articular administration of TXA was more effective than intravenous TXA in reducing rates of bleeding and blood transfusions as compared with the placebo/control group, but the regional administration of TXA can rapidly increase its concentration in the joint cavity. Parker et al 33 discovered that when the concentration of TXA was >20 mg/mL, it would change the morphology of chondrocytes in vitro, decrease their adhesion and metabolic activity, and thus increase their mortality. Since the joint needs to be replaced with a metal prosthesis in joint replacement surgery, we seem to be able to ignore the cartilage and inject TXA into the articular cavity while ensuring its reliability. The truth is that most patients undergoing ACLR are young and have high demands for the restoration of knee joint function. Therefore, we have to carefully consider whether to inject TXA regionally into the articular cavity. Felli et al 11 found that the mean 24-hour drainage volume was 59.3 ± 29.5 mL, and Chiang et al 6 determined that the 24-hour postoperative drain volume was 56.1 ± 34.1 mL with intra-articular TXA application. Results from the 2 injection methods turned out to be similar. Because of theoretical concerns regarding chondrocyte toxicity with intra-articular TXA, we recommend intravenous infusion of TXA after ACLR, although this recommendation needs to be confirmed in future RCTs.
The methods used for measuring the amount of bleeding in studies are controversial: 5 studies 6,11,19,20,24 used a drainage system, while 1 study 21 adopted the Hb balance method to calculate bleeding volume. When closed suction drains are used to measure blood loss, many confounding factors may affect the outcome, such as the depth of the suction tube inserted into the articular cavity and whether it is compressed or crimped, the indwelling time of a drain, and whether the intra-articular injection of TXA will increase regional blood coagulation and then cause the formation of blood clots in the suction tube. These factors can lead to relatively lower measurement results on blood loss. The Hb balance calculates the amount of bleeding after knee arthroplasty based on the theory of perioperative Hb balance and thus can be regarded as a clinically practical and reasonable measurement method for this reason. 12 The disadvantage of this measurement method is that it also takes into account the amount of bleeding outside the joint cavity (eg, there will be more bleeding after the harvest of autografts), resulting in a higher value recorded for the estimated bleeding volume. The Hb method can also be affected by hemodilution from intravenous fluid administration.
The postoperative bleeding volume of the TXA group measured by Lee et al 21 was 467 ± 242 mL, which was much higher than that in the other 5 studies. In the 3 studies where TXA was injected intravenously, the drug dose and the methods of measuring bleeding volume were different from those of Lee et al. These dissimilarities made the final bleeding volume incomparable. 11,20,24 Despite the fact that Chiang et al 6 performed intra-articular administration of TXA, which was the same as Lee et al, TXA dose and the measurement methods of blood loss were different between the groups. Notwithstanding that continuous bleeding existed in the first few days after ACLR, Chiang et al measured the drainage volume only on the first day after surgery, which would make the amount of bleeding measured lower than the real value. For this reason, more reasonable measurement methods should be introduced to accurately measure bleeding volume in future research.
Acute pain and hemarthrosis are the most common complications after ACLR, and hemarthrosis is the main cause of postoperative pain. 27 In 4 studies 6,11,20,24 of this review, the degree of hemarthrosis and VAS pain score in the TXA group were considerably lower than those in the control group, and the 2 differences occurred almost synchronously. These results suggested that clinical correlation can be obtained between the grade of hemarthrosis and VAS pain score after ACLR. The degree of hemarthrosis in all 4 studies was graded using the method described by Coupens and Yates. 8 Karaaslan et al 20 and Felli et al 11 found that the ROM of the TXA group was notably greater than that of the control group 2 and 7 days after surgery, respectively, while Chiang et al 6 and Lee et al 21 compared knee ROM 4 and 6 weeks after surgery, respectively, and reported no significant difference between the groups. The reason may be that TXA reduced early postoperative swelling and pain, hence allowing patients to move at a larger range. In contrast, a negligible difference lay in joint swelling and pain between the groups after 4 weeks, so the ROM was similar.
The present review revealed that TXA administration was associated with lower severity of blood loss and hemarthrosis when compared with the control. The occurrence of early postoperative infection contributed to greater hemarthrosis in patients undergoing total knee arthroplasty. 23 It has been documented that postoperative hemarthrosis after arthroscopic subcutaneous lateral retinacular release results in the scar formation, decreased ROM, and delayed rehabilitation. 35 Therefore, TXA administration with early rehabilitation may improve muscle recovery in the postoperative short term. In this review, clinical correlation was obtained between the grade of hemarthrosis and VAS pain score after ACLR. Thus, the use of TXA should lead to less narcotic usage than use of the control because of the effect of reduced pain. However, these factors were not well studied in the studies.
This review has limitations. First, the heterogeneity among administration routes, dose, and outcome measurements in studies made it impossible for data collection and analysis to be carried out. Second, given the heterogeneity and the relatively small number of original studies, we could not compare the effectiveness of intravenous and intra-articular administration of TXA to figure out which can effectually reduce postoperative bleeding and perfect clinical results. We do not really know what the correct dose is to use to have the desired effect with minimal risk. Given the toxic effect of a high concentration of TXA on articular chondrocytes, it is worthwhile for us to know the dissimilarity between the clinical effects of the 2 administration routes and then judge the comparative merits and demerits to make more useful guidance on clinical application. Third, confounding factors may have existed in the original studies; for example, the arthroscopic meniscectomy conducted with ACLR will increase the amount of postoperative bleeding. If we want to strictly evaluate the effectiveness of TXA in reducing blood loss after ACLR and in promoting better clinical results in the early postoperative period, future studies must rule out the interference caused by arthroscopic meniscectomy. Fourth, the interval time from injury to operation can be regarded as an essential confounding factor, as acute ACLR surgery could lead to increased postoperative pain and decreased ROM secondary to arthrofibrosis formation. Also, no long-term data could be recorded on outcomes, strength differences, or possible chondral toxicity effects.
Conclusion
The current best available evidence suggests that TXA administration (intravenous or intra-articular) at the time of ACLR results in decreased intra-articular bleeding (measured using a drainage system), hemarthrosis grade, and pain as compared with control. Further studies are needed to see if TXA improves long-term outcomes in patients undergoing ACLR.
Footnotes
Final revision submitted August 3, 2021; accepted August 24, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by the Natural Science Foundation of Inner Mongolia (2020MS08119). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
