Abstract
Background:
There is growing interest in various biological supplements to improve tendon healing in patients after arthroscopic rotator cuff repair. The ideal biological supplement to strengthen rotator cuff remains unknown.
Purpose:
To assess the safety and efficacy of autologous cultured dermal fibroblast (ADF) injection on tendon-to-bone healing in patients after arthroscopic rotator cuff repair.
Study Design:
Case series; Level of evidence, 4.
Methods:
Included were 6 patients who underwent arthroscopic rotator cuff repair between June 2018 and March 2020; all patients had a full-thickness rotator cuff tear (>2 cm) involving the supraspinatus and infraspinatus tendons. The patients were injected with ADF between the repaired tendon and footprint during arthroscopic rotator cuff repair using the suture bridge technique. The safety of ADFs and the procedure was evaluated at 5 weeks postoperatively, and the anatomical healing of the repaired tendon was accessed at 6 months postoperatively using magnetic resonance imaging and at 12 months using ultrasonography. Outcomes including shoulder range of motion (ROM), visual analog scale (VAS) for pain, and functional scores were measured at 6 and 12 months postoperatively.
Results:
Adverse reactions to ADF injection were not observed at 6 months after surgery. VAS and functional scores at 6 and 12 months postoperatively were significantly improved compared with preoperative scores (all P < .05). However, there was no significant difference on any ROM variable between preoperative and postoperative measurements at 6 and 12 months (all P > .05). No healing failure was found at 6 and 12 months postoperatively.
Conclusion:
There was no adverse reaction to ADF injection, and all patients had successful healing after rotator cuff repair. A simple and easily accessible ADF injection may be a novel treatment option for increasing the healing capacity of torn rotator cuff tendons. Further clinical research is needed to verify the study results.
The prevalence of rotator cuff tear is rapidly increasing, mainly because of the aging population and advancements in radiologic diagnosis. 25,45 Tears are reported in approximately 30% of individuals aged ≥60 years and nearly 60% of individuals aged ≥80 years. 25,45 Some advocate treating full-thickness rotator cuff tears surgically in order to reduce pain, restore range of motion (ROM), and strengthen muscle power. Arthroscopic rotator cuff repair has become a common surgical technique for treating rotator cuff tears, and because it is minimally invasive, it may have the additional benefits of reducing postoperative pain and improving early functional recovery. 39 Although arthroscopic surgical techniques have improved rapidly, the failure rates for large tears range between 20% and 94%. 14,16 Therefore, there is a growing interest in various biological supplement methods for enhancing tendon healing in patients after arthroscopic rotator cuff repair. One of these methods is the use of autologous cultured dermal fibroblasts (ADFs). 26
Dermal fibroblasts are easy to obtain and are similar to tenocytes used for collagen synthesis. Skin is a rich and accessible source of fibroblasts, and these skin-derived cells can easily differentiate into tenocytes. 46 By taking advantage of these benefits, researchers are gradually developing tissue engineering techniques using autologous dermal fibroblasts, particularly in the fields of plastic surgery and dermatology. Various studies 31,34,44 have proven the effect of autologous fibroblasts on soft tissue augmentation. Within orthopaedics, several clinical studies 6,9 have shown that ADF injection can be safely used in refractory tendinopathies, such as lateral epicondylitis and patellar tendinopathy, with significant improvement in pain and function. In a previous rabbit study, 26 which was based on the results of the aforementioned studies, 6,9 a tear was created iatrogenically and left for 6 weeks to establish a chronic rotator tear model. Then the torn tendons were repaired in a transosseous manner with the injection of ADFs. An increased rate of rotator cuff healing and greater collagen fiber continuity were demonstrated after the injection of ADFs. In other words, ADF injection might enhance tendon-to-bone healing after surgical repair of torn rotator cuffs. 26
To the best of our knowledge, no clinical study has analyzed the safety and efficacy of ADF injection in patients who have undergone rotator cuff repair. Based on the encouraging findings from the animal model, 26 this clinical study was initiated by an approved first-in-human protocol in patients with rotator cuff tears. We aimed to assess the safety and efficacy of autologous dermal fibroblast injection for tendon-to-bone healing in patients with full-thickness rotator cuff tear.
Methods
Study Design and Patients
The present study was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 43 The study was an observational first-in-human study, limited to 6 patients, and the protocol was approved by the Ministry of Food and Drug Safety (MFDS [formerly known as the Korea Food & Drug Administration]) of Republic of Korea and the Institutional Review Board of Seoul National University Bundang Hospital. The study was designed in accordance with the Declaration of Helsinki, and written informed consent was obtained from all patients before enrollment, 8 weeks before surgery.
The study was conducted between June 2018 and March 2020. Six patients who met the following inclusion criteria were enrolled: aged between 20 and 80 years and underwent arthroscopic rotator cuff repair for the treatment of a >2-cm full-thickness rotator cuff tear involving the supraspinatus and infraspinatus tendons. The patients received ADF injection between the repaired tendon and footprint during arthroscopic rotator cuff repair using a suture bridge technique. Excluded were patients (1) who were not eligible for skin tissue biopsy, (2) with a simultaneous subscapularis tear, and/or (3) with a medical and surgical history at the time of screening (Table 1).
Inclusion and Exclusion Criteria a

a HIV, human immunodeficiency virus.
Fibroblast Preparation
A skin biopsy of approximately 1 cm2 was obtained from an unexposed area of the buttock 9 (Figure 1), and the donor site was closed using primary repair. The harvested skin was sent to a commercial laboratory (Tego Science Inc) for fibroblast culture. Fibroblasts were cultured for 4 weeks, as described in previous reports. 3,5,24,33 These fibroblasts, at passages 4 to 6, were resuspended in phenol red–free Dulbecco’s modified Eagle medium (2 × 107 cells/mL). A series of additional efficacy release tests were performed on the final product, including the confirmation of cell count and the assessment of cell viability. In addition, fibroblast suspensions were subjected to a series of quality control checks to ensure they achieved the purity, safety, and potency approved by the MFDS of Korea. The fibroblast suspension was shipped overnight to the treatment center at 2°C to 8°C for administration the next day (Figure 1).

Fibroblast preparation. (A, B) Skin biopsy of approximately 1 cm2 was obtained from an unexposed area of the buttock. (C) The fibroblast suspension (1 mL of autologous cultured dermal fibroblasts; 2 × 107 cells/mL) was sealed in an injection vial and shipped overnight to the treatment center at 2°C to 8°C for administration.
Dosage Setting and Injection Method
In vivo toxicity analysis of the fibroblast suspension was conducted according to Good Laboratory Practice and the MFDS guidance for nonclinical studies. 13 Human fibroblasts were injected into the muscle-tendon junction of an athymic nude mouse. From the nonclinical study, it was suggested that the approximate lethal dose was ≥1 × 10 6 cells per mouse, which is equivalent to 2 × 109 cells per human. In general, nonclinical trials recommend 50 to 100 times the actual clinical dose. 13 Thus, a 1-mL ADF injection was determined.
There are 2 × 107 cells in 1 mL of ADFs, of which “biologically active cells” are 1.4 × 107 cells, with a cell viability of >70%. During rotator cuff repair, 1 mL of ADF and 1 to 2 mL of a fibrinogen/thrombin solution (fibrin glue) were simultaneously injected between the tendon and greater tuberosity.
Surgical Procedures and Perioperative Management
All procedures were performed by the senior author (J.H.O.) using 3 arthroscopic portals (anterior, lateral, and posterior), with the patient under general anesthesia in the lateral decubitus position. For patients with a stiff shoulder (forward flexion [FF] <100°, external rotation [ER] at the side <20°, or internal rotation [IR] at the back <L2 level), preoperative manipulation was applied using arthroscopic capsular release. After glenohumeral inspection, subacromial decompression was performed to remove the inflamed bursal tissue. Acromioplasty was performed using a motorized bur in all patients. After footprint preparation was performed to expose the bleeding bony surface using a ring curette, rasp, and shaver, the anteroposterior tear length and retraction were measured using a calibrated probe. Rotator cuff repair was performed using the double-row suture bridge technique with several suture anchors. 37
Details of the ADF injection technique are as follows. The fibroblast suspension was shipped overnight to the treatment center at 2°C to 8°C. This suspension was warmed to room temperature for 10 minutes before injection. For ADF injection using the fibrin sealant product, we made the ADF injection kit using an 18-gauge spinal needle, a 1-mL syringe, and an intravenous 3-way stopcock with an extension line set. The use of Tisseel (Baxter) to stabilize particulate grafts, such as platelet-rich proteins, has been reported. 4 For each intravenous 3-way stopcock, one port was connected to a 1-mL syringe containing the fibroblast suspension, a second port was connected to the Tisseel, and the last port was tightly secured to the end of the extension tube using a spinal needle for ADF injection between the tendon and the greater tuberosity after the medial row suture tie (Figure 2A).
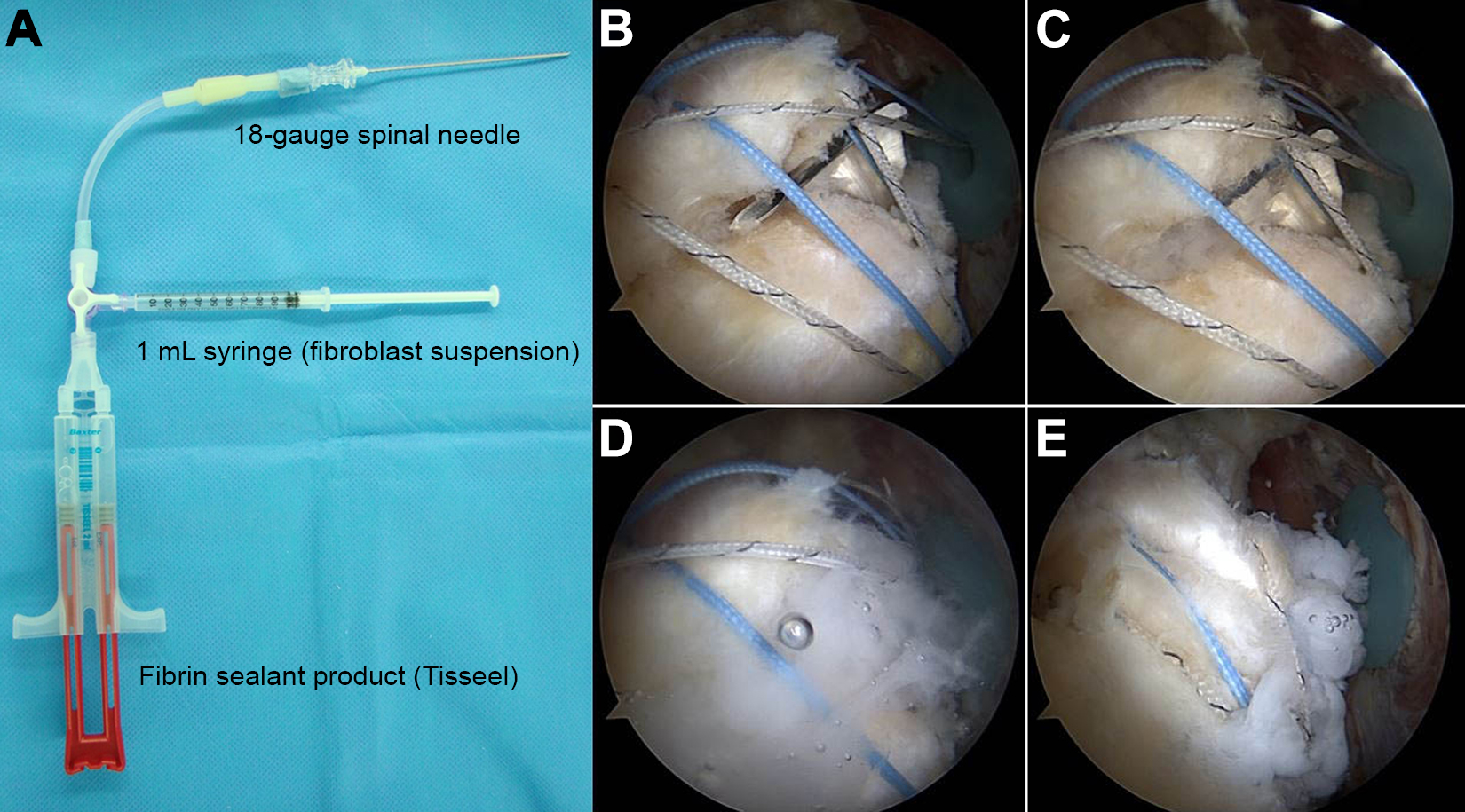
Autologous cultured dermal fibroblast (ADF) injection method. (A) The ADF injection kit includes an 18-gauge spinal needle, a 1-mL syringe, and an intravenous 3-way stopcock with an extension line set. For each intravenous 3-way stopcock, one port was connected to a 1-mL syringe containing the fibroblast suspension, another port was connected to the Tisseel (Baxter), and the remaining port was tightly connected to the end of the extension tube, with a spinal needle for ADF injection. (B-D) The ADF suspension and fiber sealant were injected simultaneously between the tendon and the greater tuberosity, after medial row sutures were tied and before the first lateral anchor was inserted deeply. (E) The first lateral anchor was deeply secured, and the second lateral anchor was finalized using tensioning for suture bridge repair.
The ADF suspension and fiber sealant must be injected simultaneously, and the injection should be between the tendon and the greater tuberosity, after medial row sutures are tied and before lateral anchors are inserted (Figure 2, B-D). After completion of the ADF injection, the first lateral anchor was deeply secured, sutures were tensioned for suture bridge repair, and the second lateral anchor was finalized (Figure 2E). It is important not to perform the injection too quickly because the suspension can be washed off with water, and if the suspension is injected too slowly, the fibrin sealant product may harden, making the injection difficult. Thus, an appropriate speed is required. We performed this procedure in an arthroscopic saline atmosphere because the ADFs were mixed with fibrin, but dry arthroscopic injection is also possible.
After subacromial procedures, the surgeon always returns to the glenohumeral joint to check for pulled-out anchors, missed foreign materials, or biceps capturing.
Rehabilitation
Postoperative rehabilitation for arthroscopic rotator cuff repair was performed for the patients in the same manner that it was performed for those who did not receive ADF injection. After surgery, an ice pack was applied to the operation site for 5 days for all patients. Postoperative pain management was administered to all patients, using the same oral pain medication, such as tramadol, and the treatment was continued for up to 7 days postoperatively. To determine the safety and effects of ADFs, immobilization was maintained using an abduction brace for 5 weeks for all patients. Shoulder shrugging, active elbow flexion/extension, active forearm supination/pronation, and active hand/wrist motion were encouraged immediately after surgery. Passive ROM was not allowed during the brace-wearing periods. After patients were weaned off the abduction brace, active-assisted and active ROM exercises were encouraged, and muscle strengthening exercises began 10 to 12 weeks after surgery. All sporting activities were allowed at 6 months after surgery. Rehabilitation was co-supervised by the Department of Rehabilitation and Orthopedic Surgery.
Protocol for Outpatient Management and Outcome Evaluations
The skin biopsy (visit 1) was obtained 8 weeks before surgery (visit 3). Then, 2 weeks after the skin biopsy procedure, a second visit (visit 2) was made to the department for stitch-out. Follow-up visits were conducted at 2, 5, 10, 24, and 52 weeks (visits 4-8) after the injection (Figure 3). At skin biopsy (visit 1), patients underwent a baseline evaluation, including medical history, shoulder history, magnetic resonance imaging (MRI) check within 6 months, physical examination, blood draws for laboratory analysis, ROM assessment, visual analog scale (VAS) for pain, and functional outcome scores.

Flow diagram of the clinical trial. ADF, autologous cultured dermal fibroblast; MRI, magnetic resonance imaging; PO, postoperative; ROM, range of motion; VAS, visual analog scale.
For the safety study, information on treatment-emergent adverse events (TEAEs), 28 adverse drug reactions (ADRs), 11 and serious adverse events (SAEs) 11 was collected with each visit. A TEAE was defined as an event that emerged during treatment, which was absent before the treatment, or the worsening of an event relative to the pretreatment state. 28 An ADR was defined as an appreciably harmful or unpleasant reaction, resulting from an intervention related to the use of a medicinal product, which predicts hazards from future administration. 11 An SAE was defined as any untoward medical occurrence at any dose, leading to death, a life-threatening adverse drug experience, prolongation of existing hospitalization, and/or significant disability/incapacity. 11 The presence of SAEs with regard to the treatment was based on the investigator’s judgment. The general condition (eg, vital signs) and the shoulder (eg, pain, swelling, skin features, infection) of all patients were assessed on the day of surgery (visit 3), 2 weeks after surgery (visit 4), and 5 weeks after surgery (visit 5). Moreover, laboratory tests, including leukocyte counts and the serum level of C-reactive protein, were conducted 5 weeks postoperatively (visit 5) to monitor adverse responses.
Efficacy outcomes were assessed using primary and secondary endpoints. The primary efficacy endpoint was the tendon-to-bone healing of the repaired rotator cuff tendon at 6 months postoperatively (visit 7) based on MRI findings 20,27 and at 12 months postoperatively (visit 8) based on ultrasonography (US) findings. 7,8,22,38,41 Shoulder MRI was performed on a 3-T MRI system (Achieva or Ingenia; Philips Medical Systems) using a dedicated shoulder coil. The following imaging parameters were used: oblique coronal fat-suppressed T2-weighted imaging (repetition time/echo time, 2400-3500/80-100 milliseconds; echo-train length, 14-18; section thickness, 2.5 mm; matrix, 256 × 256; field of view, 14 × 14 cm). A musculoskeletal radiologist with 10 years of experience interpreted the MRI and US findings and evaluated the healing failure. The tendon-to-bone healing of the repaired rotator cuff is evaluated as the integrity of the repaired rotator cuff tendon in the Sugaya classification. 42 Further, grades 4 and 5 are considered to indicate healing failure after surgery in this classification. US has been established as comparable to MRI for evaluating the status of the repaired rotator cuff. 1,38
The secondary efficacy endpoints were ROM, VAS pain (0, no pain; 10, worst pain), and functional scores at 6 and 12 months postoperatively (visits 7 and 8). ROM was measured using a goniometer, with the fixed scapular position in 3 planes: FF, ER at the side, and IR at the back. FF was measured as the angle between the arm and thorax with the elbow fully extended. ER at the side was measured as the angle between the forearm and thorax with the upper arm in adduction and the elbow in 90° of flexion. IR at the back was measured using the vertebral level that the tip of the patient’s thumb could reach in the sitting position. For convenience of analysis, we numbered the vertebral levels serially as follows: 1 to 12 for the 1st through 12th thoracic vertebrae and 13 to 17 for the 1st through 5th lumbar vertebrae. 36 VAS pain was measured in all patients preoperatively and at 6 and 12 months postoperatively. Functional outcomes using the Constant score, American Shoulder and Elbow Surgeons (ASES) score, and Simple Shoulder Test (SST) were also evaluated preoperatively and at 6 and 12 months postoperatively. 2,21 ROM and functional scores were measured by a single researcher who was blinded to the current study.
Statistical Analyses
We assessed changes in clinical scores and radiologically measured rotator cuff properties at different follow-up times using the Wilcoxon signed rank test. Analyses were performed using R software (Version 3.5.2, R Development Core Team). Patient variables such as age, sex, and tear size were analyzed, and their mean values were obtained. The mean (± standard deviation) for each patient-reported primary outcome was calculated at baseline and each subsequent follow-up visit. P < .05 was considered statistically significant. Continuous data were presented as the mean ± standard deviations, and categorical data were presented as the frequency. Paired t tests were performed to test the difference between the follow-up means and the baseline measurements.
Results
Six patients (1 man, 5 women; age range, 56-73 years) were enrolled in this study between June 2018 and March 2020. Patient characteristics are shown in Table 2. All patients were followed up for >1 year postoperatively and did not require additional treatment during this observation period.
Characteristics of the Study Patients a

a AP, anteroposterior; BMI, body mass index; RCT, rotator cuff tear.
b Age at autologous cultured dermal fibroblast injection during rotator cuff repair.
The safety outcome was the safety of ADFs and the related procedure for 5 weeks postoperatively. At each visit, no TEAEs or ADRs occurred. Similarly, no SAEs were observed for 1 year after the ADF injection. Regarding the primary efficacy endpoint, no healing failure was observed in any patient at 6 months postoperatively (visit 7) based on MRI scans (Figure 4) and at 12 months postoperatively (visit 8) based on US findings. Regarding the secondary efficacy endpoints, there was no significant difference in all ROMs preoperatively to 6 and 12 months postoperatively (all P > .05) (Table 3).

Magnetic resonance imaging scans for each patient preoperatively (visit 1) and at 24 weeks postoperatively (visit 7). Arrows indicate the tear and repair site after autologous cultured dermal fibroblast injection.
Clinical Outcomes for Each Patient Preoperatively and at 24 Weeks and 52 Weeks Postoperatively a

a Bolded values indicate statistically significant differences versus preoperatively (P < .05). ASES, American Shoulder and Elbow Surgeons; postop, postoperatively; preop, preoperatively; SST, Simple Shoulder Test; VAS, visual analog scale. Data are expressed as the mean ± standard deviation.
b Measured using the vertebral level that the patient was able to reach with the thumb and numbered serially as 1 to 12 for the 1st to 12th thoracic vertebrae, 13 to 17 for the 1st to 5th lumbar vertebrae, and 18 for any level below the sacral vertebrae.
The mean preoperative VAS pain score (4.83 ± 0.98) was significantly improved at 6 months (0.33 ± 0.82) and 12 months (0.33 ± 0.82) postoperatively (P = .034). The mean Constant score increased from 52.83 ± 13.41 preoperatively to 69.67 ± 6.22 and 72.33 ± 3.44 at 6 and 12 months postoperatively, respectively (P = .034 for both). The mean ASES score improved from a preoperative value of 60.93 ± 11.67 to 92.48 ± 6.22 at 6 months and 95.27 ± 7.09 at 12 months postoperatively (P = .036 for both). In addition, the mean preoperative SST score (4.0 ± 3.79) was significantly different from the postoperative score at 12 months (11.67 ± 0.52, P = .036), with a marginal statistical difference in the SST score at 6 months postoperatively (10.83 ± 0.75, P = .056) (Table 3, Figure 5).

Clinical scores for each patient preoperatively (visit [V] 1) and at 24 weeks (V7) and 52 weeks (V8) postoperatively. The visual analog scale (VAS) for pain, Constant score, American Shoulder and Elbow Surgeons (ASES) score, and Simple Shoulder Test (SST) score were statistically improved. *Statistically significant difference versus preoperatively (P < .05).
Discussion
In this first human clinical trial with a 1-year follow-up, we demonstrated the safety and efficacy of ADF injection for tendon-to-bone healing in patients with full-thickness rotator cuff tear. ADFs, which were derived from the skin, were observed to be safe; no adverse events were observed under close monitoring. Additionally, all the patients achieved tendon-to-bone healing of the repaired rotator cuff at 6 to 12 months postoperatively, based on MRI and US findings, respectively. The VAS pain and functional scores at 6 and 12 months postoperatively were significantly improved compared with the preoperative scores (all P < .05).
In a previous preclinical study, Kwon et al 26 investigated the effects of ADF injection on tendon-to-bone healing in a chronic rotator cuff tear model of a rabbit. They reported that ADF injection promoted tendon-to-bone healing in terms of biomechanics and histology in rabbits. Their results created the momentum for this phase 1 clinical trial on tendon-to-bone healing in human patients with rotator cuff tear. There is a series of ongoing phase 3 clinical trials on the effects of ADF injection during arthroscopic rotator cuff repair. It is important to note that this study is the first to analyze the safety and efficacy of the application of ADFs for tendon-to-bone healing in the surgical repair of full-thickness rotator cuff tear in humans. In this pilot study, we would like to emphasize the usefulness of ADF in humans for the first time, as well as technical tips of ADF application during suture bridge repair of rotator cuff tendon.
Tendon-to-bone healing is essential to reduce unsatisfactory shoulder function and the progression of osteoarthritis after arthroscopic rotator cuff repair. 12 If the repaired tendon undergoes reactive scar formation rather than histologically normal regeneration at the tendon-to-bone insertion site, tendon-to-bone attachment using these fibrous scar tissue results in poor tendon quality and weak mechanical properties compared with the native tendon-to-bone insertion site. 17,19 Furthermore, healing failure remains common, and to minimize this, various strategies have been developed to induce the regeneration of native tendon-to-bone insertion sites instead of the formation of fibrous scars, for satisfactory recovery after rotator cuff tears. 39
Unlike many types of cell-based therapy (eg, mesenchymal stem cells and adipose-derived stem cells) that require bone marrow, synovium, or adipose tissue via an open or arthroscopic biopsy, 9,26 the ADF injection is not hard to obtain using small incision biopsy of skin. It is possible to obtain the suspension with only a simple skin biopsy, without major tissue defects at the donor site. 9 This could be the biggest advantage for commercializing ADFs in the near future. Because both tenocytes and dermal fibroblasts are derived from the mesoderm and have similar properties in cell morphology and extracellular matrix components, Liu et al 29 hypothesized and demonstrated that dermal fibroblasts may replace tenocytes as seed cells for in vivo tendon engineering. Deng et al 10 also reported that human dermal fibroblasts are likely to replace tenocytes for future tendon graft development in vitro with dynamic mechanical loading in a bioreactor system. The results of these previous studies 10,26,29 provide evidence that dermal fibroblasts may act as an appropriate substitute for tenocytes, which has been the basis for accelerating the clinical application of ADFs in rotator cuff tears.
In terms of safety, all patients who received ADF injection did not develop TEAE, ADR, and SAE in the current study. Moon et al 34 reported that ADF injection might be effective and safe for correcting nasojugal grooves. In their study, all 6 patients who participated in the phase 1 trial did not develop dose limit toxicity until 4 weeks after ADF injection. 34 Further, the safety of the injection was confirmed in 46 patients who were observed at 24 weeks. At 24 weeks after the injection, 76% of the ADF injection group showed better effects compared with the placebo group in nasojugal groove improvement. 34 Based on our findings, ADF injection could be safely used for rotator cuff healing in humans as well.
In terms of tendon healing, ADF injection at the tendon-to-bone junction resulted in healing in all 6 patients in this study. During the proliferation of new blood vessels and fibroblasts to form granulation tissue, the normal healing process of connective tissue occurs. 30,32 Fibroblasts migrate to the wound and undergo vigorous mitotic activity, at which time the role of these fibroblasts is primarily to secrete extracellular matrix proteins, especially type 1/type 3 collagen and glycosaminoglycans. 32 Matthews et al 32 reported that the fibroblast population decreases as the size of the tear in the rotator cuff increases. That is, in small-sized tears, a larger population of fibroblasts also proliferates actively and exhibits an active recovery process, whereas in large-sized tears, there are fewer fibroblasts, no evidence of cell proliferation, and a reduced reparative response to healing. Therefore, our supplement strategy of ADFs that induce tendon-to-bone healing may allow satisfactory recovery after rotator cuff tears.
There are several strengths in this study. First, this is the first prospective clinical trial to evaluate the safety and efficacy of ADF injection on tendon-to-bone healing in patients with full-thickness rotator cuff tears. The results of this case series are valuable to further demonstrate the safety and efficacy of ADF application, such as phase 3 clinical trials. Second, the current study included homogeneous patients in terms of tear size, operation time, difficulties, and extent of surgery; thus, we can estimate the safety and efficacy of ADF injection more objectively.
However, the current study was subject to several limitations. First, this study was limited by its small sample size. Only 6 patients were included, with a 1-year follow-up duration. Based on the successful results of this phase 1 clinical trial, we are currently conducting a phase 3 clinical trial with a larger sample size of patients. Second, it was not easy to inject ADFs accurately into the footprint of the rotator cuff tendon attachment area. To overcome this problem, fibrin was used as a carrier for ADFs. Fibrin is a natural macromolecular material with excellent biocompatibility and biodegradability, without rejection and toxicity, 35,40 and it has been widely used in the biomedical field. Third, the optimal dose of ADF injection for tendon-to-bone healing has not been evaluated. As we mentioned previously, we attempted to calculate and apply the optimal dose as much as possible. 13 Fourth, this study did not include a control group that received fibrin alone. Therefore, we cannot rule out the possibility of a placebo effect in improving the subjective clinical score. 15,26 However, the consistent improvement in clinical scores, along with the evidence of good structural integrity after repair of rotator cuff tear based on MRI and US findings, strongly suggests that this limitation did not affect the conclusions of this study. These can all be part of future investigations and will be important considerations in our ongoing phase 3 clinical trial of the efficacy of ADFs.
Conclusion
In the current study, there were no adverse reactions to ADF injection, and all patients had successful healing after rotator cuff repair. A simple and easily accessible ADF injection may be a novel treatment option to increase the healing capacity of torn rotator cuff tendons. Further research involving more patients is necessary to prove the reliability of ADFs for rotator cuff healing.
Footnotes
Final revision submitted June 23, 2021; accepted August 4, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by a grant from the Advanced Technology Center Program (10062289; development of autologous and allogenic fibroblast therapeutics for treating rotator cuff tears), funded by the Ministry of Trade, Industry and Energy of the Republic of Korea (to S.J. and J.H.O.). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Institutional Review Board of Seoul National University Bundang Hospital (study No. B-1805/466-003).
