Abstract
Background:
Chronic exertional compartment syndrome (CECS) of the anterior leg compartment (ant-CECS) is frequently treated with a minimally invasive fasciotomy. Several operative techniques and operative devices exist, but none have been compared in a systematic and randomized manner.
Purpose:
To compare efficacy, safety, and postoperative pain of a novel operative device (FascioMax fasciotome) with a widely accepted device created by Due and Nordstrand (Due fasciotome) during a minimally invasive fasciotomy for ant-CECS.
Study Design:
Randomized controlled trial; Level of evidence, 2.
Methods:
Patients with bilateral isolated ant-CECS between October 2013 and April 2018 underwent a minimally invasive fasciotomy using the FascioMax fasciotome in 1 leg and the Due fasciotome in the contralateral leg in a single operative session. Symptom reduction at 3 to 6 months and >1 year, postoperative pain within the first 2 weeks, peri- and postoperative complications, and ability to regain sports were assessed using diaries, physical examination, and timed questionnaires.
Results:
Included in the study were 50 patients (66% female; median age, 22 years [range, 18-65 years]). No differences between the devices were found in terms of perioperative complications (both had none), minor postoperative complications including hematoma and superficial wound infection (overall complication rate: FascioMax, 8% vs Due, 6%), or reduction of CECS-associated symptoms at rest and during exercise. At long-term follow-up (>1 year), 82% of the patients were able to regain their desired type of sport, and 67% (33/49) were able to exercise at a level that was comparable with or higher than before their CECS-associated symptoms started.
Conclusion:
Both the FascioMax and the Due performed similarly in terms of efficacy, safety, and levels of pain within the first 2 weeks postoperatively.
Registration:
NL4274; Netherlands Trial Register.
Keywords
Chronic exertional compartment syndrome (CECS) of the anterior leg compartment (ant-CECS) may cause disabling symptoms such as pain and tightness. Ant-CECS is found in up to a quarter of individuals with exercise-induced leg pain who visit a sports physician. 5,31,41 This proportion is even higher in specialized centers. 7 Although the exact incidence in the general population is unknown, the fact that 1 in 2000 active military service members is diagnosed with CECS each year suggests that the condition is rather common. 50 As symptoms are often vague, most patients require specialized clinicians and additional diagnostics, such as intracompartmental pressure (ICP) measurements, before CECS is diagnosed. Patients with undiagnosed or inadequately treated ant-CECS may lose interest in sports, resulting in reduced quality of life, decreased level of fitness, and reduced overall health. 29
Treatment of ant-CECS is initially nonoperative and includes rest, nonsteroidal anti-inflammatory drugs, stretching, inlays, massage, and avoidance of provocative activities. 1,32 A more elaborate nonoperative program including gait retraining and extensive physiotherapeutic treatment may be successful in subgroups of patients. 11,19,54 Although these treatments are beneficial in some, most patients ultimately choose to undergo an operative treatment, which is generally more successful. 28,46 The differential diagnosis of CECS includes tibial stress fracture (localized pain and abnormal imaging findings), medial tibial stress syndrome (MTSS; painful palpation of distal medial tibial rim), and popliteal artery entrapment syndrome (history of intermittent claudication and abnormal arterial signal in popliteal fossa).
A fasciotomy is the preferred operative treatment of ant-CECS. 2 Techniques advocated in the literature range from entirely open to minimally invasive with the aid of a fasciotome and/or an endoscope. 13,27,34,52 These techniques have trade-offs. A fully open technique using a pair of scissors offers good visibility at the expense of a large scar. An endoscope-assisted approach combines good visibility with minimal invasiveness but is likely to be more expensive and time-consuming. Techniques that use a fasciotome are a widely used compromise. Using this simple instrument, the fascia is cut over its entire length via a 2-cm skin incision. 13,48 Although this technique performs reasonably well, the lack of direct visual contact with the tip of the fasciotome and the inability to control the movements of the fascia during the procedure may increase the likelihood of damage to adjacent structures or may lead to an incomplete fasciotomy. 9
With the goal of reducing the risk of complications associated with the use of a traditional fasciotome, we developed a novel fasciotome termed FascioMax (van Straten Medical). 8 The aim of the current study was to compare this instrument with a proven and widely used fasciotome created by Due and Nordstrand 13 (Due fasciotome) in terms of symptom reduction, safety, and pain in the first 2 weeks postoperatively. It was hypothesized that the use of the FascioMax would lead to fewer complications compared with use of the Due fasciotome.
Methods
Study Design and Sample Size
This study is a single-center randomized self-controlled clinical trial. All patients were enrolled in Máxima Medical Center, a referral center for diagnosis and treatment of exercise-induced extremity syndromes. 36,47,51 Patients underwent the experimental operative treatment (FascioMax) in 1 leg and a control operative treatment (Due) in the contralateral leg during 1 operative session. This design eliminated patient-related confounders and minimized intergroup variability. Potential introduction of bias was minimized by randomized allocation and blinding of patients to device allocation. The treating physicians were not blinded. Sample size calculation was based on noninferiority (10% margin), a power of 80%, an α of .05, a success rate of 90% for the experimental treatment, and a success rate of 83% for the control treatment. Success rate of the control treatment was based on experience, whereas the success rate of the experimental treatment was estimated. The study protocol was approved by the local medical ethics committee and uploaded in the national trial registry (Netherlands Trial Register number NL4274). All procedures adhered to the 1984 Declaration of Helsinki and its later amendments, 53 and all participants signed an informed consent form.
Patient Selection and Randomization
Starting in October 2013, all patients aged ≥18 years who had planned operative treatment of bilateral ant-CECS were eligible for the current study. Patients were diagnosed with ant-CECS if history and physical examination were suggestive of ant-CECS and if ICP was elevated in the anterior compartment. ICP was considered elevated when it exceeded 15 mm Hg at rest, 30 mm Hg at 1 minute after a provocative exercise (eg, treadmill, heel-walking, or climbing stairs), or 20 mm Hg at 5 minutes after a provocative test. Dynamic ICP measurements were performed only in suspected compartments. We refrained from routinely measuring all 4 leg compartments to limit morbidity associated with multiple needling. Details of this diagnostic procedure were described in a previous publication. 7 Patients were excluded if they had clear clinical signs of CECS in other leg compartments, had elevated ICP values in the deep flexor (dp-CECS) and/or lateral compartment (lat-CECS), had leg conditions that may interfere with subjective pain sensations (previous leg trauma or leg surgery, neuropathy, or peripheral arterial disease), or if there was a language barrier. They were informed by both the treating surgeon (M.S.) and the first author (J.B.) and received a study information brochure. Enrollment continued until the calculated sample size was reached.
Enrollment occurred between October 1, 2013, and April 12, 2018. Of the 245 patients who were diagnosed with CECS, 185 were excluded (Figure 1). A total of 8 suitable patients declined participation, resulting in 52 included participants. One participant was excluded before the operative procedure, as the preoperative questionnaire revealed that symptoms were mostly unilateral. Thus, the legs of 51 ant-CECS patients were randomized. One patients was lost to follow up directly after the operative procedure, as such short term results were based on 50 patients. As one more patient was lost to follow up after the short term questionnaire, long term results were based on 49 patients.

Patient selection and follow-up. ant-CECS, chronic exertional compartment syndrome of the anterior leg compartment.
Allocation of the devices to either the left or the right leg was randomized in blocks of 4 participants. An uninvolved individual performed randomization and blinded the allocation using consecutively numbered, closed envelopes. Allocation was concealed for the investigators and treating physicians until unblinding during the operative procedure. Allocation was kept from the patients until they completed all questionnaires.
Interventions
On the day of surgery, all patients received 2500 IU of a low--molecular weight heparin subcutaneously 1 to 2 hours before surgery. Both legs were operated in a single procedure under general anesthesia or a spinal block, depending on patient's preference. A tourniquet was not used. The anterior compartment was released through a 2-cm longitudinal incision approximately 2 to 3 cm lateral to the tibial crest halfway between the fibular head and the lateral malleolus. No additional compartments were released. After bluntly removing the fatty tissue overlying the fascia, a small longitudinal cut was made in the fascia to enable introduction of the fasciotome. The fascia toward the knee joint was further opened using the allocated fasciotome. In 1 leg, the FascioMax fasciotome (experimental treatment) was used, in the other leg a Due fasciotome (control treatment) was used. Characteristics of both devices were described previously in detail. 8,13 A video of both procedures was supplemented to a previous publication. 8
The technique for using both fasciotomes is quite similar, except that the FascioMax device consists of 2 parts (Figure 2). The FascioMax's speculum was inserted through the skin incision and was advanced toward the knee joint. One blade was always ventral to the fascia, whereas the other was advanced dorsally to the fascia. When the correct position of the tip of the ventral blade was confirmed by palpation, the fasciotome was introduced into the speculum and cut the fascia in an optimally controlled manner. In theory, this approach has the potential to reduce the risk of injury to the surrounding tissue. The Due fasciotome cut the fascia by pushing its tip toward the knee joint.
In both legs, the fascial cut was extended toward the ankle joint under direct visual contact with scissors. After digital palpation confirmed completeness of the fasciotomy, the skin was closed in 1 layer. No additional compartments were released. All patients wore compressive leg stockings continuously for 14 days. They were permitted to walk but were asked to refrain from sports until the first outpatient visit after 2 weeks. Patients were then instructed to increase their activity level slowly until they reached maximal intensity in week 8. Patients were asked to contact us after this period in case of insufficient improvement.

(A) The FascioMax system, consisting of 2 parts: a thin, regular fasciotome and a speculum that can be opened and closed to fixate the fascia during the fasciotomy. (B) The Due fasciotome: essentially a metal rod with a vertical knife.
Outcome Measurements
Both devices were compared in terms of postoperative pain, peri- and postoperative complications, and symptom reduction. The ability to regain sports was also assessed. Primary outcome was a reduction in the 5 cardinal symptoms associated with CECS (pain, tightness, muscle weakness, cramps, and altered sensation). Intensity and frequency of each symptom were assessed preoperatively, postoperatively after 3 to 6 months (short term), and more than 12 months postoperatively (long term). Participants scored pain intensity using an 11-point Numeric Rating Scale (NRS; 0 = no pain, 10 = unbearable pain). Evidence suggests that an NRS is most suitable for detecting differences in pain, has a high compliance, and allows for statistical analysis. 16,20 A 5-point verbal rating scale (VRS; no, mild, moderate, severe, or extremely severe) was deemed more suitable for assessing intensity of the other 4 symptoms (tightness, cramps, muscle weakness, and altered sensation). A VRS is patient-friendly, although statistical analyses are more complicated. Symptom frequency was also assessed with a 5-point VRS (never, sometimes, half of the time, most of the time, or always). A symptom score (intensity × frequency) was calculated for all 5 cardinal symptoms. To compare overall improvement, individual symptom scores were summed for an overall CECS symptom score (range, 0-104 points). A summary of the scoring is shown in Table 1.
Scoring of CECS Symptomsa
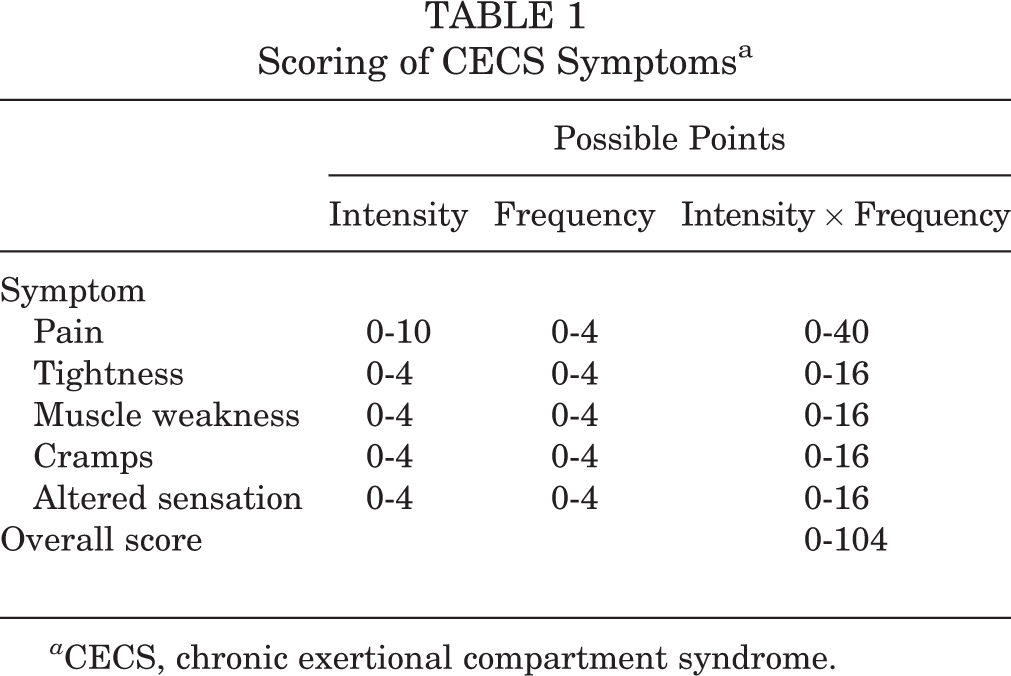
a CECS, chronic exertional compartment syndrome.
Secondary outcome measurements were intraoperative complications (substantial bleeding, incomplete fasciotomy), postoperative pain, postoperative complications, and ability to regain exercise. Postoperative pain was regarded as a measure of surgical trauma. During the first 14 postoperative days, patients scored deep anterolateral leg pain as well as superficial wound pain on an 11-point NRS (0-10). Total postoperative pain was defined as the sum of daily pain scores during the first 14 postoperative days (area under the curve). Patients also tabulated the use of analgesics.
Potential postoperative complications (hematoma, wound infection, and nerve injury) were reported by patients and registered at the postoperative outpatient clinic visit by the attending surgeon. Only wound infections that required antibiotics or operative treatment were tabulated. All visible hematomas were registered during the postoperative outpatient visit. In each patient, sensory function was assessed to identify potential nerve injuries. The ability to regain sporting activity was determined in the postoperative questionnaires.
Statistical Analysis
Data analysis was performed with SPSS Statistics Version 22.0.0.0 (IBM Corp.). When distributed normally, data were expressed as means and standard deviations, while nonparametric data were expressed as medians with ranges. Postoperative pain, symptom scores, and the combined CECS symptom score were seen as continuous variables and the intervention by either device as paired. Therefore, we used a Wilcoxon signed-rank test (nonparametric) to assess differences in postoperative pain, individual symptom reduction, and combined symptom score reduction. Differences in complication rate were assessed with a McNemar test. A P value < .05 was considered statistically significant.
Results
Patient Characteristics
Patient characteristics are tabulated in Table 2. As dictated by the study criteria, none of the patients had a history of peripheral artery disease, peripheral neuropathy, major leg trauma, or earlier leg surgery. However, 2 patients had received sclerotherapy for varices.
Characteristics of Participants (N = 50) a

a Values are reported as n (%) unless otherwise indicated.
Operative Procedure and Peri- and Postoperative Complications
Median operative time was 15 minutes (range, 11-37 minutes) for both legs together. In 98 of 100 legs, the fasciotomy succeeded at first attempt. Digital palpation revealed an incomplete fasciotomy in 2 legs (FascioMax n = 1, Due n = 1) and reintroduction of the device was necessary to accomplish a complete fasciotomy. Perioperative complications such as excessive bleeding or trauma were not observed. Additional skin incisions were not required. At the 2-week outpatient evaluation, 5 superficial hematomas were seen (3 FascioMax, 2 Due); all were treated nonoperatively. Two superficial wound infections required oral antibiotics (1 FascioMax, 1 Due). Major complications requiring reoperation or creating lasting symptoms were not encountered. Overall complication rate was 7% and similar in both groups (FascioMax, 8% vs Due, 6%; P ≥ .999).
Postoperative Pain
During the first 14 days, postoperative pain was similar in both groups (Figure 3). All patients (N = 50) had used the prescribed oral analgesics during the postoperative period. At day 14, only 4% (2/50) still used analgesics; by then, deep anterolateral leg pain and superficial wound pain were virtually absent (score ≤1) in 78% (39/50) and 74% (37/50) of the legs operated with the FascioMax, respectively. In legs operated with the Due fasciotome, these numbers were comparable (leg pain, 70%, 35/50; wound pain, 72%, 36/50). Total postoperative wound pain (P = .41) and leg pain (P = 0.65) were also similar in both groups (Figure 3).

Postoperative pain on days 1-14 after a fasciotomy for chronic exertional compartment syndrome of the anterior leg compartment. Patients scored wound pain and anterolateral leg pain daily using the Numeric Rating Scale (NRS). Area under the curve (AUC) ± SEM was calculated for patients individually.
Symptom Reduction
Figure 4 depicts the individual symptom scores before and after the operation. After 3 to 6 months, reductions of individual symptom scores were similar in both groups at rest (pain, P = .06; tightness, P = .57; muscle weakness, P = .89; cramps, P = .39; and altered sensation, P = .59) or during exercise (pain, P = .60; tightness, P = .64; muscle weakness, P = .05; cramps, P = .74; and altered sensation, P = .91). In the long term, both devices also accomplished comparable reductions of symptoms at rest (pain, P = .23; tightness, P = .88; muscle weakness, P = .52; cramps, P = .92; and altered sensation, P = .45) and during exercise (pain, P = .87; tightness, P = .82; muscle weakness, P = .24; cramps, P = .62; and altered sensation, P = .67). All 5 individual symptom scores were added to create a combined CECS symptom score (Table 1 and Figure 5). Intervention with both devices significantly reduced the combined CECS symptom score after 3 to 6 months (rest: FascioMax, P < .01; Due, P < .01; exercise: FascioMax, P < .01; Due, P < .01) and after >12 months (rest: FascioMax, P < .01; Due, P < .01; exercise: FascioMax, P < .01; Due, P < .01). No differences were found between the reductions induced by either device in rest (short term, P = .23; long term, P = .63) and during exercise (short term, P = .78; long term, P = .85) (Figure 5B).
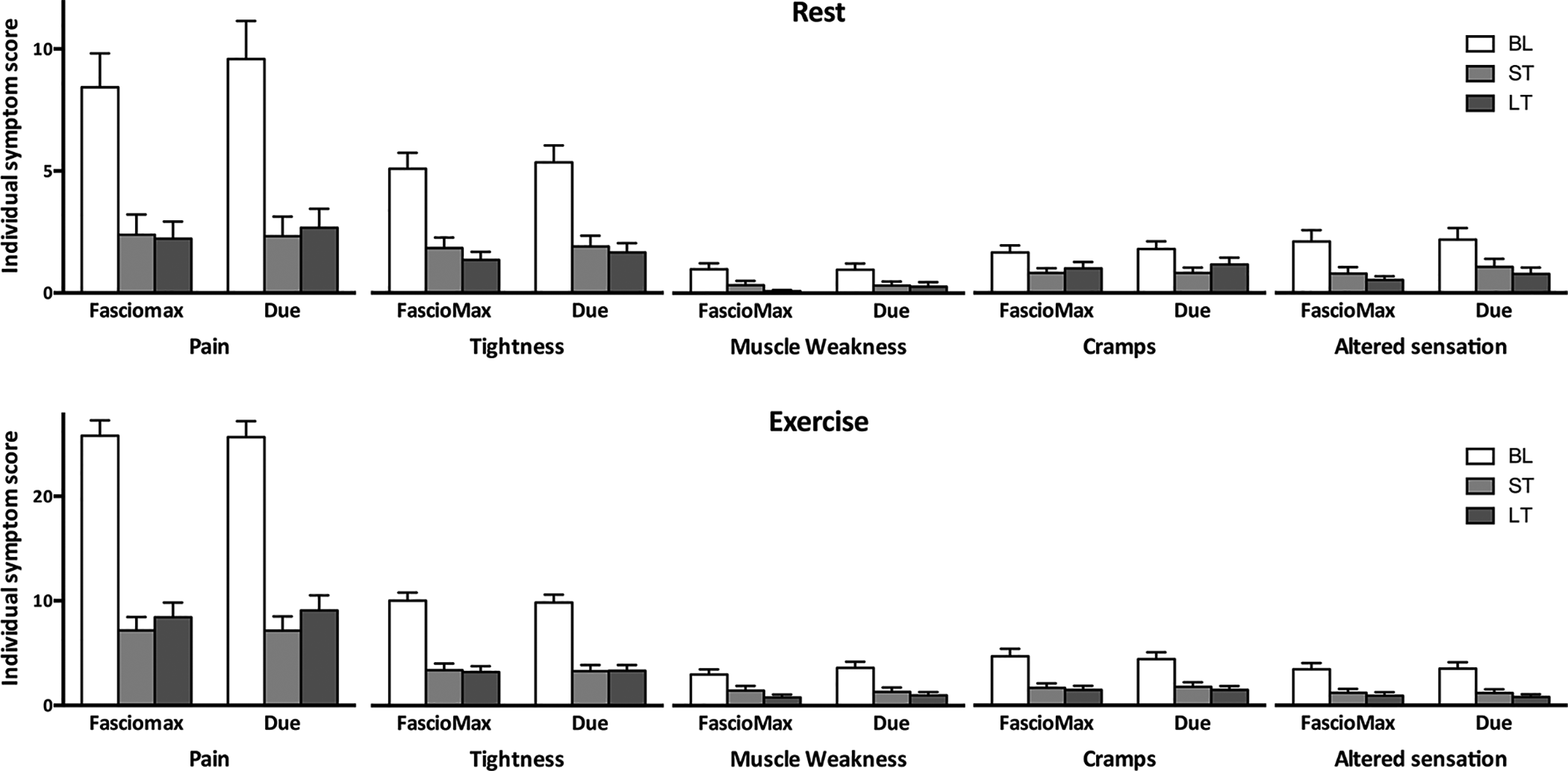
Chronic exertional compartment syndrome symptom scores at baseline (BL), 3 to 6 months postoperatively (ST) and >12 months postoperatively (LT). The symptom scores range from 0 to 40 (pain) or from 0 to 16 (tightness, muscle weakness, cramps, and altered sensation). Bars depict the mean score; error bars represent the SEM.

(A) CECS symptom scores at rest and during exercise at the preoperative (BL), 3- to 6-month postoperative (ST), and >12-month postoperative (LT) time points; *statistically significant difference. (B) After pooling (rest-ST, rest-LT, exercise-ST, and exercise-LT) we compared for each patient the proportional reductions in CECS symptom scores between legs operated with the FascioMax and legs operated with the Due fasciotome. The boxes represent the 25th-75th percentiles, and the whiskers represent the 10th-90th percentiles. Dots represent outliers. CECS, chronic exertional compartment syndrome.
Events During Follow-up Period
After the operation, 54% of the participants (27/50) were seen only during routine outpatient appointments, because the intervention had the desired result. The remaining 46% (23/50) had residual or recurrent symptoms in both legs that prompted them to schedule at least 1 additional outpatient visit during the follow-up period of at least 12 months (Appendix Table A1). Of these 23 patients, 2 were diagnosed with recurrent ant-CECS, as ICP was still elevated in the anterior compartments of both legs. They were offered a fasciectomy, but both declined because symptoms were not debilitating enough. A total of 17 patients underwent postoperative ICP measurements, which revealed dp-CECS (n = 5) or combined dp/lat-CECS (n = 2) necessitating a fasciotomy of these compartments (Appendix Table A1). Other causes for persistent leg disorders were MTSS (n = 4), bilateral noniatrogenic neuropathic pain (n = 1), bilateral hypomagnesic cramps (n = 1), bilateral fascial herniation (n = 1), or an insufficient gait pattern (n = 1). In the remaining 6 patients, the cause for the ongoing discomfort was undiagnosed.
At the final evaluation point >12 months postoperatively, all patients rated the results in both legs as excellent (n = 8), good (n = 16), reasonable (n = 10), poor (n = 11), or bad (n = 4).
Return to Sports
After 3 to 6 months, 82% of the patients (41/50) had returned to the sport they practiced before CECS-associated symptoms started. Moreover, 62% (31/50) exercised at a level that was comparable with or higher than before the symptoms had started. A total of 20% (10/50) were forced to exercise at a lower-than-desired intensity, 8% (4/50) had changed the type of sport, and 10% (5/50) had stopped sports altogether.
At the long-term follow-up, 82% of the patients (40/49) were able to participate in their favorite sport, 8% (4/49) were forced to start a new sport, and 10% (5/49) were not able to restart sports. Two-thirds of the patients (67%; 33/49) were able to exercise at a level that was comparable with or higher than before the debilitating CECS symptoms emerged.
Discussion
Several operative techniques for ant-CECS are currently used, each with advantages and limitations. An entirely open approach offers good visibility of muscle, crossing blood vessels, and nerves but at the expense of a rather large skin incision. 10,27,34 The use of a fasciotome (mostly of the Due type) limits an incision to 2 to 3 cm but the lack of continuous visual contact during the fasciotomy (“semiblind”) could increase the risk of collateral damage or an incomplete fasciotomy. 9,13,15,48 More recently, the aid of an endoscope was advocated. 23,40 Such an approach optimizes visibility without requiring a larger incision but is time-consuming and more expensive. 22,24,25,52 The present study was undertaken to compare a novel fasciotome that potentially aids in performing a more controlled, minimally invasive semiblind fasciotomy with a traditional Due fasciotome. Both devices performed similarly in terms of symptom reduction, safety, and postoperative pain when treating patients with isolated ant-CECS.
Studies that have described the effects of operative treatment for ant-CECS are very heterogeneous in terms of size, patient selection, and goals. As such, complication rates are often not reported. 30,44,45 In studies that have reported complication rates, up to 25% were found. 9,12,21,24,26,38,43 These include minor complications such as hematomas or superficial wound infections, but also major ones requiring reintervention (e.g., deep wound infection or major bleeding) or superficial peroneal nerve (SPN) injury. For instance, 1 report suggested that a semiblind fasciotomy is commonly associated with SPN damage as it was injured in 4 of 6 cadaveric legs during fasciotomy. 22 The current study found that major complications such as uncontrollable bleeding, SPN injury, muscle tissue necrosis, or reoperations were not encountered in any of the 100 legs treated. This finding is in line with 2 other publications indicating that a semiblind fasciotomy of the anterior compartment using the FascioMax (or Due) did not result in any SPN injury in more than 250 operated legs. 6,8 Furthermore, the number of minor complications was low and comparable with that found in other studies.
Little is known regarding levels of pain in the weeks immediately after ant-CECS surgery. Pain may well reflect the level of iatrogenic damage. In the current study, both devices resulted in limited levels of superficial (wound) pain and deep (muscle) pain in the first 14 days postoperatively. In addition, just 4% of the patients still required analgesics on postoperative day 14. Interestingly, insertion of the speculum during the FascioMax approach did not cause additional incisional trauma but it also did not reduce collateral trauma of tissue surrounding the opened fascia.
Reported rates of success following surgery for ant-CECS vary substantially. Some claim that residual or recurrent symptoms following a fasciotomy were virtually absent, 10,34,35 while others were considerably less successful. 21 A study in military personnel, for whom physical requirements are considerably higher, found rates of ongoing discomfort as high as 45%. 49 However, a comparison is difficult, as most publications fail to distinguish between recurrent and persistent ant-CECS, the onset of other types of leg CECS (dp-CECS or lat-CECS), or additional exercise-induced syndromes such as MTSS.
In the present study, the number of participants that experienced residual or recurrent symptoms may seem high. However, this is often not because of an incomplete fasciotomy, as only 2 patients (4%) had persistently elevated ICP in the anterior compartment. In contrast, most patients with ongoing symptoms were diagnosed with CECS of the dp and/or lat, or with MTSS. Symptoms of the anterior compartment may have been dominant before the operation. As a consequence, these other entities may have been masked earlier. Following successful treatment of ant-CECS, patients were able to increase exercise intensity, possibly unmasking other latent exercise-induced syndromes. This may also explain our high reintervention rate (18%; 9/50) compared with other studies. 4,33
CECS is associated with 5 cardinal symptoms: pain, tightness, cramps, muscle weakness, and altered skin sensation. 14 Traditionally, these symptoms are thought to arise during exercise and to subside during prolonged rest. 3 Although symptom reduction offers a reliable outcome measurement, most reports are restricted to patient satisfaction or return to sports. 10,30,34,37,39 The few studies that evaluated symptom reduction after a fasciotomy focused mostly on pain. 17 The current study assesses changes of intensity and frequency of all 5 symptoms associated with CECS. This approach enabled us to compose a symptom score and a combined symptom score, allowing for comparison of the effect of both devices. The FascioMax and the Due fasciotome reduced CECS-associated symptoms in a similar way, indicating that both devices are effective for patients with ant-CECS.
Improving the ability to exercise is an important reason for patients to undergo a fasciotomy. In fact, virtually all studies mention return to sports, or return to full activity in soldiers. It appears that most patients are able to regain sports following surgery, 18,30,44 although the number of patients who experienced full recovery without any residual symptoms is lower. 26,42,45 In active military personnel, these numbers are even more restricted. 33,49 In the current study, more than one-half of the patients (56%) had stopped participating in sports altogether because of CECS-associated symptoms. The fasciotomy resulted in the ability to again practice the desired type of sport in most patients (82%). However, one-third of our patients were forced to exercise at a lower than desired level or decided to shift to another type of sport. The ongoing presence of untreated additional exercise-induced syndromes such as MTSS or CECS of other leg compartments likely contributes to this percentage. Although all patients were evaluated by an experienced sports physician and underwent ICP measurements, we did not routinely perform radiographs, magnetic resonance imaging, or vascular workup. As such, we may have missed concurrent conditions during inclusion, thereby increasing the number of patients with ongoing symptoms. Although most patients were able to fully regain sports, no less than 30% rated their outcome as “poor” or “bad.” We believe that this suboptimal rating is explained in part by the high expectations that some of these athletes may have had. Improved preoperative counseling may help to manage such expectations.
The present study is the first to our knowledge to compare 2 devices during a minimally invasive fasciotomy for ant-CECS in a randomized and controlled manner. The main limitation is related to the study power. Although the calculated required sample size was reached, differences between the 2 devices were substantially smaller than anticipated. In order to reach the predetermined power, the number of included participants would need to be increased 5-fold, a number that is unrealistic for a condition such as ant-CECS. As we decided to terminate the study at the current number of participants, small differences in complications or symptom reduction may not have been detected. Second, we performed ICP measurements only of suspected compartments to minimize ICP-related morbidity. As a consequence, we may have missed other types of CECS that may have become symptomatic once exercise levels were intensified after successful ant-CECS surgery. This approach has likely affected the reintervention rate and has resulted in additional interventions during the follow-up period. We have tried to minimize bias by postponing the long-term questionnaire until reintervention was performed in both legs. Third, the CECS symptom score we introduced is not yet validated for assessing symptoms associated with CECS and symptom reduction following therapy. However, as shown by the differences in patient-reported outcomes at rest and during exercise, and before and after surgery, the symptom score was responsive to changes in the experienced symptoms. Fourth, a single surgeon with ample experience in both techniques performed all fasciotomies. It is possible that differences in operative results, complication rates, and symptom reduction may have been different if 2 (or more) surgeons with various levels of expertise had performed the procedures.
Conclusion
A minimally invasive fasciotomy of the leg anterior compartment with the aid of the FascioMax fasciotome is similar to the generally accepted Due fasciotome in terms of efficacy, complication rate, and postoperative pain. Awareness of concurrent exercise-induced conditions is required for optimal patient satisfaction.
Footnotes
Final revision submitted July 15, 2021; accepted July 26, 2021.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Máxima Medical Center (study No. 1330).
APPENDIX
Diagnoses and Treatments in Participants Requiring Additional Outpatient Clinic Visits a

| ID | Sex | Age, y | Rating (LT) b | Reason for Additional Visit | Additional Examination | Diagnosis | Treatment |
|---|---|---|---|---|---|---|---|
| 1 | F | 43 | Poor | Recurrent symptoms | ICP; ant/lat: not elevated | No definite diagnosis | None |
| 3 | M | 26 | Reasonable | Symptoms, medial leg | ICP; elevated pressure dp | Dp-CECS | Fasciotomy dp |
| 4 | M | 29 | Reasonable | Symptoms, medial leg | ICP; elevated pressure dp | Dp-CECS | Fasciotomy dp |
| 5 | F | 23 | Poor | Symptoms, medial leg/shins | ICP; ant/dp: not elevated | Bilateral MTSS | Fasciotomy dp |
| 6 | F | 22 | Reasonable | Recurrent symptoms | None | MTSS, possible dp-CECS | None |
| 8 | F | 21 | Good | Symptoms, medial/lateral leg | ICP; dp/lat: elevated pressure | Dp/lat-CECS | Fasciotomy dp/lat |
| 11 | F | 24 | Poor | Recurrent symptoms | ICP; ant: elevated pressure | Recurrent ant-CECS | None during follow-up |
| 12 | F | 20 | Reasonable | Symptoms, medial leg | None | Bilateral MTSS | Fasciotomy dp |
| 13 | F | 38 | Poor | Recurrent symptoms | ICP; ant/dp/lat: not elevated | Noniatrogenic neurogenic | Prolotherapy |
| 14 | F | 42 | Good | Symptoms, medial/lateral leg | ICP; dp/lat: elevated pressure | Dp/lat-CECS | Fasciotomy dp/lat |
| 18 | F | 21 | Good | Symptoms, medial leg | ICP; dp: not elevated | Muscle cramps | Magnesium supplements |
| 20 | M | 27 | Good | Symptoms, medial leg | ICP; dp: elevated pressure | Dp-CECS | Fasciotomy dp |
| 21 | F | 22 | Bad | Aggravation symptoms | Thorough multidisciplinary analysis | No definite diagnosis | None |
| 25 | F | 19 | Good | Symptoms, medial leg | ICP; dp: elevated pressure | Dp-CECS | Fasciotomy dp |
| 26 | F | 29 | Bad | Persistent symptoms | ICP; ant: not elevated | No definite diagnosis | Prolotherapy, physiotherapy |
| 27 | F | 21 | Bad | Recurrent symptoms | ICP; ant/lat: not elevated | No definite diagnosis | Analgesics, physiotherapy |
| 31 | M | 21 | Poor | Persistent symptoms | ICP; dp/ant/lat: not elevated | Insufficient gait pattern | Gait-training/inlays |
| 34 | F | 24 | Poor | Symptoms, lateral leg | None, symptoms not severe enough for ICP | No definite diagnosis | None |
| 37 | F | 22 | Bad | Recurrent symptoms | None, symptoms not severe enough for ICP | No definite diagnosis | None |
| 43 | F | 19 | Poor | Recurrent symptoms | ICP; ant: elevated pressure | Recurrent ant-CECS | None during follow-up |
| 45 | F | 19 | Reasonable | Symptoms, medial leg | None, diagnosis clear | Bilateral MTSS | Prolotherapy |
| 48 | F | 25 | Poor | Recurrent symptoms | ICP; ant: not elevated | Fascial herniation | None during follow-up |
| 51 | M | 22 | Good | Symptoms, medial leg | ICP; dp: elevated pressure | Dp-CECS | Fasciotomy dp |
a ant, anterior compartment; CECS, chronic exertional compartment syndrome; dp, deep flexor; F; female; ICP, intracompartmental pressure; lat, lateral compartment; LT, long term; M, male; MTSS, medial tibial stress syndrome.
b All patients who rated their outcome as “poor” or “bad” were offered outpatient consultation.
