Abstract
Background:
Different indications and treatment options for combined injuries to the anterior cruciate ligament (ACL) and medial collateral ligament complex (MCL) are not clearly defined.
Purpose:
To perform a modified Delphi process with the Committee for Ligament Injuries of the German Knee Society (DKG) in order to structure and optimize the process of treating a combined injury to the ACL and MCL.
Study Design:
Consensus statement.
Methods:
Scientific questions and answers were created based on a comprehensive literature review using the central registers for controlled studies of Medline, Scopus, and Cochrane including the terms medial collateral ligament, anterior cruciate ligament, MCL, ACL, and outcome used in various combinations. The obtained statements passed 3 cycles of a modified Delphi process during which each was readjusted and rated according to the available evidence (grades A-E) by the members of the DKG Ligament Injuries Committee and its registered guests.
Results:
The majority of answers, including several questions with >1 graded answer, were evaluated as grade E (n = 16) or C (n = 10), indicating that a low level of scientific evidence was available for most of the answers. Only 5 answers were graded better than C: 3 answers with a grade of A and 2 answers with a grade of B. Only 1 answer was evaluated as grade D. An agreement of >80% (range, 83%-100%) among committee members was achieved for all statements.
Conclusion:
The results of this modified Delphi process offer a guideline for standardized patient care in cases of combined injuries to the ACL and MCL.
Combined injuries of the anterior cruciate ligament (ACL) and the medial collateral ligament (MCL) complex represent the most common combined ligamentous injuries of the knee joint. 44
Therapy options after isolated ACL rupture are clearly defined. 58 With isolated ruptures of the ACL, the ligament can be retained only in rare cases such as femoral ruptures. 65 Otherwise, the ACL is usually reconstructed using various autologous grafts, such as the hamstring tendons (semitendinosus and gracilis tendons), quadriceps tendon, or patellar tendon. The use of an allograft is less common and often inhibited by particular national regulations. Patients who cope well in everyday life and subjectively report a stable knee joint may also be treated nonsurgically.
In contrast, an isolated MCL rupture has long been a domain of nonoperative therapy. 47 Isolated or partial ruptures of the MCL are mostly treated nonoperatively in a brace that protects the knee from buckling inward, which may limit flexion or extension. To date, the duration of brace application, brace limitations, and duration of nonweightbearing in cases of MCL rupture are not clearly defined. Furthermore, there are no commonly accepted criteria used to define the outcome after nonoperative MCL treatment as successful or unsuccessful.
Whereas the treatment path for an isolated MCL rupture lacks a clear consensus, indications and treatment options for combined injuries to the ACL and MCL are even less clearly defined. A systematic evaluation of clinical results is difficult given a heterogeneous population of patients who frequently have further concomitant injuries affecting the menisci, cartilage, or other ligaments of the knee joint as well as different rupture patterns of the MCL, including the superficial medial collateral ligament (sMCL), deep medial collateral ligament (dMCL), and posterior oblique ligament (POL), as shown in Figure 1. Consequently, the treatment of these injuries is often guided by clinical experience and personal preference of the treating surgeon.

The medial collateral ligament complex consisting of the superficial medial collateral ligament (sMCL), deep medial collateral ligament (dMCL), and posterior oblique ligament (POL).
The aim of this study was to perform and evaluate a modified Delphi process 42 with the Committee for Ligament Injuries of the German Knee Society (DKG). The study was intended to structure and optimize the process of treating a combined injury to the ACL and MCL.
Methods
The consensus group consisted of the DKG Ligament Injuries Committee and its registered guests. All committee members are specialized knee surgeons highly experienced in treating single- and multiligament injuries who, due to their professional specialization and scientific career, are among the leading professionals in their field in Germany. This ensured that all those involved in the consensus finding had sufficient experience with the pathology discussed.
The process of this consensus project is outlined in Figure 2. Initially, a comprehensive review of the literature using the central registers for controlled studies of Medline, Scopus, and Cochrane was performed. The following terms were included in the search and used in various combinations: medial collateral ligament, anterior cruciate ligament, MCL, ACL, and outcome. In addition, reference lists in the studies identified were searched for relevant studies. Clinical studies with evidence levels between 1 and 5 were included in this analysis. Animal and cadaveric studies were excluded for the assessment of the level of evidence and, if relevant, were added as additional information.

Flowchart of the procedure used to determine the consensus for combined injuries to the anterior cruciate ligament and the medial collateral ligament complex. DKG, German Knee Society.
In the first cycle, an initial and orienting questionnaire was sent to the committee members. Three authors (D.G., T.P., and R.B.) were responsible for developing the questions and answers based on the previous literature review. Questionnaires were sent by email, and the data were processed anonymously. The questionnaire allowed open answers containing the qualifications of the committee members as well as a wide range of questions about the care of combined injuries of the ACL and MCL.
This initial questionnaire was used to create ideas and concepts with regard to the research question and to incorporate the clinical experience of the committee members. Based on the answers of the committee members and the results of the literature research, important questions in 4 subject areas (indications, nonoperative therapy, operative therapy, postoperative follow-up treatment) were answered verbalizing scientific statements. The respective scientific evidence was given at the end of each statement according to Table 1.
Level of Scientific Evidence Given at the End of Each Statement Based on the Type of Studies Available in the Current Literature
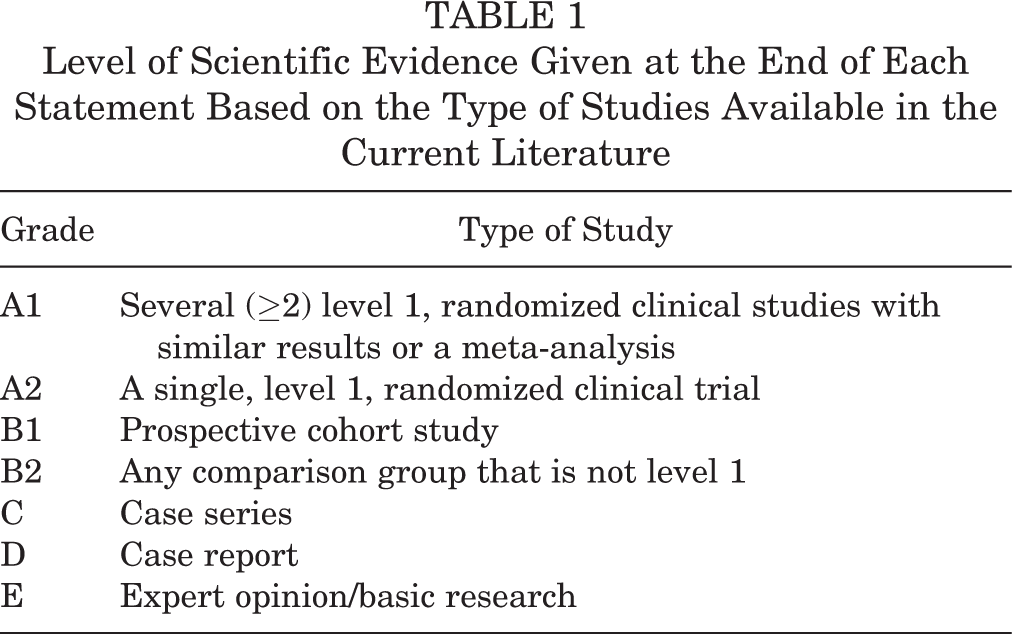
In the second cycle, the obtained statements were rated by the committee members using a 5-point Likert scale in addition to the option “I can’t judge.” For each statement, the formulation of free-text comments was possible (for example, to explain the evaluation or to express the rejection of the relevance of the statement). The first author (D.G.) then analyzed the answers for a subsequent submission to the committee members by a further questionnaire with statements from the third cycle. Finally, the revised draft was resubmitted to the committee members for a final evaluation. Here, an answer of “I agree” or “I do not agree” was required. The possibility of individual comments was no longer given. In all cycles of the modified Delphi process, the committee members received feedback with the evaluation of the total responses of all committee members to the respective statements. As usual in literature, a satisfactory consensus was defined as 80% agreement. 10
Among all participants, the questionnaires were created using the SoSci program (SoSci Survey GmbH). This program allows the completeness of the questionnaires to be traced automatically. The data were processed using SPSS (Version 20.0; IBM) and Excel 2019 (Microsoft).
Results
The majority of answers, including several questions with >1 graded answer, were evaluated as grade E (n = 16) or C (n = 10), indicating that a low level of scientific evidence was available for most of the answers. Only 5 answers were graded better than C: 3 answers with a grade of A and 2 answers with a grade of B. Only 1 answer was evaluated as grade D. For all statements, an agreement of >80% (range, 83%-100%) among committee members was achieved. The following section is a summary of the questions and answers for the 4 subject areas:
Questions and Answers
Indication
1. Does treatment of a combined ACL/MCL injury depend on the pattern of injury?
In the absence of reliable evidence, it is the opinion of this working group that the treatment of a combined ACL/MCL injury might depend on the pattern and location of injury and might follow detailed, individualized treatment regimens. 54 The lack of evidence supporting operative or nonoperative treatment options of the MCL in combined ACL/MCL injury might be caused by inadequate individualization of treatment regimens (grade E; agreement, 100%).
2. What is the role of stress radiographs in combined ACL/MCL injury?
Chronic cases: Stress radiographs can play a role in quantifying medial instability in chronic MCL insufficiency 71 (grade C; agreement, 100%).
Acute cases: In the absence of reliable evidence, it is the opinion of this working group that the application of stress radiographs in acute cases might be limited due to pain and knee effusion and might not have clinical relevance (grade E; agreement, 100%).
3. What is the role of the history and the clinical examination in combined ACL/MCL injury?
The practitioner should obtain a relevant history and perform a musculoskeletal examination of the lower extremities, since these are effective diagnostic tools for ACL and concomitant injuries † (grade A1; agreement, 100%).
4. What is the role of magnetic resonance imaging (MRI) in combined ACL/MCL injury?
The practitioner should obtain an MRI scan to confirm ACL and MCL injury and to identify concomitant knee pathology such as other ligamentous, meniscal, or articular cartilage injury ‡ (grade A1; agreement, 100%).
5. Should total ruptures of the ACL during combined ACL/MCL injury be treated nonoperatively or operatively?
The ACL can be surgically addressed in combined ACL/MCL injury. § Based on the available literature, there is no evidence that indications for operative treatment of the ACL during combined ACL/MCL injury differ from those in isolated ACL injury (grade B2; agreement, 91%).
6. Should total ruptures (grade III) located at the femoral insertion of the MCL during combined ACL/MCL injury be treated nonoperatively or operatively?
Femoral total ruptures (grade III) of the MCL in combined ACL/MCL injury can be treated operatively 28,36 (grade C; agreement, 100%).
7. Should total ruptures (grade III) located in the midsubstance of the MCL during combined ACL/MCL injury be treated nonoperatively or operatively?
Midsubstance total ruptures (grade III) of the MCL can be treated operatively 28 (grade C; agreement, 91%).
8. Should total ruptures (grade III) located at the tibial insertion of the MCL during combined ACL/MCL injury be treated nonoperatively or operatively?
Tibial total ruptures (grade III) of the MCL, including and not limited to Stener-like lesions, 8 can be treated operatively 9,28 (grade C; agreement, 100%).
9. Should tibial or femoral bony avulsions of the MCL during combined ACL/MCL injury be treated nonoperatively or operatively?
Tibial and femoral bony avulsions of the MCL may be treated operatively 20 (grade D; agreement, 100%). Screws with a spike washer 45 and/or suture anchors 20,45 may be used to refix bony avulsions of the MCL (grade D; agreement, 100%).
10. Should partial ruptures (grades I and II) of the MCL during combined ACL/MCL injury be treated nonoperatively or operatively?
Partial tears (grades I and II) of the MCL during combined ACL/MCL injury should be treated nonoperatively 26,46 (grade A1; agreement, 91%).
Nonoperative Therapy
11. Should a brace be used for nonoperative treatment of the MCL in combined ACL/MCL injury?
If nonoperative treatment of the MCL in combined ACL/MCL injury is feasible, the patient can wear a brace for 6 weeks ∥ (grade C; agreement, 100%).
12. Should range of motion be limited during nonoperative treatment of the MCL in combined ACL/MCL injury?
Multiple nonoperative treatment regimens regarding range of motion are reported, with no evidence supporting one option over the other. 22,23,26,46 In the absence of reliable evidence, it is the opinion of this working group that during nonoperative treatment of the MCL in combined ACL/MCL injury, range of motion might be limited for 6 weeks. A feasible treatment regime might include the following: (1) 0 to 2 weeks postinjury, full knee extension to 10° knee flexion; (2) 2 to 4 weeks postinjury, full knee extension to 60° knee flexion; and (3) 4 to 6 weeks postinjury, full knee extension to 90° knee flexion. Patients might be reevaluated frequently, and the treatment regime might be individually adapted to prevent stiffness or joint instability (grade E; agreement, 87%).
13. How much weight should the patient bear during nonoperative treatment of the MCL in combined ACL/MCL injury?
There is conflicting evidence regarding nonoperative weightbearing regimens, with studies promoting direct full weightbearing, no weightbearing, or partial weightbearing. 22,23,46,61,62 In the absence of reliable evidence, it is the opinion of this working group that in a valgus malaligned knee, partial weightbearing might be required, whereas in a varus malaligned knee or in straight legs, full weightbearing might be tolerated (grade E; agreement, 96%).
14. At what point should physical therapy be initiated during nonoperative treatment of the MCL in combined ACL/MCL injury?
In the absence of reliable evidence, it is the opinion of this working group that physical therapy might start as early as possible during nonsurgical treatment of the MCL in combined ACL/MCL injury (grade E; agreement, 91%).
15. At what point should success of nonoperative treatment be evaluated during nonoperative treatment of the MCL in combined ACL/MCL injury?
In the absence of reliable evidence, it is the opinion of this working group that if nonoperative treatment of the MCL in combined ACL/MCL injury is initiated, success of nonoperative treatment might be initially evaluated after 6 weeks (grade E; agreement, 100%).
16. At what point after successful nonoperative treatment of the MCL in combined ACL/MCL injury should the ACL be treated surgically?
In the absence of reliable evidence, it is the opinion of this working group that if nonoperative treatment of the MCL is successful, the ACL might be reconstructed at least 6 weeks after initiation of nonoperative treatment, as long as full range of motion is intact (grade E; agreement, 96%).
17. When should nonoperative treatment of the MCL in combined ACL/MCL injury be considered inefficient?
In the absence of reliable evidence, it is the opinion of this working group that nonoperative treatment of the MCL in combined ACL/MCL injury might be inefficient if patients continue to experience persistent medial grade 2+ or 3+ instability based on the Hughston classification in full extension and/or at 20° to 30° of knee flexion and/or instability during daily activities or exercise (grade E; agreement, 100%). 27
18. When should nonoperative treatment of the ACL in combined ACL/MCL injury be considered inefficient?
In the absence of reliable evidence, it is the opinion of this working group that nonoperative treatment of the ACL should be considered inefficient if a persistent Lachman test grade II/III and/or a pivot-shift test grade II/III and/or instability during daily activities or sport is present (grade E; agreement, 100%).
Operative Therapy
19. How should the MCL be treated if nonoperative treatment of the MCL in combined ACL/MCL injury fails?
If nonoperative treatment of the MCL and/or ACL fails, the failed ligament can be reconstructed 12,38,72 (grade C; agreement, 100%).
20. At what point should an acute total rupture of the MCL in combined ACL/MCL injury be treated operatively?
In the absence of reliable evidence, it is the opinion of this working group that acute total ruptures of the MCL including the sMCL, dMCL, and POL in combined ACL/MCL injury might be surgically addressed as early as possible (grade E; agreement, 96%).
21. Which technique should be used to treat acute femoral or tibial total ruptures of the MCL in combined ACL/MCL injury?
Acute tibial and femoral total ruptures of the MCL can be sutured and/or refixed 28,61,62 (grade C; agreement, 100%).
22. Which technique should be used to treat acute total ruptures of the MCL located at the midsubstance in combined ACL/MCL injury?
Acute midsubstance total ruptures of the MCL can be treated with ligament bracing 5 and/or suturing 28,61,62 (grade C; agreement, 91%).
In the absence of reliable evidence, it is the opinion of this working group that a suture tape might be used for bracing the sMCL and that the suture tape might be fixed with anchors and/or screws in an isometric position (grade E; agreement, 83%).
23. If the MCL is reconstructed in combined ACL/MCL injury, should all parts of the MCL (sMCL, dMCL, and POL) be addressed?
If the MCL is reconstructed in combined ACL/MCL injury, the sMCL and POL can be reconstructed 11,36,37 (grade B1; agreement, 100%).
24. Which grafts are desirable to use for reconstruction of the MCL in combined ACL/MCL injury?
In the absence of reliable evidence, it is the opinion of this working group that autogenic hamstring tendon grafts 32,37,50,70 might be used to reconstruct the MCL. Options might include the autogenic peroneus split graft or allografts. A good option to avoid weakening the medial side of the knee by graft harvesting might entail using tendons of the contralateral side. 50 The MCL graft choice in combined ACL/MCL injury might be influenced by the ACL graft choice, leg alignment, and type of sport (grade E; agreement, 100%).
25. Which grafts are desirable to use for reconstruction of the ACL in combined ACL/MCL injury?
In the absence of reliable evidence, it is the opinion of this working group that autogenic ipsilateral hamstring tendon grafts might not be used for reconstruction of the ACL in combined ACL/MCL injury. Options might include the ipsilateral autogenic quadriceps tendon, ipsilateral patellar tendon, contralateral hamstring tendons, or allografts (grade E; agreement, 96%).
26. What is the role of osteotomy in MCL injury in combined ACL/MCL injury?
In the absence of reliable evidence, it is the opinion of this working group that in chronic MCL insufficiency and/or revisions in patients with >5° valgus malalignment, an osteotomy, either single- or multistaged, might be considered (grade E; agreement, 100%).
Postoperative Follow-up Treatment
27. Should a brace be used after operative treatment of the MCL in combined ACL/MCL injury?
After surgical treatment of the MCL in combined ACL/MCL injury, the patient can wear a brace. Time for wearing the brace varies in the literature between 2 and 9 weeks, most commonly 6 weeks ¶ (grade C; agreement, 96%).
28. Should range of motion be limited after operative treatment of the MCL in combined ACL/MCL injury?
Multiple postoperative treatment regimens regarding range of motion are reported, with no evidence supporting one option over the other. # In the absence of reliable evidence, it is the opinion of this working group that after surgical treatment of the MCL in combined ACL/MCL injury, range of motion might be limited for 6 weeks. A feasible treatment regimen might include the following: (1) 0 to 2 weeks postoperative, full knee extension to 10° knee flexion; (2) 2 to 4 weeks postoperative, full knee extension to 60° knee flexion; and (3) 4 to 6 weeks postoperative, full knee extension to 90° knee flexion (grade E; agreement, 91%).
29. How much weight should the patient bear after operative treatment of the MCL in combined ACL/MCL injury?
There is conflicting evidence regarding postoperative weightbearing regimens, with studies promoting direct full weightbearing, no weightbearing, or partial weightbearing.** In the absence of reliable evidence, it is the opinion of this working group that the patient may bear partial weight for 6 weeks after surgical treatment of the MCL in combined ACL/MCL injury (grade E; agreement, 100%).
30. At what point should physical therapy be initiated after operative treatment of the MCL in combined ACL/MCL injury?
Physical therapy can start as early as possible after surgical treatment of the MCL in combined ACL/MCL injury 22 (grade C; agreement, 96%).
Discussion
The most important finding of this survey study is that for all statements, an agreement of >80% among committee members could be achieved. This implies that there seems to be a high agreement among the investigated cohort of specialized orthopaedic surgeons regarding the treatment of combined ACL/MCL ruptures. In the absence of clear scientific evidence for everyday clinical practice, this survey can thus serve as a clear orientation to optimize the treatment of combined injuries of the ACL and MCL. The following section describes the important statements that were addressed with this modified Delphi process.
A detailed history and physical examination are necessary. According to the evidence-based clinical practice guideline for the management of ACL injuries adopted by the American Academy of Orthopaedic Surgeons, 58 the history should include, at minimum, the mechanism of injury, history of hearing/feeling a popping sensation, ability to bear weight, ability to return to play, history of mechanical symptoms of locking or catching, localization of pain if possible, and any history of prior knee injuries. 13,35,52 A history of hearing or feeling a popping sensation with associated swelling is important in suspecting or predicting an ACL injury. 49 A physical examination should include, at minimum, a neurovascular examination with documentation of distal perfusion and tibial/peroneal nerve function, assessment for joint line tenderness or obvious stepoff/deformity, evaluation for an effusion, assessment of varus and valgus laxity at 0° and 30° of extension, and evaluation of anterior-posterior and rotational laxity. 3,15,30,59,63 A Lachman test should be performed, which has been shown to be sensitive for diagnosing an ACL injury. 7 An MRI should be performed to establish a diagnosis. In chronic cases, it can be useful to take radiological stress images.
Total ruptures of the MCL, irrespective of whether they occur at the femoral insertion, tibial insertion, or midsubstance, can be treated operatively in the context of combined injuries with the ACL. However, in a rabbit model, MCLs, injured near either end, healed more slowly than those injured in their midsubstance and developed unphysiological insertion morphology. 16 An in vitro study showed that single-bundle ACL reconstruction alone was not able to restore anterior tibial translation, valgus rotation, and external rotation of the intact knee with combined ACL and sMCL injuries. 73 The in situ forces in the ACL graft in response to a valgus torque of 5 Nċm at 60° of knee flexion are almost 2.5 times higher in the ACL-reconstructed knee with MCL deficiency than in the intact knee. 41 In a sheep model with a combined ACL/MCL injury, the ACL graft stabilized the knee initially but became loose over time. As a result, the nonsurgically treated, healing MCL may have been required to take on excessive loads and was unable to heal sufficiently compared with an isolated MCL injury. 1
According to the working group, the acute total rupture of the MCL with all its components should be treated operatively as soon as possible. Femoral and tibial ruptures can be sutured and reattached. Tibial and femoral bony avulsions of the MCL may be refixed using screws with a spikewasher or suture anchors. Care must be taken not to confuse Pellegrini-Stieda lesions with an acute bony avulsion. Pellegrini-Stieda lesions are calcifications in the soft tissue at the medial side of the knee, often located near the medial femoral condyle, resulting from macrotrauma or repetitive microtrauma. 43,67 In the event of ruptures being localized to the midsubstance, the sMCL can undergo intraligamentous bracing and/or be sutured with a tape after isometric testing has been performed. In contrast, partial ruptures of the MCL should be treated nonoperatively.
Commonly accepted indications for surgical treatment of the ACL, in most cases mainly triggered by an instability in daily life or during sporting activities, are not influenced by an additional MCL injury. However, the need for ACL surgery is certainly more frequent, because the instability is usually increased, especially with total ruptures of the MCL.
In the opinion of the working group, for nonoperative treatment as well as for postoperative follow-up treatment, the range of knee joint motion should be limited by a semirigid brace under partial (if necessary) or full weightbearing.
The importance of a limited range of motion on MCL healing becomes clear when reviewing the function of the individual parts of the MCL. In neutral rotation of the knee, none of the parts of the MCL—neither the sMCL, the dMCL, nor the POL—experiences an increase in length with the knee in 10° of knee flexion compared with full extension. First, the anterior fibers of the sMCL increase in length when the knee flexes >10°. Then the length of the sMCL increases gradually. The anterior fibers of the dMCL start increasing their length at approximately 60° of knee flexion. The posterior fibers of the sMCL, the posterior fibers of the dMCL, and the POL do not increase in length during the path of knee flexion and extension. In contrast, they become more lax with increasing knee flexion angle. 69
Physical therapy should start as early as possible. The success of nonoperative MCL treatment should be checked by manual valgus stress testing initially after 6 weeks. The ACL should therefore undergo surgery at least 6 weeks after the initiation of nonoperative therapy for the MCL. Free range of motion of the knee joint should also have been achieved. Persistent grade 2+ to 3+ instabilities based on the Hughston classification and especially clearly perceptible instabilities during daily and sporting activities are a clear sign of a failure. 27
A recent project defining a successful outcome after ACL injury and reconstruction achieved a consensus for the following measures in operative and nonoperative management:
The absence of giving way
Return to sports
Quadriceps and hamstrings strength >90% of the uninvolved limb
No more than a mild knee joint effusion
Patient-reported outcomes 40
In chronic MCL insufficiencies, both the sMCL and the POL can be reconstructed with a tendon graft. The choice of MCL graft for combined ACL/MCL injuries can be influenced by the choice of ACL graft, the leg axis, and the type of sport. Ipsilateral hamstring tendons should be avoided as a graft whenever possible in order to avoid further weakening the medial side of the knee joint. In a human cadaveric study, semitendinosus and gracilis muscles stabilized valgus moments applied to the MCL-insufficient knee with the knee near full extension. 25 A varus corrective osteotomy can be considered for chronic MCL insufficiency and/or revisions in patients with >5° valgus, either single- or double-staged. An osteotomy leads to the loss of medial thrust on weightbearing in chronic MCL laxity. 6
This consensus has some limitations. Carrying out the process online resulted in a large spatial distance between the committee members. Consequently, the major part of communication was limited to comments made within the framework of the questionnaire. Therefore, no open discussion of questions and statements was possible. However, this might also be an advantage, because the possible biasing influence of opinion-leading individuals in the decision-making process was naturally limited.
Another limitation of this study is that the working group consisted exclusively of trauma surgeons and orthopaedic surgeons. The lack of colleagues who use only nonoperative treatments could implicate a tendency toward operative care.
The committee members do not have any financial advantage from operative care of the MCL. Based on the German diagnosis-related groups (DRG)-based remuneration system, a combination of ligament interventions (as many combined procedures in general) is not remunerated more than the single intervention of a single ACL reconstruction.
Care was taken so that the creation of the statements did not conflict with the scientific evidence. The individual opinion may therefore have been influenced by literature. The level of evidence also influenced the wording of the statements. Hence, some statements were created as “optional” rather than “should” or “must” statements.
Conclusion
The results of this modified Delphi process offer a guideline for standardized patient care in the context of combined injuries to the ACL and MCL. This may mean that less efficient and/or unnecessary diagnostic and/or therapeutic instruments or interventions are limited, potentially reducing the cost of the individual treatment in the future.
Finally, a possibly lower failure rate, which could be achieved by optimizing the therapy, could result in lower costs for the health care system.
Footnotes
Notes
Authors
Thomas Pfeiffer, MD (Department of Orthopaedic Surgery, Trauma Surgery, and Sports Medicine, Cologne-Merheim Medical Center [CMMC], Witten-Herdecke University, Cologne, Germany); Wolf Petersen, MD (Klinik für Orthopädie und Unfallchirurgie, Martin Luther Krankenhaus, Berlin, Germany); Andreas Imhoff, MD (Department of Orthopaedic Sports Medicine, Hospital Rechts der Isar, Technical University of Munich [TUM], Munich, Germany); Mirco Herbort, MD (OCM Clinic Munich, Munich, Germany); Andrea Achtnich, MD (Department of Orthopaedic Sports Medicine, Hospital Rechts der Isar, Technical University of Munich [TUM], Munich, Germany); Thomas Stein, MD (Sporthologicum Frankfurt, Center for Sport & Joint Injuries, Frankfurt am Main, Germany; Department of Sports Medicine, Goethe University Frankfurt, Frankfurt am Main, Germany); Christoph Kittl, MD (Department of Trauma, Hand and Reconstructive Surgery, Westphalian Wilhelms University Muenster, Muenster, Germany); Christian Schoepp, MD (Department of Arthroscopic Surgery, Sports Traumatology, and Sports Medicine, BG Klinikum Duisburg gGmbH, Duisburg, Germany); Ralph Akoto, MD (Department of Trauma Surgery, Orthopaedics and Sports Traumatology, BG Hospital Hamburg, Hamburg, Germany; Department of Orthopaedic Surgery, Trauma Surgery, and Sports Medicine, Cologne-Merheim Medical Center [CMMC], Witten-Herdecke University, Cologne, Germany); Jürgen Höher, MD (Sportsclinic Cologne, Cologne, Germany; Department of Orthopaedic Surgery, Trauma Surgery, and Sports Medicine, Cologne-Merheim Medical Center [CMMC], Witten-Herdecke University, Cologne, Germany); Sven Scheffler, MD (Sporthopaedicum Berlin, Medizinische Hochschule Brandenburg Theodor-Fontane [MHB], Berlin, Germany); Amelie Stöhr, MD (OCM Clinic Munich, Munich, Germany); Thomas Stoffels, MD (OC Stadtmitte, Berlin, Germany); Julian Mehl, MD (Department of Orthopaedic Sports Medicine, Hospital Rechts der Isar, Technical University of Munich [TUM], Munich, Germany); Tobias Jung, MD (Center for Musculoskeletal Surgery, Charité - University Medicine Berlin, Germany); Andree Ellermann, MD (Arcus Clinics Pforzheim, Pforzheim, Germany); Christian Eberle, MD (Arcus Clinics Pforzheim, Pforzheim, Germany); Cara Vernacchia, DO (Physical Medicine and Rehabilitation, Northwestern University Shirley Ryan Ability Lab, Chicago, Illinois, USA); Patricia Lutz, MD (Department of Orthopaedic Sports Medicine, Hospital Rechts der Isar, Technical University of Munich [TUM], Munich, Germany); Matthias Krause, MD (Department of Trauma and Orthopaedic Surgery, University Medical Center, Hamburg-Eppendorf, Hamburg, Germany); Natalie Mengis, MD (Arcus Clinics Pforzheim, Pforzheim, Germany); Peter E. Müller, MD (Department of Orthopaedic Surgery, Physical Medicine and Rehabilitation, University Hospital of Munich [LMU], Munich, Germany); Thomas Patt, MD (Orthopedium, Delft, the Netherlands); Raymond Best, MD (Sportsclinic Stuttgart, Department of Orthopaedic and Sports Trauma Surgery, Stuttgart, Germany; University of Tuebingen, Department of Sportsmedicine, Tuebingen, Germany).
Final revision submitted July 15, 2021; accepted July 26, 2021.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
