Abstract
Background:
Recent evidence suggests that there may be an increased risk of infection for patients undergoing a corticosteroid injection before, during, or after knee arthroscopy.
Purpose:
To systematically review the literature to evaluate the risk of postoperative infection in patients undergoing intra-articular corticosteroid injections (CSI) before, during, or after knee arthroscopy.
Study Design:
Systematic review; Level of evidence, 3.
Methods:
A systematic review was performed by searching the PubMed, Cochrane Library, and Embase databases to identify studies that evaluated the rate of postoperative infection in patients undergoing knee arthroscopy who received an intra-articular CSI during the perioperative period. The search phrase used was “knee AND arthroscopy AND injection AND (infection OR revision).” A subanalysis was also performed to analyze infection rates based on the timing of the corticosteroid injection in relation to arthroscopy.
Results:
Four studies met the inclusion criteria, representing 11,925 patients undergoing knee arthroscopy with an intra-articular CSI administered during the perioperative period (mean follow-up, 5.3 months) and 247,329 patients without a corticosteroid injection during the perioperative period (mean follow-up, 5.9 months). Patients who received an injection experienced a statistically significantly higher rate of postoperative infection (2.2%) when compared with patients who did not receive an injection (1.1%; P < .001). When analyzed by the timing of the injection, patients receiving an injection preoperatively or intraoperatively experienced a statistically significantly higher rate of postoperative infection (3% and 2.6%, respectively) when compared with patients receiving an injection postoperatively (1.4%; P = .001 for both).
Conclusion:
Patients undergoing knee arthroscopy who receive an intra-articular CSI during the perioperative period can be expected to experience significantly higher postoperative infection rates when compared with patients not receiving an injection. Furthermore, patients receiving a corticosteroid injection pre- or intraoperatively may experience significantly higher rates of postoperative infection when compared with patients receiving an injection postoperatively.
Intra-articular corticosteroid injections (CSIs) are a commonly employed adjunct in both the diagnosis and treatment of knee pathology. Given the ubiquity of knee arthroscopy, 12,22 it is important to investigate the risk of infection in patients undergoing arthroscopy who received an intra-articular CSI in the perioperative period. 2,5 The potent anti-inflammatory properties of corticosteroids are effective in relieving pain secondary to intra-articular pathology, 26 reducing postoperative analgesic consumption and pain scores, 13,20 and elucidating the origin of knee pain for clinical decision-making purposes. 15 However, there is some concern that corticosteroids suppress the intra-articular immune response, making the joint vulnerable to postoperative infection. 29 , 30
The increased risk of infection after preoperative CSI has been validated in the arthroplasty literature. 7,21,28 The question has similarly been investigated in patients undergoing shoulder, 29 ankle, 30 hip, 27 and knee arthroscopy. 2,5 Early series demonstrating corticosteroid-associated infection risk in patients who underwent knee arthroscopy were small and underpowered because of the low overall incidence of infection after knee arthroscopy, 2,19 estimated to be between 0.07% and 0.42%. 5 Recent studies, although more highly powered, demonstrate heterogeneity in both the timing and clinical outcomes of perioperative intra-articular injection. 4,6,27 The risk and optimal timing of CSI in the arthroscopy population therefore remains unclear. The purpose of this study was to systematically review the literature to evaluate the risk of postoperative infection in patients undergoing intra-articular CSI before, during, or after knee arthroscopy. The authors hypothesized that patients receiving a corticosteroid injection in the perioperative period would be at a higher risk of infection after knee arthroscopy.
Methods
This systematic review was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines using a PRISMA checklist. Two independent reviewers (J.W.B., L.E.K.) searched the PubMed, Embase, and Cochrane Library databases up to December 10, 2020. The electronic search strategy used was as follows: “knee AND arthroscopy AND injection AND (infection OR revision).” A total of 72 studies were reviewed by title and/or abstract to determine study eligibility based on inclusion criteria. In cases of disagreement, a third reviewer (M.J.K.) made the final decision. Inclusion and exclusion criteria followed the participants, interventions, comparators, outcomes, study (PICOS) design strategy. Studies selected for inclusion met the following criteria: (1) participants: patients diagnosed with intra-articular knee pathology; (2) intervention: intra-articular injections of corticosteroid and subsequent or preceding arthroscopic procedure; (3) comparator: no intra-articular injection in the pre- or perioperative period; and (4) outcomes: incidence of postoperative infection; and (5) study design: studies with evidence levels 1 to 4 that were published in English. The exclusion criteria included studies that did not meet the aforementioned inclusion criteria. Four studies were determined to meet the inclusion criteria (Figure 1). To ensure that studies from the same group did not include overlapping patients, each study's lead author was contacted and asked to verify that the patient populations from each study were completely independent of one another. This was performed as described for the 2 papers of Cancienne et al. 5,6 Data extraction from each study was performed independently (J.W.B.) and then reviewed by a second author (L.E.K.). There was no need for funding or a third party to obtain any of the collected data. Risk of bias was assessed according to the risk of bias in nonrandomized studies of interventions (ROBINS-I) risk of bias tool, 24 which incorporates an assessment of bias because of confounding, selection of participants, deviations from intended interventions, completeness of outcome data, selection of outcomes reported, and other sources of bias.
Study Methodology Assessment
The modified Coleman Methodology Score (MCMS) 8 was used to evaluate study methodology quality. The MCMS score is a reliable way to evaluate the quality of the included studies based on a number of factors, including study design, sample size, methodology, included descriptions, and so forth. The MCMS has a scaled potential score ranging from 0 to 100. Scores ranging from 85 to 100 are excellent, 70 to 84 good, 55 to 69 fair, and <55 poor.
Statistical Analysis
A weighted average was calculated for numerical demographics (percentage male, follow-up). Chi-square tests were used to calculate statistical significance for infection data.
Results
Four studies (1 with level 1 and 3 with level 3 evidence) met the inclusion and exclusion criteria (Figure 1), including a total of 259,254 patients who underwent knee arthroscopy (CSI, 11,925; non-CSI, 247,329). The mean follow-up time was 5.3 months for the CSI group and 5.9 months for the non-CSI group, and the percentage of men was 36.3% and 35% in the CSI and non-CSI groups, respectively (Table 1). Only 1 study 14 reported on patient age, in which the mean age of patients who received CSI was 49 ± 11 years (range, 19-68 years).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Studies Included in the Review a
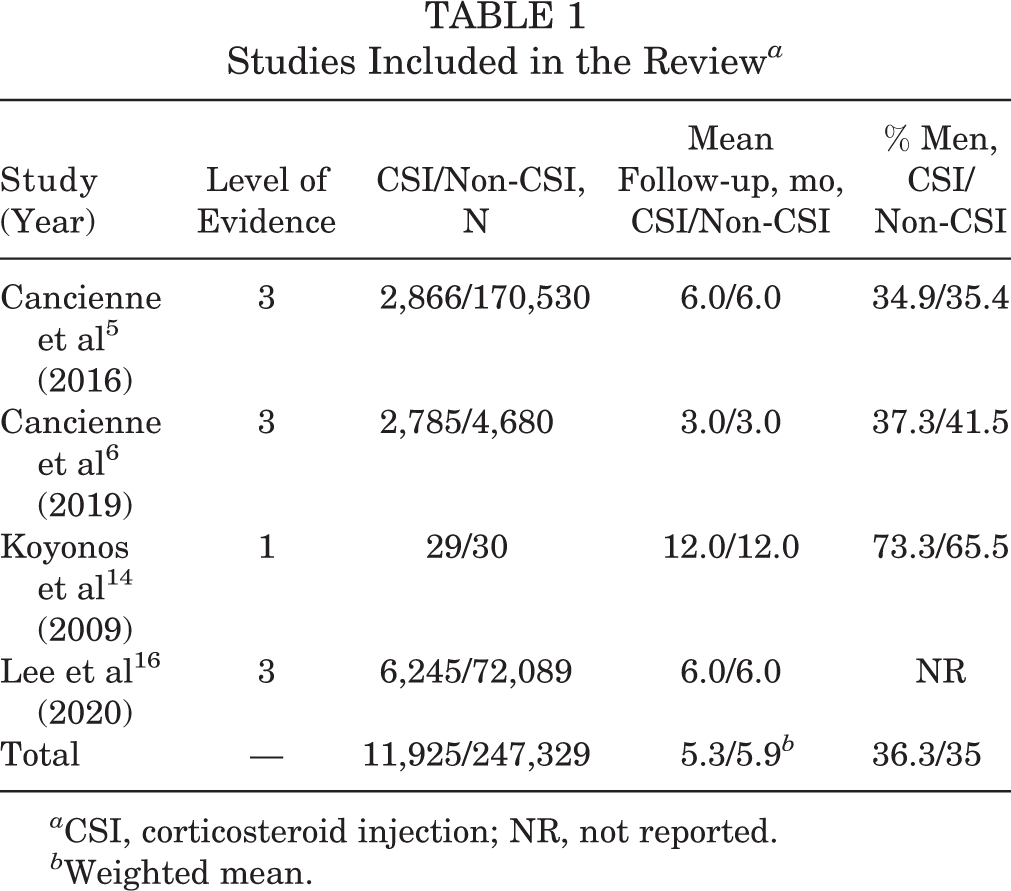
a CSI, corticosteroid injection; NR, not reported.
b Weighted mean.
Corticosteroid Formulation
In 2 studies, 5,6 patients received intra-articular injections of 1 mg triamcinolone acetonide, 10 mg triamcinolone acetonide, 20 mg methylprednisolone acetate, 40 mg methylprednisolone acetate, or 80 mg methylprednisolone acetate. One study 14 reported administering 40 mg methylprednisolone to patients who received CSI, and 1 study 16 did not specify corticosteroid formulation.
Injection Technique
No studies described the injection administration technique.
Modified Coleman Methodology Score
Table 2 shows the MCMS scores from the 4 included studies. Three studies 5,6,16 received fair scores and 1 study 14 received a good score.
Modified Coleman Methodology Score a
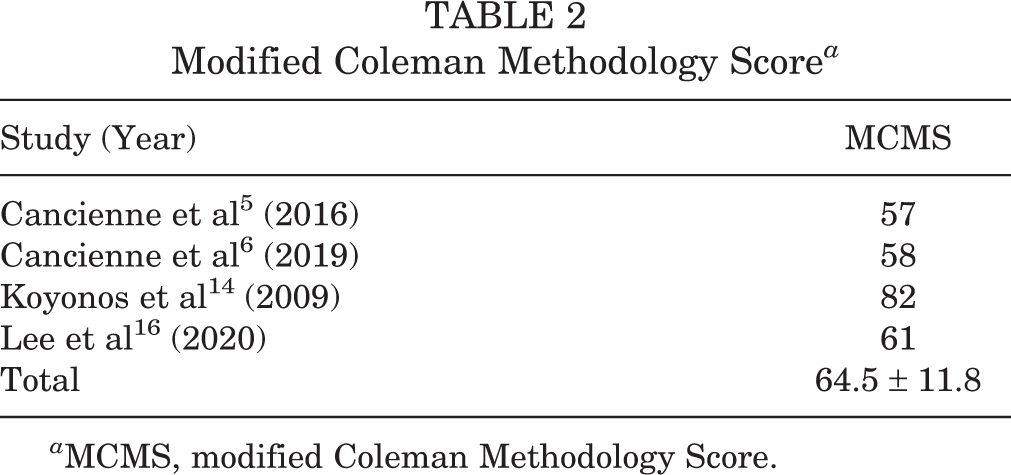
a MCMS, modified Coleman Methodology Score.
Characteristics
All 4 studies 5,6,14,16 reported no significant differences in age between CSI and non-CSI groups, 3 studies 5,14,16 reported no differences in sex, 2 studies 5,6 reported no differences in obesity, and 2 studies 5,6 reported no differences in smoking status or diabetes. All 4 studies 5,6,14,16 reported no differences in type of the arthroscopy procedure performed. One study 16 reported a significant difference in both diabetes and smoking status, in which the injection group included a significantly higher proportion of diabetic patients and smokers (P < .05).
Procedures Performed
One study 14 included only patients undergoing arthroscopic meniscectomy, while another study 6 included patients undergoing either arthroscopic meniscectomy or plica debridement. Two studies 5,16 included any patient undergoing arthroscopic meniscectomy, debridement, loose body removal, and/or microfracture.
Methodologic Quality Assessment
The results of the methodologic quality assessment of the 4 included studies using the ROBINS-I risk of bias tool are presented in Figure 2. All 4 studies 5,6,14,16 showed a moderate risk of bias because of confounding, as there were no prognostic variables that predicted baseline intervention and no patients who switched between interventions during the study period. No studies excluded eligible patients or used variable follow-up times based on intervention (low risk of bias), no studies deviated from the intended intervention (low risk of bias), and all studies clearly classified treatment type (low risk of bias). Only 1 of the 4 studies 14 described the use of a blinded outcome assessor (low risk of bias), although identical postoperative protocols were used in each cohort in the remaining 3 studies 5,6,16 (moderate risk of bias). No studies showed bias because of missing data (low risk of bias). Because of the nature of the primary outcome (infection), all 4 studies 5,6,14,16 demonstrated serious risk of bias because of the use of the treating physician as the nonblinded outcome assessor. Finally, no studies showed bias because of selective reporting (low risk of bias). A Cohen kappa score of 0.82 reflected a very good agreement between reviewers.

Risk of bias graph. Risk of bias is presented as a percentage across all included studies.
Infection Rates
All 4 studies 5,6,14,16 reported on infection rates. Only 1 study 16 described how infection was diagnosed, which was defined as infected postoperative seroma, pyogenic arthritis, or cellulitis. The remaining 3 studies 5,6,14 did not specify how infection was defined. In 3 studies, 5,6,16 the Current Procedural Terminology and Clinical Modification codes were used to identify patients who experienced an infection postoperatively. Overall, 3 of the 4 studies 5,6,16 found a significantly higher rate of infection at the latest follow-up among patients who received an intra-articular CSI before, during, or after arthroscopy (P < .05) (Table 3).
Infection Rates a

a Infections are reported as number of infections/total number of patients (%) at the latest follow-up. CSI, corticosteroid injection.
Infection Rates by Timing of Injection
The timing intervals between injection and arthroscopy are listed in Table 4. Patients were allocated to 1 of the 3 distinct groups depending on if they received their injection pre-, intra-, or postoperatively.
Time between Corticosteroid Injection and Arthroscopy

The overall postoperative infection rate for patients receiving an intra-articular CSI preoperatively was 3% compared with 2.6% if administered intraoperatively (P = .85) and 1.4% if administered postoperatively (preoperatively vs postoperatively P = .001; intraoperatively vs postoperatively; P = .001) (Table 5).
Infection Rates by Timing of Injection a

a Infections are reported as number of infections/total number of patients (%) at the latest follow-up.
Discussion
Intra-articular CSI is commonly employed in the treatment of knee pathology and often precedes operative intervention. Given the potential implications, it is important to clarify the relationship between CSI and postoperative infection in patients undergoing arthroscopic procedures. The results of this systematic review suggest that patients undergoing knee arthroscopy who receive an intra-articular CSI perioperatively experience significantly higher postoperative infection rates when compared with patients not receiving an injection. Additionally, preoperative and intraoperative injections are associated with significantly higher postoperative infection rates as compared with injections administered postoperatively.
Corticosteroids are frequently used in the nonoperative treatment of chondral and meniscal pathology and have proven efficacious in pain relief. 11,25,26 When nonoperative treatment fails, some surgeons advocate for intra-articular CSI at the time of arthroscopy, as the anti-inflammatory properties of corticosteroids are thought to mitigate the pain of inflamed synovial tissue and contribute to hemostasis. 10 This practice has been shown to reduce postoperative analgesic consumption and pain scores and hasten recovery. 13,14,20 Although intra-articular CSI does provide demonstrable clinical benefit, there remains concern that perioperative injection increases the risk of postoperative infection.
Early studies of infection rates in patients undergoing knee arthroscopy demonstrated a significant association between intra-operative CSI and postoperative infection. 1,2,19 Armstrong et al 1 reviewed 4,256 knee arthroscopies and reported an infection rate of 0.42%, noting that the most significant risk factor for postoperative infection was intraoperative injection of methylprednisolone. Other risk factors for postoperative infection included prolonged surgical time, increased number of procedures during surgery, and history of prior procedures on the ipsilateral knee. 1 The authors proposed that corticosteroid-induced suppression of the local immune response predisposes the joint to infection, a hypothesis that has since been endorsed by others. 2,3,13,20,27 Chronic systemic steroid use has also been associated with significantly increased complication rates after arthroscopy. 9,31 Westermann et al 31 noted a 3-fold increased rate of readmission because of surgical-site infection and wound complications in patients with chronic steroid use who had knee and shoulder arthroscopy.
Despite these early findings, subsequent studies demonstrated no increased risk of infection in patients undergoing arthroscopy who received CSI in the perioperative period. 4,14,20,23 In a randomized study of 59 knees, Koyonos et al 14 demonstrated no increased infection risk in patients undergoing intra-articular CSI at the time of meniscectomy compared with patients receiving no steroid. Similarly, in their respective cohorts of 60 and 77 patients undergoing CSI at the time of knee arthroscopy, Rasmussen et al 20 and Smith et al 23 reported no postoperative infections. While these studies highlight the clinical benefit of intraoperative CSI, each was likely insufficiently powered to detect a difference in infection rates because of the low overall incidence of infection after arthroscopy. Rates of infection after knee arthroscopy are estimated to be between 0.07% and 0.42% 5 and are thus unlikely to be adequately evaluated in small cohorts. The present systematic review includes a substantially larger sample size, enabling an analysis sufficiently powered for the detection of significant differences in infection rates after arthroscopy.
In the present systematic review, 3 of the 4 included studies found a significantly higher rate of infection in patients who received an injection in the perioperative period. 5,6,16 Similar findings have been demonstrated in the arthroplasty literature 7,21,28 as well as in patients undergoing shoulder arthroscopy, 29 with significantly decreased rates of infection noted when CSI is performed >3 months before surgery. This 3-month duration may relate to the biologic half-life of injected corticosteroids within large joints. 18 However, this relationship has not been clearly validated, 17,27 and the optimal timing of perioperative CSI in the setting of arthroscopy remains unclear. Lee et al 16 demonstrated an increased risk of postoperative infection in patients undergoing CSI within 2 weeks of knee arthroscopy but found no increased risk of infection outside of this window. Similarly, Cancienne et al 6 noted an increased risk of infection in patients injected within 4 weeks after knee arthroscopy yet demonstrated no increased infection risk after this time period. The pooled analysis of the studies included in the present systematic review demonstrated a significantly decreased infection risk in patients undergoing CSI postoperatively compared with pre- or intraoperatively.
Overall, the results of this systematic review demonstrate a 2.2% risk of infection in patients undergoing perioperative CSI compared with a 1.1% risk in patients not undergoing injection. While the absolute risk of postoperative infection remains low in both groups, perioperative CSI appears to approximately double the risk of infection in patients undergoing knee arthroscopy. When considering CSI in the perioperative period, surgeons should evaluate comorbidities and other risk factors for infection, the patient’s pain level, and the diagnostic or therapeutic benefit potentially afforded by CSI. Surgeons should avoid administering injections shortly before or during arthroscopy procedures to reduce the risk of infection. Ultimately, the decision to pursue CSI is a risk-benefit analysis that should entail discussion with the patient, and CSI should not be portrayed to patients as a risk-free procedure.
The strengths of this systematic review include the use of comparative studies with low levels of bias. The limitations of this study should also be noted. Most notably, there were only 4 primary studies included in this review. There was heterogeneity among studies in the timing and formulation of corticosteroid injection, and only 1 study provided an explicit definition of postoperative infection. 16 Only 1 of the 4 included studies, which was likely underpowered because of its small sample size, was of level 1 evidence, and none of the included studies used blinding in their methodology. Additionally, the differences in study methodology, sample size, and inherent bias among the included studies may contribute to the differences in infection rates observed in the present review. Finally, surgical time and perioperative antibiotic coverage were assessed in the included studies. Further high-quality, randomized studies are needed to better elucidate the optimal timing of CSI in the perioperative period.
Conclusion
Patients undergoing knee arthroscopy who receive an intra-articular CSI during the perioperative period can be expected to experience significantly higher postoperative infection rates when compared with patients not receiving an injection. Furthermore, patients receiving a corticosteroid injection pre- or intraoperatively may experience significantly higher rates of postoperative infection when compared with patients receiving an injection postoperatively.
Footnotes
Final revision submitted March 8, 2021; accepted April 14, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: O.M.-D. has received research support, consulting fees, and royalties from Stryker and nonconsulting fees from Smith & Nephew; and has stock/stock options in MITA. A.J.S. has received research support from ISO-Biologics and consulting fees from DePuy. E.C.M. has received research support from Arthrex, Biomet, Breg, Mitek, Ossur, Smith & Nephew, and Stryker; education payments from Gemini Mountain; consulting fees from Zimmer and Medical Device Business Services; speaking fees from Arthrex; and royalties from Elsevier and Zimmer Biomet. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
