Abstract
Background:
It is not clear whether the mechanical strength of adjustable-loop suspension devices (ALDs) in anterior cruciate ligament (ACL) reconstruction is device dependent and if these constructs are different from those of an interference screw.
Purpose:
To compare the biomechanical differences of 2 types of ALDs versus an interference screw.
Study Design:
Controlled laboratory study.
Methods:
ACL reconstruction was performed on porcine femurs and bovine extensor tendons with 3 types of fixation devices: interference screw, UltraButton (UB) ALD, and TightRope (TR) ALD (n = 10 for each). In addition to specimen testing, isolated testing of the 2 ALDs was performed. The loading protocol consisted of 3 stages: preload (static 150 N load for 5 minutes), cyclic load (50-250 N at 1 Hz for 1000 cycles), and load to failure (crosshead speed 50 mm/min). Displacement at different cycles, ultimate failure load, yield load, stiffness, and failure mode were recorded.
Results:
In specimen testing, displacement of the ALDs at the 1000th cycle was similar (3.42 ± 1.34 mm for TR and 3.39 ± 0.92 mm for UB), but both were significantly lower than that of the interference screw (7.54 ± 3.18 mm) (P < .001 for both). The yield load of the UB (547 ± 173 N) was higher than that of the TR (420 ± 72 N) (P = .033) or the interference screw (386 ± 51 N; P = .013), with no significant difference between the latter 2. In isolated device testing, the ultimate failure load of the TR (862 ± 64 N) was significantly lower than that of the UB (1879 ± 126 N) (P < .001).
Conclusion:
Both ALDs showed significantly less displacement in cyclic loading at ultimate failure than the interference screw. The yield load of the UB was significantly higher than that of the other 2. The ultimate failure occurred at a significantly higher load for UB than it did for TR in isolated device testing.
Clinical Relevance:
Both UB and TR provided stronger fixation than an interference screw. Although difficult to assess, intrinsic differences in the mechanical properties of these ALDs may affect clinical outcomes.
Keywords
Many factors affect anterior cruciate ligament (ACL) reconstruction outcomes, but the graft fixation device is a crucial determinant of revision rates after primary ACL reconstruction. 13 In the past, the interference screw was the gold standard fixation system, but it was shown to be associated with complications, such as cyst formation, poor integration, graft migration, or tunnel widening, resulting in knee instability or complete failure. 10,38 As an alternative to the interference screw, especially for use at the femoral site, suspension devices were proposed. Suspension devices have an advantage over interference screws of allowing greater contact between the graft and the internal surface of the tunnel, which translates to better and faster integration of the graft. 38
In recent years, suspension devices with adjustable loops (ALDs) have been introduced. Their main advantage over fixed-loop devices is that they can fill bone tunnels of different lengths, eliminating the need for fixed-loop devices of different loop sizes. However, they represent a concern for some surgeons since some biomechanical studies have concluded that when the system is subjected to daily cyclic loading from knee movements, elongation of the loop can occur and lead to residual laxity. 1,4,12,15,29 Other biomechanical studies have indicated that the elongation of some ALDs is comparable with that of fixed loop devices 26,28,37 ; however, a recent meta-analysis of laboratory studies found that there were significant differences between the biomechanical properties of different types of ALDs and that 1 of the fixed-loop devices (EndoButton CL; Smith & Nephew) showed significantly less cyclic displacement in comparison with that shown by ALDs. 18 Clinical and radiological outcomes have been shown to be comparable between ALD and interference screws, 5,24 and similar performance data for ALDs and fixed-loop devices have been reported. 2,6,21,31 Therefore, there is a lack of consensus regarding the biomechanical properties and clinical outcomes of ALDs in ACL reconstruction.
ACL reconstruction outcomes depend on the type of fixation, environmental biology (graft and bone), and the technical ability of the surgeon. Currently, there is much information about mechanical properties of available ALDs. However, little is known about these mechanical properties when a device is tested in isolation or as a part of the bone-tendon-device ensemble, using the same protocols. The purpose of this study was to compare the biomechanical performance of 3 ACL reconstruction fixation systems using in vitro biomechanical tests: 2 ALDs and an interference screw. We also aimed to assess whether the ALDs performed differently from one another in isolated device testing and if that difference was reproducible in a biological test setting. Our hypothesis was that the 3 systems would show the same biomechanical behavior in terms of fixation strength, cyclic displacement, and stiffness.
Methods
Three fixation devices were tested: UltraButton (UB) ALD (Smith & Nephew), TightRope (TR) ALD (Arthrex), and a 9 × 30—mm titanium 6Al-4 V interference screw (Propel). Two types of in vitro tests were carried out: specimen testing, which tested the complete ACL-femoral reconstruction system of the 3 fixation devices using animal tissues, and isolated device testing, which was used to assess each ALD alone, without taking into account the contribution of the tissues or the tissue-device interface. Because of the operating principles of the devices in this type of test, only the ALDs (UB and TR) could be tested.
Specimen Testing
Thirty digital bovine extensor tendons and the same number of skeletally mature porcine femurs obtained from a local slaughterhouse were used in this study. Bovine tendons were used because they mimic the biomechanical properties of the tendons (semitendinosus and gracilis) used in ACL reconstructions, 11 and porcine femurs were used because they are a reasonable alternative to use of human bone. 20 Tendons were harvested just after the animals were sacrificed, wrapped in gauze soaked in normal saline, and packed in individual bags. Soft tissue was removed from the femurs, and the proximal part was sectioned to fit the custom-made jaw of the specimen testing system. Tendons and bones were frozen at –20°C and were thawed to room temperature 12 hours before testing. The specimens were kept moist throughout the handling period using a nebulizer with normal saline. Tendon diameter was measured using a custom-made caliper. Only 6.5 mm–diameter tendons were used. Then, tendons were folded, obtaining an approximately 9-mm equivalent diameter, and a suture was sewn to each branch of the tendon to ease its handling.
To attach the ALD, a guide pin was drilled into the femur at the level of the ACL origin through the lateral femoral cortex. A 20 mm–long bone tunnel was created from the ACL origin point using a 9 mm–diameter cannulated drill bit then a 4.5 mm–diameter cannulated drill bit with the same guide pin to completely pierce the bone. A single tendon was folded back over the loop of the device, creating a 2-stranded graft, and was introduced into the bone tunnel until the button protruded from the external cortex and flipped, fixing the device to the bone. The loop of the ALD was then adjusted to completely fill the 9-mm tunnel with the graft; manual tension was applied to the graft, according to usual clinical practice. For ACL reconstructions with the interference screw, a full 9 mm–diameter tunnel was drilled in the femur, and the standard introduction technique was applied.
A custom-made fixation apparatus was used in the lower jaw of the testing machine (EFH/5/FR; Microtest SA), allowing a 45° angle between the femur and the vertical axis. Serrated jaws were used to attach the tendons to the upper part of the testing machine. In all ACL reconstructions, approximately 30 mm of tendon was left outside the tunnel, simulating the intra-articular distance of an intact ACL. 30 The testing machine applied load in the direction of the femoral tunnel, which represented the worst-case load scenario (Figure 1).
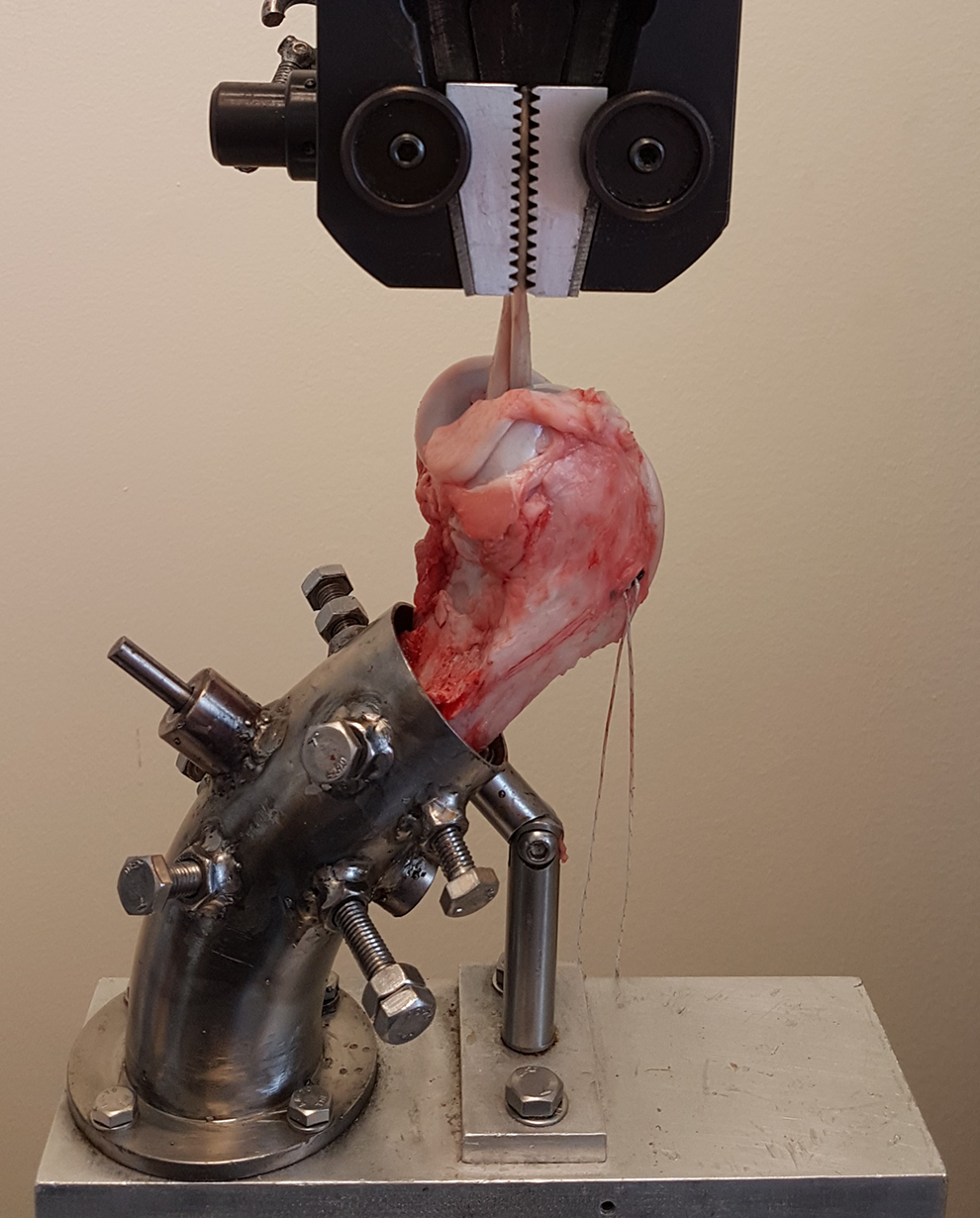
UltraButton anterior cruciate ligament reconstruction in the testing machine.
Ten ACL reconstructions were tested for each type of fixation device at 1 Hz, simulating typical loading frequency when walking.
17
The loading protocol was as follows: Preload: displacement at 20 mm per minute until 150 N and stable load for 5 minutes. Cyclic load: 50 to 250 N at 1 Hz for 1000 cycles. Final ramp: load until failure at 50 mm/min displacement rate.
The preload simulated pretension of the graft to avoid the influence of the initial graft viscoelasticity, allowing focus on the performance of the fixation devices. The first 10 cycles were considered to represent the intraoperative preconditioning of the graft, recommended and performed by many surgeons. The displacement was then zeroed because it is supposed that, at this time, the definitive fixation in the tibia of the graft is carried out, so the relevant clinical displacements occur from this moment onward. The 50- to 250-N loading protocol allows results to be comparable with those of many other researchers. 8,12,29 Moreover, the initial intraoperative tension applied to the graft seems to always maintain a certain amount of tension on the ACL reconstruction, so to mimic an actual rehabilitation protocol, a minimum tension of 50 N was preferred to a null load.
Data for 1 in every 10 cycles were recorded at a sampling rate of 300 Hz. Data were obtained with the displacement sensor and the load cells of the testing machine. Displacement was obtained from the raw data at the medium load of each cycle (150 N) and recorded for cycles 50, 100, 500, and 1000. In the final part of the test, 3 results were obtained: the ultimate failure load, yield load, and stiffness. The ultimate failure load was the maximum load value obtained in the test. Stiffness was defined as the slope of the linear part of the load-displacement graph, assuming linearity when the correlation coefficient was ≥0.99, and yield load was defined as the load at the intersection of the force-displacement graph and the line to determine the stiffness with a 0.2% offset. The mechanism of failure was recorded (cortical blowout, suture tear, tendon failure, or tendon slippage).
Isolated Device Testing
We used 10 UB ALDs and 10 TR ALDs to carry out isolated device testing. To simulate the femoral cortex support of the button, a 4.5 mm–diameter hole was drilled into a stainless steel plate, and the button was passed through the hole then flipped to secure it to the plate, allowing load application perpendicular to the button (Figure 2). The plate was fixed to the lower clamp of the machine with 2 bolts. The loop length of both ALDs was set to 40 mm, in accordance with instructions from the manufacturers. The same loading protocol as that of the specimen test was applied except that no preconditioning was performed since it was not considered necessary because no viscoelastic elements (tendons) were being tested. Data were also obtained in the same manner. Thus, the displacement was set to zero at the beginning of the first load cycle, at an average load of 150 N. The mechanism of failure was also recorded (suture tear or button fracture).

UltraButton in the isolated device testing setup.
Statistical Analysis
For specimen testing, the 3 devices were compared using an analysis of variance and Tukey post hoc test to detect differences between groups. Using 10 specimens per group, similar to what has been used by other researchers, 12,29 allowed us to achieve an effect size of 0.6, which is considered large by Cohen guidelines. 9 For isolated device testing, an independent 2-samples t test was used to detect significant differences in displacement (at different numbers of cycles), ultimate failure load, and stiffness. A post hoc power analysis revealed that with a power of 80%, 10 samples per group would allow detection of a difference of 0.5 mm in displacement at the 1000th cycle and 125 N of ultimate failure load. To verify the validity of the parametric methods, normality was verified using the Shapiro-Wilk test, and equality of variances was verified using the Levene test. All statistical analyses were performed using the SPSS Version 24.0 software (IBM Corp), and a significance level of .05 was used.
Results
Specimen Testing
The mean graft displacements versus the number of cycles for the 3 devices are shown in Figure 3. Displacements for all cycle comparisons (50, 100, 500, and 1000) were similar for the TR and UB but were significantly lower (P < .001) than those measured for the interference screw. Increases in displacement were not proportional to the number of cycles. For example, in the first 100 cycles, average displacements were 30%, 34%, and 33% of the total displacements for the TR, UB, and interference screw, respectively.

Mean cyclic displacement of the graft in the anterior cruciate ligament (ACL) in vitro reconstructions.
Table 1 shows specimen testing data. The ultimate failure loads were similar for the TR and UB, and both were significantly higher than that of the interference screw. The UB had a significantly higher yield load than did the TR and the interference screw, with no significant difference between the latter 2. No significant difference was observed in stiffness among all 3 devices.
Specimen Testing Results a

a Data are reported as mean ± SD unless otherwise indicated. Bold P values indicate statistically significant difference between groups (P < .05). IS, interference screw; TR, TightRope; UB, UltraButton.
Regarding the failure mode, cortical blowouts were observed in 50% of both the UB and the TR (Figure 4) failures, whereas the other 50% of failures were “tendon failure” for the UB and “suture tear” for the TR. The basic failure mode of the interference screw was tendon slippage with very large displacement for the ultimate failure load.
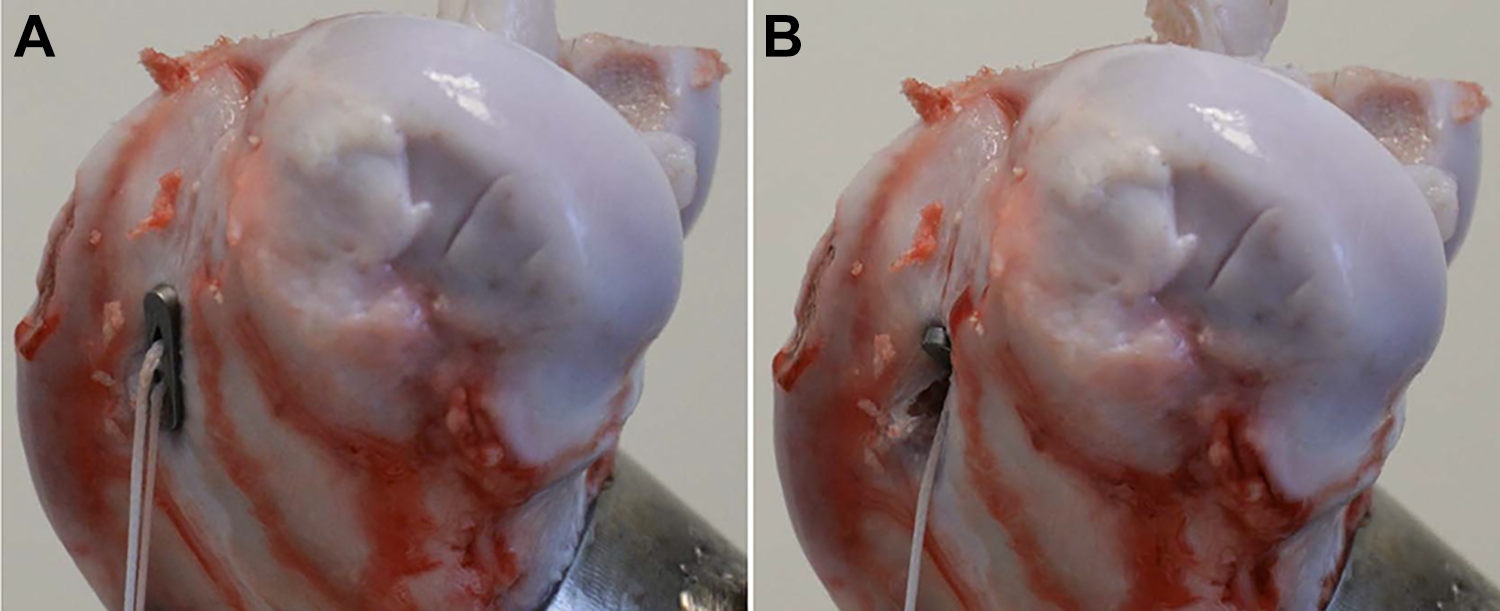
TightRope device (A) before failure and (B) after cortical blowout of the button with subsequent tendon slippage.
Isolated Device Testing
Table 2 shows the results of the comparison between the TR and UB tested in isolation (without biological tissues). The displacements were significantly higher for the UB than for the TR at low cycles (10, 50, and 100), but the differences were not significant at high cycles (500 and 1000). The mean ultimate failure load of UB was statistically higher than that of TR; however, stiffness was equivalent. Regarding the failure mechanism, most of the devices experienced a suture tear, but 3 of 10 UB ALDs fractured at the button.
Isolated Device Testing Results a

a Data are reported as mean ± SD unless otherwise indicated. Bold P values indicate statistically significant difference between groups (P < .05). TR, TightRope; UB, UltraButton.
Discussion
The main finding of this study was that the interference screw showed significantly higher displacements from cyclic loading than those shown by the TR and UB ALDs. Displacements for the interference screw after 500 cycles were >3 mm; however, both ALDs showed displacements <3 mm up to 500 cycles and <4 mm after 1000 cycles. Slippage is a cause of concern because many fixation devices, including the ALDs and the interference screw tested in this study, have shown residual slippage >3 mm in biomechanical tests. 4,12,32,34 This 3-mm limit has been proposed previously as the clinical limit of allowable slippage. 4,29,37 However, postoperative laxity of >5 mm does not appear to lead to a worse clinical outcome at long-term follow-up. 16 In fact, clinical outcomes of ALDs and interference screws have been reported to be satisfactory. 5,6,24,33 Others have found that constructs that use screw fixation had lower ultimate failure loads and higher total elongation compared with those of suspension device constructs, similar to the findings of our study. 39
Stiffness is also an important factor in ACL reconstruction since this surgical procedure attempts to restore the natural properties of an intact ACL. Stiffness is the force applied to the graft divided by its displacement, so stiffness represents the ability of the graft, regardless of its initial length, to withstand loads without excessive deformation. Stiffness was similar for all 3 devices and in the range of previously reported stiffness (242 ± 28 N/mm) of intact ACLs. 40 Yield load expresses the capacity to withstand actual loads better than ultimate failure load in specimen tests because very high displacements at ultimate failure would lead to clinical failure. The yield load was similar for TR and the interference screw, whereas UB displayed a statistically superior yield load to both. This could mean a clinical advantage, but clinical studies to assess this are needed.
Using constructs with porcine bone and bovine tendons, Smith et al 37 observed no statistically significant differences between UB and TR for total or dynamic elongation. In the present study, similar results were found; however, Glasbrenner et al 15 found, using porcine tibias and porcine flexor tendons, that UB showed higher elongation under cyclic loading than that shown by TR. This suggests that studies that control for concurrent variables, such as differing surgeon expertise for devices or bone and tendon inherent variability, should be performed.
The way to assess ACL fixation devices without the influence of biological tissues is using isolated device testing. In our study, these tests showed similar performance for both ALDs except that UB had a higher ultimate failure load than did TR. In previous isolated device tests, similar conclusions were obtained, albeit using a different load protocol. 1 In a recent study comparing UB with 2 other ALDs (RigidLoop Adjustable and ProCinch RT), after 5000 cycles, UB showed the largest elongation at failure, although all 3 demonstrated elongations <3 mm over an extended number of cycles at high loads. 36 However, Cheng et al 8 observed that the mechanical properties of the GraftMax ALD were comparable with those of TR.
Mode of failure is also important in assessing the biomechanical properties of a fixation device. In our study, 9 of 10 failures observed for the interference screw constructs were slippages, similar to the findings of other studies. 3,7,25 In half of the biological specimens of the TR and the UB ALDs, cortical blowout was the mode of failure. For the remaining TR constructs, the failure mode was suture failure, whereas for the remaining UB constructs, the failure mode was tendon failure. From a clinical point of view, this could suggest that UB will withstand traumatic loads at least until bone or tendon failures. Similar modes of failure have been found by other authors; in a meta-analysis of 6 previous studies, 18 TR suture rupture was the mode of failure in 83.8% of cases. Other authors have found tendon slippage to be the main cause of failure with UB ALDs, 37 along with bone breakage, and also suture failures for 75% of TR construct failures. In isolated device testing, sutures failures were observed, except in 3 UB tests, and the ultimate failure load was statistically higher for UB than for TR.
The suitability of ALDs as a graft fixation device in ACL reconstructions is still a matter of debate, 14,22,41 and the present study tries to provide relevant information. It may occur that the corresponding applied load does not simulate the load in an average clinical rehabilitation protocol but instead simulates that of a worst-case scenario. The actual maximum load withstood by the ACL in the early period after a reconstruction is a very difficult value to determine. On one hand, published values of ACL peak load in normal activities vary widely (eg, 156 N to 1000 N for walking), 35 and, on the other, rehabilitation protocols in ACL reconstructions also vary widely; thus, the forces defining the mechanical state of the knee after ACL reconstructions are highly variable. 23 Therefore, doubts remain whether cyclic loading causes excessive lengthening of the loop. While biomechanical tests are very informative in determining the baseline performance of the devices, controlled clinical trials performed using different devices with the same conditions are necessary to be able to draw reliable clinical conclusions.
This study had some limitations. First, the results did not take into account biological healing in a real clinical situation, which is very important in stabilizing the construct. Second, only a 9 mm–diameter metal interference screw was tested. Different interference screw results might have occurred with the use of screws of different materials or size, and also interference screw fixation strength may have been different if a graft with a bone plug had been tested. Third, no retensioning of the ALD was performed because a lack of consensus exists about its application 19,27 ; however, because this was a comparison of 2 ALDs using the same protocol, results can be assumed to be valid without retensioning. Finally, the loading protocol used did not fully unload the ACL reconstruction, maintaining a certain amount of tension in the ALDs. This tension helps to avoid elongation because the ALDs work on a “finger-trap” technology. However, it is not clear whether the initial tension given to the graft at the time of the surgery was enough to always maintain this tension during the rehabilitation period.
Conclusion
Both UB and TR ALD constructs showed significant less displacement in cyclic loading at ultimate failure load than that shown by the interference screw construct. The yield load of the UB construct was significantly higher than that of the other 2. No differences were observed in the ultimate failure load among ALDs when tested as part of bone tendon–device constructs. However, the ultimate failure load was significantly higher for UB than for TR when the devices were tested in isolation.
Footnotes
Acknowledgment
Final revision submitted March 5, 2021; accepted March 24, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: The UltraButton devices used in this work were provided free of charge by Smith & Nephew Spain, and the TightRope devices used in this work were provided free of charge by Arthrex Spain (G.L.G.). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
