Abstract
Background:
The rate of elbow medial ulnar collateral ligament (MUCL) injury and surgery continues to rise steadily. While authors have failed to reach a consensus on the optimal graft or anchor configuration for MUCL reconstruction, the vast majority of the literature is focused on the young, elite athlete population utilizing autograft. These studies may not be as applicable for the “weekend warrior” type of patient or for young kids playing on high school leagues or recreationally without the intent or aspiration to participate at an elite level.
Purpose:
To investigate the clinical outcomes and complication rates of MUCL reconstruction utilizing only allograft sources in nonelite athletes.
Study Design:
Case series; Level of evidence, 4.
Methods:
Patient records were retrospectively analyzed for individuals who underwent allograft MUCL reconstruction at a single institution between 2000 and 2016. A total of 25 patients met inclusion criteria as laborers or nonelite (not collegiate or professional) athletes with a minimum of 2 years of postoperative follow-up. A review of the medical records for the included patients was performed to determine survivorship free of reoperation, complications, and clinical outcomes with use of the Summary Outcome Determination (SOD) and Timmerman-Andrews scores. Statistical analysis included a Wilcoxon rank-sum test to compare continuous variables between groups with an alpha level set at .05 for significance. Subgroup analysis included comparing outcome scores based on the allograft type used.
Results:
Twenty-five patients met all inclusion and exclusion criteria. The mean time to follow-up was 91 months (range, 25-195 months), and the mean age at the time of surgery was 25 years (range, 12-65 years). There were no revision operations for recurrent instability. The mean SOD score was 9 (range, 5-10) at the most recent follow-up, and the Timmerman-Andrews scores averaged 97 (range, 80-100). Three patients underwent subsequent surgical procedures for ulnar neuropathy (n = 2) and contracture (n = 1), and 1 patient underwent surgical intervention for combined ulnar neuropathy and contracture.
Conclusion:
Allograft MUCL reconstruction in nonelite athletes demonstrates comparable functional scores with many previously reported autograft outcomes in elite athletes. These results may be informative for elbow surgeons who wish to avoid autograft morbidity in common laborers and nonelite athletes with MUCL incompetency.
Injury to the medial ulnar collateral ligament (MUCL) at the elbow has been increasingly recognized since Dr. Frank Jobe performed the first UCL reconstruction on Tommy John in 1974 and reported his experience with athletes in 1986. 23 As the UCL serves as the primary static restraint to valgus force of the elbow, injuries can be particularly devastating for high-performance overhead athletes. Over the past decade, there has been an increase in the frequency of MUCL injuries not only in high-level athletes but also in adolescent and youth athletes. 19,38,43 As a response, a significant body of research has been published in the past 2 decades attempting to understand the optimal anatomic configuration of MUCL grafts and anchors during reconstruction. ‡ While the results of MUCL reconstruction are generally reliable in throwing athletes, the vast majority of this literature is still focused on the young, elite athlete population utilizing autograft. 9
To our knowledge, only 1 series has reported on the use of allograft for MUCL reconstruction. 40 The population investigated in that particular study consisted almost exclusively of young patients, with only a few patients older than 25 years of age. Similarly, all patients were competitive collegiate (n = 48) or high school (n = 45) overhead athletes, and the outcomes reported therefore reflect return-to-activity level and may not be generalizable or useful to the average laborer or occasional player who seeks a functional elbow but does not aspire to competitive athletics. In addition, performing autograft harvests can add a degree of complexity and increase the risk for further complications and morbidity. 20 Although rare, superficial infections, continued pain and dysfunction of the graft site, hamstring weakness when the gracilis tendon is harvested, 34 and accidental median nerve harvest when trying to harvest the palmaris tendon have all been described. 27
The acutely focused population characteristics and graft limitations in previous studies present an opportunity to contribute to the MUCL reconstruction literature and guide appropriate treatment for both academic and community elbow surgeons. The purpose of this study was to (1) describe a population of nonelite athletes who underwent MUCL reconstruction using allograft, (2) report complications and reoperation rates, and (3) analyze functional outcome scores at a mean of 8 years of follow-up. We hypothesized that allograft MUCL reconstruction would adequately restore elbow stability for these patients with a low reoperation rate and acceptable functional outcomes.
Methods
After institutional review board approval, a retrospective chart and allograft registry review was conducted to identify all patients undergoing MUCL reconstruction between 2000 and 2016 at a single institution. These dates were selected to provide the potential for a minimum 2-year follow-up, with the aim of obtaining a longer mean follow-up period. The institution joint and allograft registry captures patient and clinical variables at the time of allograft implantation. Clinical follow-up appointments were requested from each patient to provide an accurate evaluation and physical examination. Those unable to complete in-person follow-up were asked to complete a validated, patient-reported outcome survey that also assessed reoperation at an outside institution or any complication, including those that might have not been reflected in our institution’s electronic medical record.
Patient Characteristics and Selection Criteria
A total of 86 patients received primary MUCL reconstruction during the study period. Allograft reconstructions were performed in 44 patients (51%) after discussion and ensuring that the patient understood the requirement for their elbow. Those with limited or lower return-to-sport stakes and demands were offered allograft reconstruction. Inclusion criteria for further cohort analysis were primary procedures with allograft tendon graft utilization and a minimum 2-year follow-up. Exclusion criteria included revision MUCL reconstructions; simultaneous lateral collateral ligament reconstruction; collegiate, professional, or semiprofessional athletes; or other elite-level athletes, defined as competition at an Olympic or Junior Olympic level. All MUCL reconstructions were performed for clinically demonstrated instability with symptomatic continued medial elbow pain or functional impairment with activity. Patients had a positive moving valgus stress test and evidence of valgus instability on examination. Radiographs were obtained as a part of the initial evaluation. Magnetic resonance imaging was obtained to determine the extent of the UCL tears. Despite rehabilitative therapy, medication, and activity modification, these patients were unable to return to work or play at a recreational level of their chosen sport and desired reconstructive efforts. Furthermore, before surgical intervention, all patients failed to respond to exhaustive nonoperative treatment modalities, ranging from over-the-counter pain medication to physical therapy to corticosteroid-based injections.
Twenty-five patients with a mean follow-up of 91 months (range, 25-195 months) met all criteria and were included in the final cohort analysis. There were 17 male and 8 female patients with a mean age of 25 years of age (range, 12-65 years) at the time of surgery. Five (20%) of these patients were laborers with occupations as a mechanic (n = 1), machinist (n = 1), cook (n = 1), and farmer (n = 2). Fewer than half (12/25) of the patients were injured during their involvement with throwing sports, while the remainder were involved in other various athletic and nonathletic activities at the time of injury (Table 1). Twenty-three of 25 patients underwent MUCL reconstruction on the dominant arm. Patient characteristics are summarized in Table 1.
Patient Characteristics for Those Undergoing MUCL Allograft Reconstruction a

a Values are reported as n (%) unless otherwise indicated. BMI, body mass index; MUCL, medial ulnar collateral ligament.
Operative Technique
All MUCL reconstructive efforts were performed utilizing a standard approach through a flexor-pronator mass split. Allografts used during the study period were predominantly plantaris (n = 19), with some split semitendinosus (n = 5) and peroneus longus (n = 1) also being utilized. A variety of techniques were utilized, including modified Jobe (n = 16), docking (n = 7), biotenodesis screw (n = 1), and biointerference screw (n = 1). Concomitant ulnar nerve transposition was not routinely performed, but it was performed at the surgeon’s discretion based on preoperative symptoms and intraoperative nerve assessment and stability. In this cohort, 8 of 25 patients underwent concomitant ulnar nerve transposition. In a similar fashion, the necessity for concomitant osseous intervention was guided by clinical and intraoperative evaluation; 10 of 25 patients had either excision of osseous abnormalities at the ligament insertion/origin or heterotopic ossification/enthesophyte debridement.
Postoperative Rehabilitation
After surgery, patients were typically immobilized for 2 weeks. Afterward, passive range of motion (ROM) was initiated and slowly progressed with the goal of achieving full ROM by 6 to 8 weeks postoperatively. Once full ROM was achieved, gentle strengthening was initiated and slowly progressed over the coming months. For throwing athletes, a return-to-throw program was typically initiated at 4 months postoperatively. Nonthrowing athletes were typically released for full return to all activities around 6 months postoperatively.
Clinical Outcome Assessment
The primary clinical determinants for these patients were the Summary Outcome Determination (SOD) scores 6,41 and the subjective portion of the Timmerman-Andrews score. 1,16 These outcome scores were considered most applicable and relevant to the average nonelite patient. Of note, final scores were acquired after all elbow surgeries, including revisions and reoperations when they occurred. The SOD score (–10 to 10, with positive numbers representing improvement after surgery and negative numbers indicating the patient is worse after treatment) is a relatively straightforward tool that compares surgical outcomes with the preoperative status of the patient (see the Appendix). The Timmerman-Andrews score (0-100) focuses on the primary components that affect functionality for routine activities: pain, swelling, locking/catching, and activity level. 41,42 While the Conway-Jobe rating score was also performed for some patients, it was deemed invalid for the majority of our patients, many of whom were simply aiming to relieve pain and return to a functional level for recreation and work. 13 Secondary outcomes included rates of complication and reoperation for recurrent instability.
Statistical Methods
Descriptive statistics were utilized to report frequencies and percentages for categorical variables. Continuous variables were compared between groups using a Wilcoxon rank sum test. The alpha level was set at .05 for statistical significance.
Results
At the most recent follow-up, none of the 25 elbows included in this study had undergone revision surgery for recurrent instability, nor were any of the index procedures considered a failure. There were a total of 5 complications (20%), 4 of them requiring subsequent procedures. Two patients underwent ulnar nerve transposition for symptomatic ulnar neuropathy, and 1 patient required subsequent release for a contracture with 40° of flexion. One patient sustained a severe contracture with a limited arc of motion from 40° to 80° of flexion, along with ulnar neuropathy symptoms, and subsequently elected to undergo surgical intervention for combined ulnar nerve transposition and arthroscopic osteocapsular arthroplasty. One patient developed a minor painless contracture with 10° of flexion that did not affect activities of daily living; this patient elected against surgical intervention (Table 2). Of note, all complications occurred in patients with plantaris allograft (n = 5); there were no complications in patients who received a split semitendinosus or peroneus longus graft.
Clinical Outcomes for MUCL Allograft Reconstruction a
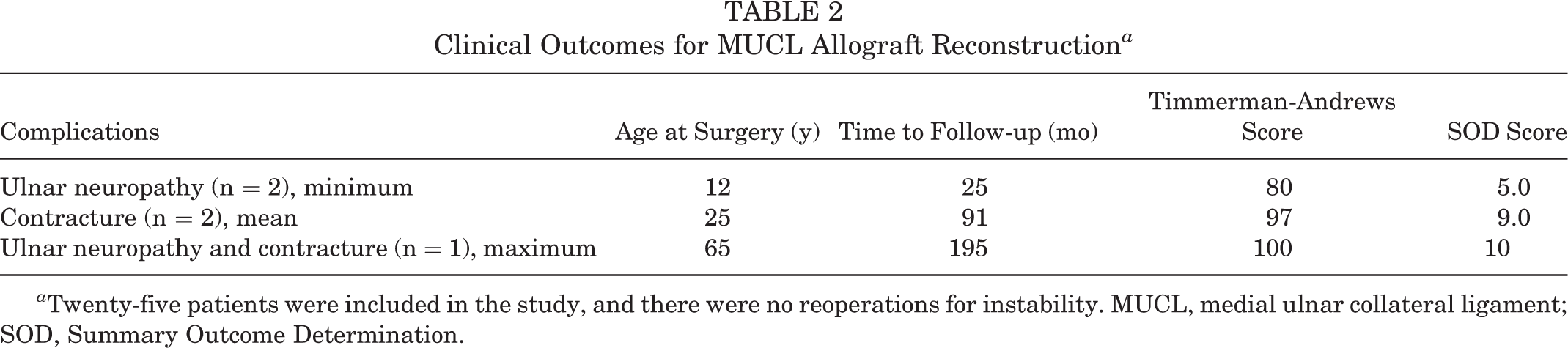
a Twenty-five patients were included in the study, and there were no reoperations for instability. MUCL, medial ulnar collateral ligament; SOD, Summary Outcome Determination.
In terms of clinical outcomes, the mean SOD score was 9 (range, 5-10) at the most recent follow-up. The mean subjective Timmerman-Andrews score was 97 (range, 80-100). All but 1 patient were able to participate in sporting activities at the final follow-up. A subgroup analysis of the outcome scores based on the allograft type was performed. With respect to the mean SOD score, no differences (P = .4) were observed between plantaris (mean score, 9 ± 1), split semitendinosus (mean score, 9 ± 0.4), and peroneus longus (mean score, 8) grafts. Similar findings of no difference were observed with the mean Timmerman-Andrews scores (P = .53) between plantaris (mean score, 96 ± 6), split semitendinosus (mean score, 98 ± 4), and peroneus longus (mean score, 95) grafts.
Discussion
MUCL disruption remains a major concern for patients who seek to return to high-demand activity. While the large majority of MUCL injuries occur in competitive athletes who throw overhead, 40 there is a paucity of clinical data in the literature investigating MUCL reconstruction techniques and graft types in the nonprofessional athlete or laborer after failed nonoperative treatment. This study analyzed clinical outcomes and complication rates of MUCL reconstruction utilizing only allograft sources (plantaris, semitendinosus, and peroneus longus) in 25 laborers and recreational and nonelite athletes. Ultimately, allograft UCL reconstruction demonstrated excellent clinical outcomes at an average of 8 years of follow-up in terms of SOD scores (mean score, 9), subjective Timmerman-Andrews scores (mean score, 97), and a high return-to-activity rate. There were no subsequent revision operations for recurrent instability or failed index MUCL reconstructions. Additionally, clinical outcomes based on the type of allograft failed to reach statistical significance.
Use of allograft for MUCL reconstruction has been described in athletes who throw overhead. In 2013, Savoie et al 40 evaluated the outcomes of hamstring allograft MUCL performed by 3 academic surgeons at a mean of 39 months of follow-up. They reported that 95% of their patients successfully returned to sports, with 88% at the same level of competition or higher, and a majority (85%) did so within 12 months or less. Furthermore, this study reported a favorable complication rate of 6%, notable for ulnar neuropathies, hematoma formation, and a medial humeral epicondyle fracture after return to sports. Obviously, donor site–related morbidity was completely avoided, and no clinical failures of the MUCL reconstruction or revision surgery were reported. The authors concluded that reconstruction of the MUCL ligament with allograft tissue appears to be an effective alternative to autograft reconstruction, with the added benefit of eliminating autograft harvest–related complications.
The current study utilized 3 types of allograft, with a longer-term mean follow-up at 91 months. Similar clinical outcomes were noted, with a high return-to-work and return-to-sport rate and encouraging elbow function in terms of patient-reported outcome scores. Interestingly, a relatively high complication rate of 20% was observed. These complications consisted of 2 ulnar nerve neuropathies, 2 elbow contractures, and 1 combined ulnar nerve neuropathy with a contracture. This rate is consistent with others reported in the literature for MUCL reconstruction (range, 6%-46.3%). 14,19 However, it is higher than that reported in the only other previous allograft UCL paper (Savoie et al). 40 In focusing more specifically on our complication profile, we observed a trend of soft tissue–based problems, which we think can be partially attributed to the older age in this cohort, leading to a likely higher incidence of scar formation. Regardless, it is important to note the lack of donor site morbidity because of allograft use. Additionally, as demonstrated in the Savoie et al paper, 40 our series also demonstrated no revision operations for recurrent instability or failed index MUCL reconstructions.
The reported rate of revision UCL reconstruction varies from 1% to 15%. 8,15,28,30,35,44 Earlier studies that reported revision UCL rates of 1% to 2% included athletes with any overhead component, including sports such as wrestling, soccer, gymnastics, cheerleading, and pole vaulting; 8 this is in sharp contrast to the observed revision rate among baseball pitchers, which is much higher at 5% to 15%. 8,35 In a study of professional baseball pitchers, there was an increasing trend of revision UCL from 2010 to 2016 in both Major League Baseball (MLB) and Minor League Baseball (MiLB) players. 10 In this work, the overall mean revision UCL rate was 6.7%, with a rate of 9.4% in MLB and 5.2% in MiLB players (P = .004).
Data regarding the use of different graft tissues (hamstring, palmaris longus, and plantaris) are mainly observed with autograft sources. Erickson et al, 14 in 2019, demonstrated no differences in clinical outcomes, performance, or return-to-sport rates between hamstring and palmaris longus autograft in professional baseball players. Similar findings have also been observed by Griffith et al, 19 who demonstrated no difference in return-to-sport rates, return to the same level of play, complication profile, or need for revision. Additionally, biomechanical studies evaluating tendon grafts for MUCL reconstruction demonstrated no differences between palmaris longus grafts and larger-diameter semitendinosus grafts with respect to stiffness, load to elongation, or cycles to failure. 36 To our knowledge, the current study is one of the first studies of MUCL reconstruction analyzing multiple allograft sources, and we report no demonstrated differences in outcome score and return to activity with the varying graft types.
Generally, it is understood that MUCL insufficiency requires treatment to restore valgus elbow stability, participate in athletics, and enjoy uninhibited performance of activities of daily living. 5 Nonoperative management is often the initial treatment of choice and continues to be a suitable definitive option in a certain subset of patients with MUCL injuries. Nonoperative candidates are often patients with a history, examination, and imaging consistent with low-grade partial tearing of the UCL; nonelite throwers; or those with goals of care not dependent on placing repetitive high valgus stresses across the elbow. Standard nonoperative programs for UCL injuries include a period of rest, dedicated rehabilitation, and a progressive return to activity. Often, these programs are tailored to the specific demands of the patients, with elite throwers requiring more structured rehabilitation, sport-specific return-to-throwing programs, and possible use of orthobiologics. 11 For the nonelite athletes and laborers, it continues to be unclear whether nonoperative management or surgical treatment leads to better functional outcomes. 5 However, as demonstrated in the current study, performing a trial of nonoperative management followed by allograft MUCL reconstruction may serve as a reasonable treatment pathway resulting in acceptable outcomes for laborers and nonathletes.
There were limitations to the present study primarily related to its retrospective design. Although it spanned 16 years, the infrequency of MUCL reconstruction with allograft use led to a very small sample size and obvious selection bias. As such, our results on the various allograft sources should be interpreted in the context of the paper and our decreased ability to detect a difference. Moreover, as described earlier, our investigation utilized the Timmerman-Andrews outcome measure, which is a nonvalidated scoring system based largely on symptoms. In our lower-demand population of nonelite athletes and laborers, our scoring may likely be lower especially when compared with elite-level athletes. As such, our findings in this subset are focused and may not be as generalizable to patients with extreme demands on their elbow for performance.
The progression of surgical technique, concomitant procedures, and rehabilitation likely influenced the results and may limit the extent of our findings. Specific examples include the use of concomitant procedures addressing possible chronic processes (enthesophytes and heterotopic ossification), which may be a confounding variable to our positive outcomes with respect to pain relief independent of UCL treatment. Additionally, given the limited participation or access to certain records, we were unable to analyze for other comorbidities, confounding variables, and radiographic assessments (stress shielding changes) that could influence our results, and we therefore suspect that the study may be underpowered to detect some differences in certain patient outcomes. As such, larger prospective or matched cohort series are needed to better understand and characterize the unique differences of allograft MUCL reconstruction in the nonelite athlete.
Conclusion
The results of allograft reconstruction make this approach a reasonable alternative to autograft in the nonelite athlete. These results are useful for both the academic and community elbow surgeon who cares for common laborers and nonelite athletes with MUCL tears who desire to avoid the morbidity of autograft harvest.
Footnotes
Final revision submitted April 27, 2020; accepted May 11, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.W.O. has received royalties from Acumet, DonJoy/Aircast, and Wright Medical. J.S.-S. has received research support from Stryker; consulting fees from Exactech, Merck Sharp & Dohme, Tornier, and Wright Medical; speaking fees from Acumed, Arthrex, and Stryker; royalties from Elsevier, Oxford University Press, and Stryker; and hospitality payments from Gemini Medical. C.L.C. has received hospitality payments from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Mayo Clinic (ref No. 18-010653).
