Abstract
Background:
The extravasation of fluid into the intra-abdominal space is recognized as a possible complication of hip arthroscopic surgery/endoscopy. The exposure of anatomic areas to elevated pump pressures and high volumes of irrigation fluid increases the risk of fluid leakage into anatomic spaces around the hip joint, especially to the abdomen and pelvis.
Purpose:
To estimate the incidence and risk factors related to intra-abdominal fluid extravasation (IAFE) after hip endoscopy or arthroscopic surgery.
Study Design:
Cohort study; Level of evidence, 2.
Methods:
A prospective study was carried out between June 2017 and June 2018. A total of 106 hip procedures (endoscopy or arthroscopic surgery) performed for extra- or intra-articular abnormalities were included. Before and after surgery, in the operating room, ultrasound was performed by a trained anesthesiologist to detect IAFE. The hepatorenal (Morison pouch), splenorenal, retroaortic, suprapubic (longitudinal and transverse), and pleural spaces were examined. Patients were monitored for 3 hours after surgery to assess for abdominal pain. The data collected included maximum pump pressure, duration and volume of irrigation fluid (Ringer lactate), total surgical time, and traction time.
Results:
The incidence of IAFE was 31.1% (33/106; 95% CI, 23.1%-40.5%). The frequency of IAFE was 52.9% (9/17) in cases with isolated extra-articular abnormalities and 15.9% (7/44) in cases with isolated femoroacetabular impingement; in cases with both extra- and intra-articular abnormalities, the frequency was 37.8% (17/45). An intervention in the subgluteal space was identified as a risk factor for IAFE (odds ratio, 3.62 [95% CI, 1.47-8.85]). There was no statistically significant difference between groups (with vs without IAFE) regarding total surgical time, maximum pump pressure, or fluid volume. Postoperative abdominal pain was found in 36.4% (n = 12) of cases with IAFE compared with 2.7% (n = 2) of cases without extravasation (P < .001). No patient with IAFE developed abdominal compartment syndrome.
Conclusion:
IAFE was a frequent finding after hip arthroscopic surgery/endoscopy in patients with extra-articular abnormalities. Exploration of the subgluteal space may increase the risk of IAFE. Pain and abdominal distension during the immediate postoperative period were early warning signs for IAFE. These results reinforce the need for careful intraoperative and postoperative monitoring by the surgeon and anesthesiologist to identify and avoid complications related to IAFE.
Keywords
Hip arthroscopic surgery and endoscopy are rapidly evolving surgical procedures indicated for the management of many extra- and intra-articular abnormalities, respectively. Arthroscopic surgery has shown good functional results, with an estimated rate of minor and major complications of 4.2% and 1.7%, respectively. 17 The most common complications are traction-related injuries, chondrolabral damage, deep vein thrombosis, pulmonary embolism, and fluid extravasation. 2,12,17
The exposure of anatomic areas to elevated pump pressures and high volumes of irrigation fluid increases the risk of fluid leakage into anatomic spaces around the hip joint, especially to the abdomen and pelvis. Although the main risk factors for intra-abdominal fluid extravasation (IAFE) during hip arthroscopic surgery or endoscopy are not fully identified, some studies have found an association between IAFE and iliopsoas muscle tenotomy, 6 extensive capsulotomy, 11 prolonged surgical times, 5 and elevated pump pressures. 10
IAFE can elevate intra-abdominal pressure and increase the risk of abdominal compartment syndrome (ACS), which may lead to the development of metabolic acidosis, hypothermia, and cardiac arrest. 1 The frequency of IAFE in patients undergoing hip surgery has been studied mainly in those with a diagnosis of intra-articular abnormalities and has been estimated at rates between 0.16% and 16%. 7 –9 However, the incidence and risk factors have not been specifically evaluated. The aim of this study was to estimate the incidence and risk factors of IAFE after hip endoscopy or arthroscopic surgery. Our hypothesis was that the incidence of IAFE would be more frequent than that described in the literature. Additionally, we believed that there would be specific procedures associated with a higher risk of IAFE.
Methods
We performed a prospective observational study of consecutive patients undergoing hip endoscopy and/or arthroscopic surgery between June 2017 and June 2018. This study was approved by an institutional review board, and written informed consent was obtained from all participants. We conducted the study at a hip preservation surgery referral center with experience in the management of extra-articular abnormalities, mainly in treatment of the subgluteal space.
Participants
Included in the study were patients of both sexes older than 18 years and with a diagnosis of intra- and/or extra-articular abnormalities treated with primary hip arthroscopic surgery and/or endoscopy in the study period. Patients with intra-abdominal fluid during the preoperative examination were excluded. A total of 106 participants were included; 66.0% were female, and the mean age was 47.3 ± 14.3 years.
Procedures
All hip surgery was performed by a single senior surgeon (B.A.-B.) with the patient under regional and general anesthesia. Each patient was placed in the supine position on an orthopaedic traction table using a perineal post. The Trendelenburg position was not used in this study. The anterior hip space was intervened through the anterior, anterolateral, and anterior distal accessory portals. Access to the peritrochanteric space was achieved through the accessory upper and distal portals. The subgluteal space was accessed through the posterolateral portal and posterior proximal accessory portal.
The surgical procedure was performed with a POWER LED 175 arthroscope with 30° and 70° lenses (Karl Storz), a Flow Control irrigation system (Stryker), and a hand vaporization system (VAPR; DePuy) irrigating Ringer lactate with 500 µg of adrenaline for every 3 L of infused solution. T-capsulotomy of the iliofemoral ligament was performed, and traction was used when the central hip space was intervened. No traction was used during the intervention of the subgluteal and peritrochanteric spaces. Intravenous diclofenac 75 mg and dipyrone 1 g were given as analgesics at the beginning of surgery. Intravenous oxycodone 3 mg was administered at the end of the procedure.
Ultrasound Assessment of IAFE
The presence of IAFE was evaluated using an ultrasound device (Sonosite M-Turbo; Fujifilm) with a low frequency convex probe (1-6 MHz). Ultrasound was performed with the patient awake in the supine position at the beginning as well as at the end of the procedure in the operating room. Head and feet were placed at the same level during the ultrasound. All ultrasound examinations were performed by a trained anesthesiologist (M.P.-V.) who completed a formal training course in the Focused Assessment with Sonography for Trauma (FAST) scan method.
During the examination, the hepatorenal (Morison pouch), splenorenal, retroaortic, and suprapubic (longitudinal and transverse) spaces were explored. The left and right pleural spaces were also examined. The hepatorenal and splenorenal spaces were assessed by placing the probe in the abdominal right and left upper quadrants, respectively (Figure 1, A and B). In the retroaortic space, the epigastrium was examined by placing the probe where the aortic artery before its bifurcation and the corresponding vertebral body were best visualized (Figure 1C). The suprapubic space was assessed by placing the probe in the pelvis in a longitudinal and transverse orientation (Figure 1D), and the pleural space was evaluated in the bases of both lungs (Figure 1E). The diagnosis of IAFE was made by the presence of extravasated fluid in any of the spaces; the amount of fluid was not quantified (Figure 2).

Probe placement for the identification of cases with fluid extravasation: (A) hepatorenal (Morison pouch) space, (B) splenorenal space, (C) retroaortic space, (D) suprapubic space, and (E) pleural space. Reprinted with permission from Aguilera-Bohórquez B, Cantor E, Ramos-Cardozo O, Pachón-Vásquez M. Intraoperative monitoring and intra-abdominal fluid extravasation during hip arthroscopy. Arthroscopy. 2020;36(1):139-147. Copyright 2020 by Elsevier.

Presence of fluid in the spaces evaluated: (A) hepatorenal (Morison pouch) space, (B) splenorenal space, (C) retroaortic space, (D) transverse suprapubic space, and (E) pleural space. IAFE, intra-abdominal fluid extravasation.
Data Collection
Hip abnormalities, the American Society of Anesthesiologists (ASA) class, procedures, and intervened hip spaces were recorded. The procedures included in the extra-articular anterior spaces were psoas tenotomy and anterior inferior iliac spine resection. In the peritrochanteric space, tensor fascia lata tenotomy and abductor insertion were included. Sciatic neurolysis and lesser trochanteric osteotomy were classified in the subgluteal space. Procedures associated with the management of femoroacetabular impingement (FAI) were classified in the anterior space. The data collected included the maximum pump pressure, duration and volume of irrigation fluid (Ringer lactate), total surgical time, and traction time. The patients were observed during the first 3 hours after surgery to assess abdominal pain (measured using the visual analog scale; VAS) and abdominal distension.
Statistical Analysis
The sample size was calculated to estimate the incidence of IAFE using the formula for cross-sectional studies. There were 101 hip arthroscopic procedures required, assuming an expected prevalence of 21%, a 95% CI, and a precision of ±8%. In this study, we did not calculate a priori power to detect differences between groups.
The estimation of the incidence of IAFE was made using the Wilson approximation with its respective 95% CI. Continuous variables were summarized as mean ± SD or median and interquartile range (IQR).
The patients were divided into 2 groups according to the presence of IAFE. A comparison of the groups for continuous variables was performed using the Student t test (for independent samples) or Mann-Whitney U test. Binary and categorical characteristics were compared using the chi-square test or Fisher exact test. Traction time was classified into groups: 0 minutes (without traction), 1-30 minutes, 31-60 minutes, and >60 minutes.
A logistic regression model was applied to determine factors associated with IAFE. The odds ratio (OR) was estimated with the 95% CI, and goodness of fit was evaluated using the Hosmer-Lemeshow test. Model selection was performed using a backward selection methodology based on the Akaike information criterion. A level of significance of .05 was used. All analyses were carried out using Stata 13 (StataCorp).
Results
Fluid extravasation was found in 33 of the 106 patients (31.1%; 95% CI, 23.1%-40.5%). Among the patients with IAFE, 28 (84.8%) had fluid in the suprapubic space, 18 (54.5%) in the hepatorenal space, 13 (39.4%) in the splenorenal space, 6 (18.2%) in the retroaortic space, and 4 (12.1%) in the pleural space. The incidence of IAFE was 52.9% (9/17) in cases with extra-articular abnormalities, 15.9% (7/44) in cases with FAI (intra-articular abnormality), and 37.8% (17/45) in cases with both extra- and intra-articular abnormalities (Figure 3). No patient with IAFE developed ACS.
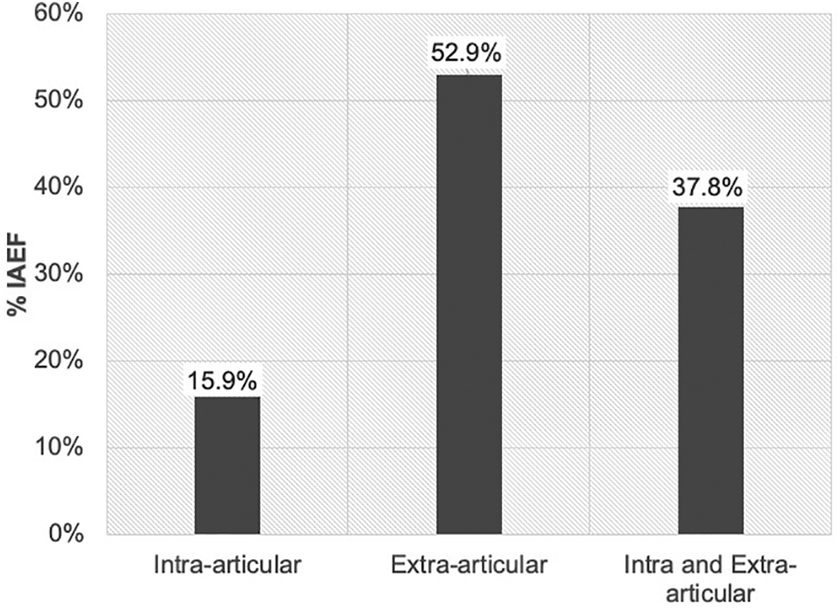
Incidence of intra-abdominal fluid extravasation (IAFE) according to diagnosis. Extra-articular category included cases with deep gluteal syndrome, subspine impingement, ischiofemoral impingement, or external snapping hip. Hips with femoroacetabular impingement were classified in the intra-articular category.
The demographic and clinical characteristics of the patients according to the study groups are described in Table 1. No statistically significant differences were found in age, body mass index, or ASA class between patients with and without IAFE (Table 1).
Demographic and Clinical Characteristics a

a Data are shown as n (%) unless otherwise indicated. AIIS, anterior inferior iliac spine; ASA, American Society of Anesthesiologists; BMI, body mass index; DGS, deep gluteal syndrome; FAI, femoroacetabular impingement; IAFE, intra-abdominal fluid extravasation; IFI, ischiofemoral impingement; IQR, interquartile range; SSI, subspine impingement.
b Statistically significant difference between groups (P < .05).
c Only 70 out of 73 patients have this information available.
d Only 103 out of 106 patients have this information available.
In the majority of cases with IAFE, the subgluteal space was intervened (72.7%), with statistically significant differences between the groups (P = .004). The anterior peripheral space was more frequently intervened in the patients without IAFE (P = .023). Tenotomy of the tensor fascia lata, sciatic nerve decompression, and lesser trochanteric osteoplasty were the procedures more frequent in the IAFE group. No statistically significant differences were found in total surgical time, maximum pump pressure, or fluid volume between the groups (Table 1).
A decrease in the incidence of IAFE was observed with an increase in traction time (Table 2). Those with a traction time of 0 minutes had a higher probability of IAFE than those with a time between 31 and 60 minutes (52.6% vs 25.6%, respectively; OR, 0.31 [95% CI, 0.09-0.96]). After adjusting for potential confounders, patients who required management in the subgluteal space were 3.6 times more likely to have IAFE (OR, 3.62 [95% CI, 1.47-8.85]). Based on the multivariable model, subgluteal space exploration was identified as an independent risk factor for the presence of IAFE (Table 2).
Risk Factors Associated With the Presence of IAFE a

a IAFE, intra-abdominal fluid extravasation; OR, odds ratio.
bP < .10.
cP < .05.
Postoperative abdominal pain was found in 36.4% (n = 12) of the cases with IAFE compared with 2.7% (n = 2) of the cases without extravasation (P < .001). The median VAS score in the IAFE group during the first 3 postoperative hours was 8.0 (IQR, 6.2-8.0). Abdominal distention was found in 39.4% (n = 13) of the cases with extravasation compared with 8.2% (n = 6) of the cases without IAFE (P < .001). There were 5 patients with IAFE who had both abdominal pain and abdominal distension.
Within the IAFE group, 5 patients were admitted to the intensive care unit for close observation, with a length of stay of 2 days. All of these patients were admitted because of severe pain, abdominal distension, and shortness of breath. Extravasated fluid was found in the pleural space in 3 cases. None required surgical management for IAFE. Furosemide was administered in 2 cases, and 3 cases did not receive any treatment other than observation. All patients were discharged without symptoms and had a full recovery.
Discussion
In this study, we found an incidence of IAFE of 31.1% (n = 33) identified through ultrasound. In the literature, the estimate varies from 0.16% to 16%. 7,10 This difference could be explained by the high percentage of cases with deep gluteal syndrome or ischiofemoral impingement (IFI) in our study. Surgical treatment in the subgluteal space has been identified as a risk factor for IAFE. The incidence of IAFE in cases with isolated FAI was 15.9%. This result may be more representative of findings by other studies because most of the patients who are treated in other centers require hip arthroscopic surgery for the management of intra-articular abnormalities.
Kocher et al, 10 based on a survey of experts from the Multicenter Arthroscopy of the Hip Outcomes Research Network (MAHORN) group, estimated an incidence of IAFE of 0.16% (40 cases) in a sample of 25,648 hip arthroscopic procedures. However, the MAHORN study only included symptomatic cases that required medical treatment. In a similar study by Haskins et al 7 of 100 hip arthroscopic procedures, the incidence of asymptomatic IAFE was 16% through ultrasound, which is similar to our estimate in patients with only FAI. On the other hand, Hinzpeter et al 9 reported a higher incidence of IAFE (47.5%) through axial computed tomography (CT). CT is a more sensitive diagnostic method to detect small amounts of fluid compared with ultrasound. Nonetheless, ultrasound has advantages such as low cost, immediate availability, and portability. Additionally, it avoids the risk of high ionizing radiation exposure to the patient, which is associated with CT.
In particular, the incidence of IAFE was less frequent in hips with both intra- and extra-articular abnormalities than hips with isolated extra-articular abnormalities. This finding could be explained by the use of traction during the surgical treatment of intra-articular disease, which has been suggested as a possible protective factor to IAFE. In 36 patients who underwent arthroscopic surgery for FAI, Hinzpeter et al 9 described a positive relationship between extravasated volume and a longer period without traction (r = 0.29; P = .070). Similarly, our data showed a decrease in the incidence of IAFE with increasing traction time, although these findings were not statistically significant in the multivariate model.
In this study, the main risk factor identified for the presence of IAFE was exploration of the subgluteal space, which included the surgical management of patients with deep gluteal syndrome or IFI. According to Tan et al, 16 neurolysis of the sciatic nerve and tenotomy of the piriformis can open an access route through the greater sciatic notch, which could allow the leakage of fluid into the abdominal cavity. However, no reports of cases with symptomatic IAFE associated with sciatic nerve decompression were found. 4,7,10 Regarding the patients with IFI, we hypothesize that during lesser trochanteric osteoplasty, an access route through the lesser sciatic notch is created, which can allow fluid migration toward the abdomen. It is important to underline that all patients with IFI had clinical signs and symptoms and were diagnosed by a 3-dimensional dynamic study.
In our series, psoas tenotomy was performed in 7 cases, and none of the patients presented with fluid extravasation. In the literature, psoas tenotomy has been associated with a higher risk of IAFE, 4,5,10 especially when it is performed at the beginning of the surgical procedure. In the MAHORN study, 63% of the 40 cases with symptomatic IAFE had undergone psoas tenotomy. On the other hand, Hanypsiak et al, 6 in a study performed with 6 hemipelvis human cadaveric specimens to identify possible access routes through which fluid enters the abdominal cavity and retroperitoneal space, found a greater leakage of fluid in the specimens that underwent psoas tenotomy compared with the specimens that underwent capsulotomy only. It is noteworthy that with our clinical practice, we routinely perform psoas tenotomy at the end of surgery after releasing the limb and removing the perineal post with pressures below 20 mm Hg, which could decrease the risk of extravasation and explain our findings.
The use of higher pump pressures during hip arthroscopic surgery has been identified as a risk factor for IAFE. 5,8,10 Although this was not evidenced in our study, this finding is similar to that reported by Haskins et al. 7 Several studies have recommended the close monitoring of fluid balance and the use of pump pressures between 40 and 50 mm Hg to reduce the risk of extravasation. 12,13
Although all patients were placed in the supine position without elevation maneuvers, which may increase the risk of fluid leakage into the abdominal cavity, the frequency of IAFE was higher than that reported in the literature. Hip arthroscopic surgery/endoscopy can be performed in the supine, lateral, prone, or Trendelenburg position. However, patient positioning on the surgical table depends on the surgeon’s preference. 14 In a systematic review conducted by Ekhtiari et al, 4 symptomatic IAFE was reported in patients who underwent surgery in the supine and lateral decubitus positions; however, no evidence was found that the patient’s position is a potentially important factor for IAFE. In contrast, Stafford et al 15 recommended the lateral position under the hypothesis that its use may reduce the need for increased fluid pressure during surgery within the peripheral space.
In this study, we found that approximately 2 of 3 patients who developed IAFE (20/33) had symptoms of pain or abdominal distension during the first 3 hours after surgery. Among the patients with pain and abdominal distension, leakage of fluid was found in 85.7% (12/14) and 68.4% (13/19), respectively. Pain around the hip and lower limbs after hip arthroscopic surgery/endoscopy is a common postoperative symptom, mainly during the first 2 days after surgery. 3 However, abdominal pain detected in the recovery room should be considered a warning sign to perform an abdominal examination. Ultrasound should be considered for the early detection of IAFE and for establishing appropriate management to decrease complications associated with this condition. Abdominal distention has been identified as one of the main signs associated with the development of ACS after hip arthroscopic surgery/endoscopy. 4 The review by Ekhtiari et al 4 found abdominal distension in 8 of 9 patients with ACS, which suggests that this is an early warning sign for the identification of ACS. It is important for the surgical team to be aware of the presence of IAFE as a first step to prevent complications.
Among the cases with IAFE, 39.4% (13/33) had no symptoms/signs associated with the extravasation of fluid. Hinzpeter et al 9 found, in 19 (47.5%) hips with leakage of fluid into the iliopsoas and/or retroperitoneal-intraperitoneal space, an extravasated volume of 3.63 ± 1.43 L measured by CT. However, they did not report symptoms or complications attributable to extravasated fluid. 9
In patients undergoing hip arthroscopic surgery/endoscopy, we recommend performing an abdominal examination after surgery to detect early cases with IAFE. If the patient has severe abdominal pain, ultrasound may be indicated to detect extravasated fluid in the abdomen. If IAFE occurs, the patient should be kept under close observation to monitor hemodynamic parameters.
Limitations
This study has several limitations. First, all hip arthroscopic/endoscopic procedures were performed by 1 senior surgeon, and variables such as traction time and pump pressure can differ from other surgeons, which could modify the incidence of IAFE. In addition, the heterogeneity of procedures included in this study could be recognized as a confounding factor, which does not allow for the generalization and estimation of the frequency of IAFE with accuracy. Second, ultrasound was not conducted independently by a second observer; therefore, interobserver reliability was not calculated. Additionally, the patients’ symptoms were not correlated with the extravasated volume; this should be addressed in future studies.
Conclusion
IAFE was a frequent finding after hip arthroscopic surgery/endoscopy in patients with extra-articular abnormalities. Subgluteal space exploration may increase the risk of IAFE. Pain and abdominal distension during the immediate postoperative period were early warning signs for IAFE. These results reinforce the need for careful intraoperative and postoperative monitoring by the surgeon and anesthesiologist to identify and avoid complications related to IAFE.
Footnotes
Acknowledgment
The authors appreciate the Research Institute of Centro Médico Imbanaco for its support during the development of this project. They also thank Evelyn Bedoya for assistance with statistical analysis and Dr Juan Gómez Hoyos for comments and suggestions.
Final revision submitted February 27, 2020; accepted March 12, 2020.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Centro Médico Imbanaco Institutional Review Board (code CEI-281).
