Abstract
Background:
A concomitant rotator cuff tear (RCT) with frozen shoulder is a common but challenging clinical scenario. The effect of frozen shoulder on clinical outcomes is open to discussion.
Purpose/Hypothesis:
This study aimed to evaluate the effect of preoperative frozen shoulder on postoperative clinical outcomes of an RCT. We hypothesized that the treatment results of an RCT concomitant with preoperative frozen shoulder would be comparable with those of an isolated RCT.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A total of 212 patients were divided into 2 groups: 154 in the non–frozen shoulder (NFS) group and 58 in the preoperative frozen shoulder (FS) group. All patients underwent a thorough preoperative evaluation that included range of motion (ROM) and the pain visual analog scale, functional visual analog scale, Constant score, and American Shoulder and Elbow Surgeons (ASES) score. The same evaluation was performed at 6 months and 1 and 2 years postoperatively. The healing status of the repaired RCT of all patients was evaluated by postoperative magnetic resonance imaging.
Results:
The FS group showed statistically significantly worse functional outcomes than the NFS group at 6 months and 1 year postoperatively (P < .05). At 2 years postoperatively, active assisted ROM was equivalent between the groups, although the ASES and Constant scores were significantly lower for the FS group (P < .033 and P < .001, respectively). The retear rates were 5.3% and 12.3% for the FS and NFS groups, respectively (P = .013).
Conclusion:
Preoperative frozen shoulder positively affected rotator cuff healing but negatively affected most functional outcomes, including ROM, at 6 months and 1 year postoperatively. At 2 years after surgery, there was no significant difference in active motion, but outcome scores remained lower in the FS group. For patients with preoperative frozen shoulder, a delay in surgery for additional physical therapy might not be necessary. The retear rate for the NFS group was more than double that for the FS group in the current study, which indicates that surgery for an RCT combined with frozen shoulder might provide better results in the long term because of an intact, healed rotator cuff.
Rotator cuff tears (RCTs) are a common cause of shoulder pain and dysfunction, as is frozen shoulder. 2,28 Unfortunately, some patients have both abnormalities. While the operative management of RCTs is widely accepted as a reasonable treatment modality, the management of combined preoperative frozen shoulder and RCTs is still under debate. It is unclear whether to wait for frozen shoulder to improve before surgery or to surgically treat frozen shoulder and RCTs early.
The overall incidence of postoperative stiffness after rotator cuff repair reportedly ranges from 4.9% to 23%, while that of primary frozen shoulder is 2% to 5% in the general population. 9 Frozen shoulder can be broadly categorized into 2 subgroups depending on the cause: primary frozen shoulder is idiopathic in origin, whereas secondary frozen shoulder can be explained by fractures, operative procedures, cervical herniated discs, and other shoulder lesions. 7,22 In cases of a concomitant RCT, it has been reported that preoperative frozen shoulder should be managed separately before RCT repair because the former negatively affects postoperative outcomes. 19 However, a 2008 study showed that frozen shoulder can be managed with a single-stage operative procedure without any changes in clinical outcomes if capsular release and manipulation are added to the index procedure. 20 In addition, some studies have reported that preoperative frozen shoulder positively affects the postoperative outcomes of RCT repair. 18 Thus, how preoperative frozen shoulder affects RCT repair outcomes remains to be elucidated.
This study aimed to evaluate the effect of preoperative frozen shoulder on postoperative clinical outcomes of RCT repair. We hypothesized that the treatment results for RCT concomitant with preoperative frozen shoulder would be comparable with those for isolated RCT.
Methods
Patient Selection
This study was approved by an institutional review board. Between April 2011 and March 2013, a total of 696 patients were diagnosed with an RCT and underwent arthroscopic RCT repair in our hospital by a single senior surgeon [J.C.Y.]. Patients were included if they had undergone RCT repair at our hospital. Patients were excluded for concomitant instability or other diagnoses such as calcific tendinitis, superior labral anterior-posterior tears, paralabral cysts, infections, or glenohumeral joint osteoarthritis. Additionally, patients who underwent previous shoulder surgery with or without rotator cuff repair, had a partial-thickness RCT, underwent incomplete and partial RCT repair because of the poor condition of rotator cuff tissue, 27 completed a short follow-up period (<6 months), or did not undergo postoperative magnetic resonance imaging (MRI) for the evaluation of tendon integrity were excluded. The patients who met the study criteria were divided into 2 groups: those with frozen shoulder (FS group) and those without (non–frozen shoulder [NFS] group). According to a previously validated protocol, 24 the definition of frozen shoulder was as follows: (1) active assisted shoulder range of motion (ROM) less than 120° for forward flexion, less than 30° for external rotation at the side, and less than the L3 level for internal rotation at the back 1,6,8,17 ; and (2) limited active ROM visually identified on the affected shoulder versus that on the nonaffected shoulder, even if ROM was better than the ROM criteria listed above.
Among the 230 patients who met the study criteria, 18 were excluded because they were lost to follow-up. Of the remaining 212 patients, 154 were assigned to the NFS group and 58 to the FS group. We divided the patients into 3 subgroups according to tear diameter using the classification system described by Post et al 23 : small, <1 cm; medium, 1-3 cm; and large to massive, >3 cm.
Surgical Procedures
Manipulation was performed in the supine position under general anesthesia. The glenohumeral joint was forced to full ROM in forward flexion. All operative procedures were performed with the patient in the lateral decubitus position under general anesthesia. Arthroscopic findings (Figure 1) of hyperemic inflammatory capsules were observed in all patients in the FS group.

Right shoulder, viewed through the anterior portal, from the rotator interval to the anteroinferior direction with the patient in the lateral decubitus position. Arthroscopic finding of a hyperemic, inflamed posterior capsule.
After a thorough examination of the glenohumeral joint space, the patients in the FS group underwent rotator interval release and capsular release from 1 o’clock to 9 o’clock in an anterior direction using a radiofrequency device or arthroscopic scissors (Figure 2).
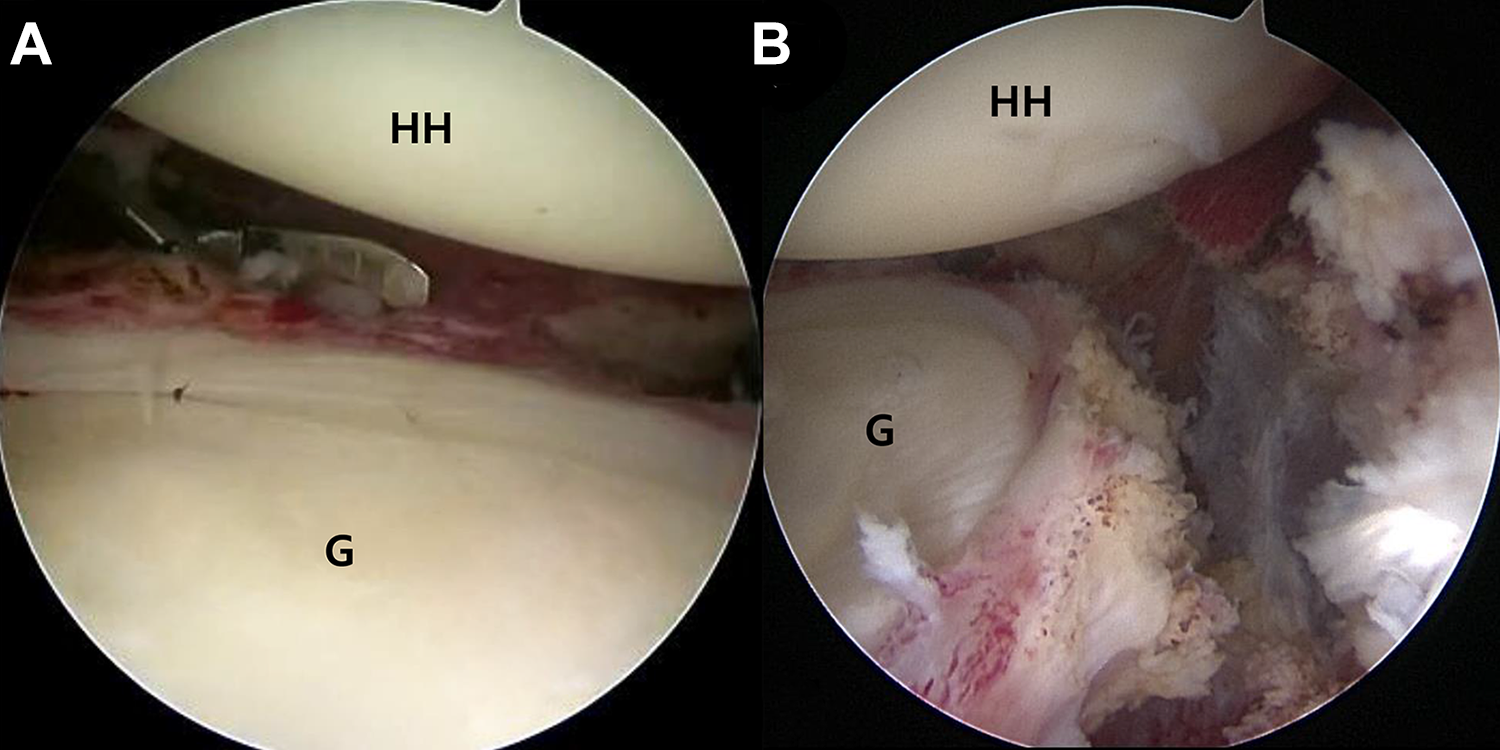
Left shoulder, viewed through the anterior portal, with the patient in the lateral decubitus position. (A) Using arthroscopic scissors, posteroinferior release was performed inferiorly, extending to the 6-o’clock position. (B) Arthroscopic photograph after pancapsular release. G, glenoid; HH, humeral head.
Thereafter, the scope was moved to the subacromial space. If there were signs of impingement (eg, joint space narrowing, acromion bony spur protrusion, and a hook- or curve-shaped acromion) on simple radiographs, subacromial decompression and acromioplasty were performed. After subacromial decompression, the RCT was observed and treated depending on its mobility, while proper perimuscular soft tissue release and repair were performed using bioabsorbable anchors with the double-row technique (ie, transosseous-equivalent suture bridge). Finally, the stability of the repair construct and the overall repair configuration were confirmed via a probe examination.
Postoperative Rehabilitation
All patients underwent immobilization using a 30° abduction pillow. In general, 4 weeks of immobilization was recommended for patients with small- or medium-sized tears and 6 weeks for those with large or massive tears. The same postoperative protocol was used in both groups. After the immobilization period, passive ROM exercises were started after 4 weeks. Active assisted ROM exercises were started once full passive ROM was recovered. Strengthening exercises were started between 10 and 12 weeks postoperatively and continued for approximately 3 to 6 months.
Outcome Evaluation
All patients underwent a thorough preoperative evaluation that included active assisted shoulder ROM (ie, forward flexion, external rotation at the side, internal rotation at the back, and abduction) as well as the pain visual analog scale (PVAS), functional visual analog scale (FVAS), Constant score, Simple Shoulder Test (SST), and American Shoulder and Elbow Surgeons (ASES) score. The same evaluation was performed at 6 months, 1 year, and 2 years postoperatively. A single independent specialist in shoulder physical therapy [S.M.L.] administered all clinical evaluations on an outpatient basis using a goniometer and scoring sheet. The examiner was blinded to the patients’ preoperative status.
Radiological Assessment
Postoperative MRI (3.0-T Gyroscan Intera Achieva scanner; Philips) was performed in all 212 patients at a mean of 5.62 ± 1.08 months. 14 Oblique coronal, oblique sagittal, and transverse planes on T2-weighted MRI were used to classify postoperative rotator cuff integrity into 5 categories as described by Sugaya et al. 25 We defined type IV and V tears as retears. There were 2 orthopaedic shoulder specialist fellows who evaluated all images independently without clinical information, and then each repeated the MRI evaluation 2 weeks later.
Analysis of RCT Size by Group
Several studies have shown that tear size is the greatest predictor for retears. 3,5,10,15,16 The initial tear size significantly affects clinical outcomes after arthroscopic RCT repair. Therefore, we analyzed the clinical outcomes by RCT size of the 2 groups.
Statistical Analysis
All data were analyzed using SPSS software (version 12.0; IBM) with 95% CIs. We used the chi-square test to examine sex, presence of diabetes mellitus, presence of thyroid disease, and tear type. For other variables, such as age, follow-up duration, clinical scores (ie, PVAS, FVAS, SST, Constant, and ASES), and ROM, an independent t test was used for the variables that were normally distributed and the Wilcoxon signed-rank test for the variables that were not normally distributed. P values <.05 were considered statistically significant. The intraclass correlation coefficient was used to assess the interobserver and intraobserver reliabilities for the detection of postoperative rotator cuff integrity on MRI. We considered an intraclass correlation coefficient of ≥0.7 as good or excellent reliability.
Results
There were no significant differences in baseline demographics between the FS and NFS groups except for the presence of thyroid disease as a comorbid preoperative condition; the FS group had a higher incidence of thyroid disease than the NFS group. The patients’ demographic data are summarized in Table 1.
Baseline Patient Demographics a

a Data are presented as mean ± SD unless otherwise indicated. Bolded P value indicates significant differences between groups (P < .05). FS, frozen shoulder; NFS, non–frozen shoulder; RCT, rotator cuff tear.
The NFS group showed significantly better preoperative clinical scores than the FS group except for the PVAS score (P = .407) (Table 2).
Preoperative Clinical and Functional Outcomes a

a Data are presented as mean ± SD (range). Bolded P values indicate significant differences between groups (P < .05). ASES, American Shoulder and Elbow Surgeons; FS, frozen shoulder; FVAS, functional visual analog scale; NFS, non–frozen shoulder; PVAS, pain visual analog scale; ROM, range of motion.
b Internal rotation measured to the highest vertebral level that can be reached with the patient’s thumb.
The postoperative clinical outcomes at 6 months and 1 year were also similar between groups, except for the PVAS, FVAS, and ASES scores (6 months: P = .401, .899, and .138, respectively; 1 year: P = .761, .094, and .150, respectively). Overall, the NFS group showed better clinical outcomes than the FS group (Tables 3 and 4). There were no significant differences in the 2-year postoperative clinical outcomes between the groups, with the exception of the ASES and Constant scores (P = .033 and P < .001, respectively) (Table 5).
6-Month Postoperative Clinical and Functional Outcomes a

a Data are presented as mean ± SD (range). Bolded P values indicate significant differences between groups (P < .05). ASES, American Shoulder and Elbow Surgeons; FS, frozen shoulder; FVAS, functional visual analog scale; NFS, non–frozen shoulder; PVAS, pain visual analog scale; ROM, range of motion.
b Internal rotation measured to the highest vertebral level that can be reached with the patient’s thumb.
1-Year Postoperative Clinical and Functional Outcomes a

a Data are presented as mean ± SD (range). Bolded P values indicate significant differences between groups (P < .05). ASES, American Shoulder and Elbow Surgeons; FS, frozen shoulder; FVAS, functional visual analog scale; NFS, non–frozen shoulder; PVAS, pain visual analog scale; ROM, range of motion.
b Internal rotation measured to the highest vertebral level that can be reached with the patient’s thumb.
2-Year Postoperative Clinical and Functional Outcomes a
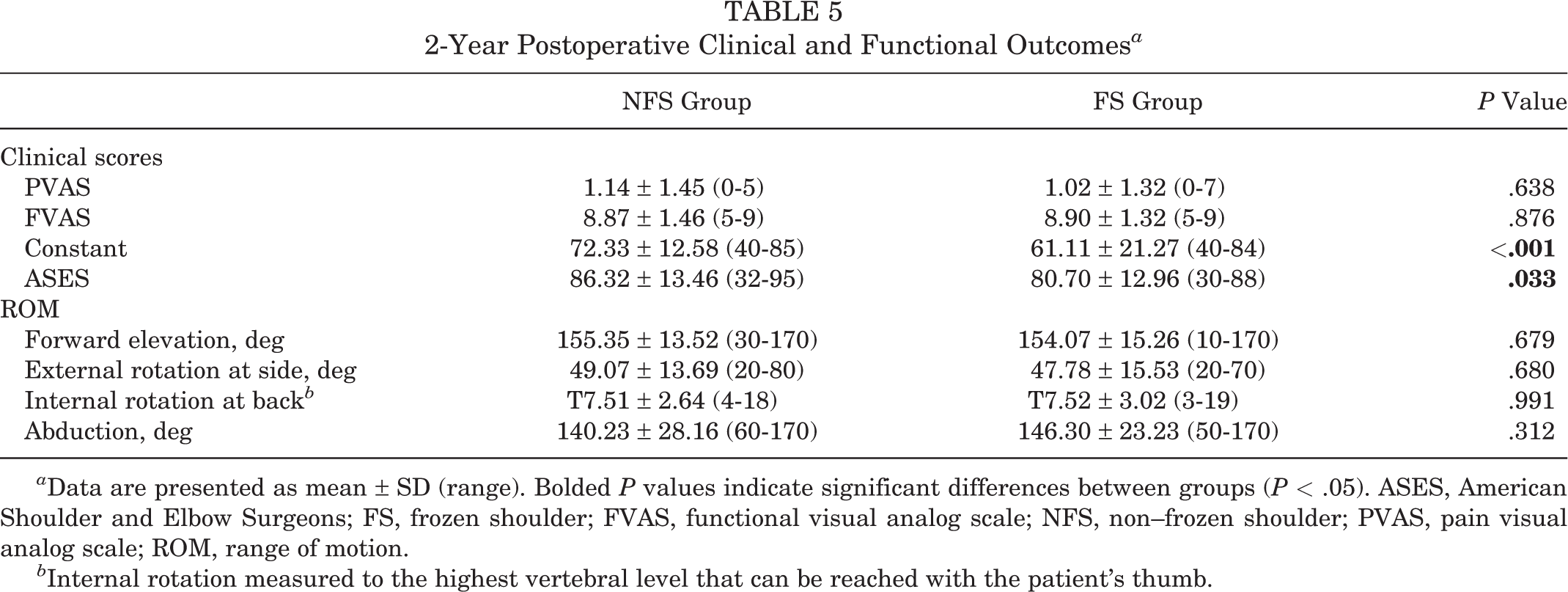
a Data are presented as mean ± SD (range). Bolded P values indicate significant differences between groups (P < .05). ASES, American Shoulder and Elbow Surgeons; FS, frozen shoulder; FVAS, functional visual analog scale; NFS, non–frozen shoulder; PVAS, pain visual analog scale; ROM, range of motion.
b Internal rotation measured to the highest vertebral level that can be reached with the patient’s thumb.
Retear Rates
Follow-up MRI was performed at a mean of 5.6 months (range, 4-7 months). Among the 58 patients in the FS group, 3 had a retear (5.3%), whereas a retear occurred in 19 of the 154 patients in the NFS group (12.3%). This difference was statistically significant, with a P value of .013. The interobserver and intraobserver reliabilities for the MRI evaluations was excellent (kappa = 0.85 and 0.94, respectively).
Analysis of RCT Size by Group
Interestingly, there were no significant differences in postoperative clinical outcomes between the patients with small versus large RCTs (Figures 3 and 4). On the other hand, for medium-sized RCTs, patients in the NFS group showed better preoperative and postoperative (6 months and 1 year) clinical outcomes than those in the FS group. However, there was no significant intergroup difference in the 2-year postoperative outcomes (Figure 5).

Comparison of outcomes between patients with frozen shoulder (group 1) and without frozen shoulder (group 2) and concomitant small-sized rotator cuff tears, including the pain visual analog scale (PVAS) score, functional visual analog scale (FVAS) score, American Shoulder and Elbow Surgeons (ASES) score, Constant score, forward flexion (FE), abduction (ABD), external rotation (ER), and internal rotation (IR). *P < .05. **P < .01. ***P > .05.

Comparison of outcomes between patients with frozen shoulder (group 1) and without frozen shoulder (group 2) and concomitant large-sized rotator cuff tears, including the pain visual analog scale (PVAS) score, functional visual analog scale (FVAS) score, American Shoulder and Elbow Surgeons (ASES) score, Constant score, forward flexion (FE), abduction (ABD), external rotation (ER), and internal rotation (IR). *P < .001. **P > .05.

Comparison of outcomes between patients with frozen shoulder (group 1) and without frozen shoulder (group 2) and concomitant medium-sized rotator cuff tears, including the pain visual analog scale (PVAS) score, functional visual analog scale (FVAS) score, American Shoulder and Elbow Surgeons (ASES) score, Constant score, forward flexion (FE), abduction (ABD), external rotation (ER), and internal rotation (IR). *P < .05. **P < .01. ***P < .001. ****P > .05.
Preoperative and Postoperative Stiffness
At 1 year after surgery, stiffness was noted in 50% of patients in the FS group and 39% of patients in the NFS group. The patients with preoperative frozen shoulder were more likely to have stiffness at 1 year postoperatively (P = .024).
Discussion
Our study results showed that frozen shoulder before RCT repair could negatively affect functional outcomes, including the objective recovery of ROM, during the 1-year postoperative period. However, we found that 2 years after surgery, there was no significant difference in active motion, PVAS scores, or FVAS scores between the FS and NFS groups. In addition, preoperative frozen shoulder had a positive effect on healing of the rotator cuff. Interestingly, preoperative frozen shoulder did not significantly affect the postoperative clinical outcomes of patients with small or large RCTs, but it did affect those of patients with medium-sized RCTs. Therefore, it is not necessary to increase the time to surgical treatment through additional physical therapy for preoperative frozen shoulder. Moreover, the retear rate was significantly lower in the FS group, so it might be recommended to perform surgery during this period when frozen shoulder combined with an RCT is present.
Frozen shoulder, either as a primary disease entity or as a secondary condition with causes such as certain surgical procedures and systemic disease, is known to affect patient quality of life by producing long-term pain and residual loss of ROM. 11 RCT repair of frozen shoulder has resulted in good clinical outcomes for several decades, and surgeons are now interested in reducing complications and improving early and long-term clinical outcomes. Frozen shoulder combined with RCT repair is a major concern for surgeons, especially in the early postoperative period, because preoperative frozen shoulder is a known risk factor for postoperative shoulder stiffness. 7
There are 3 commonly accepted stages in frozen shoulder. 22 The first stage is the “freezing” stage, which is recognized mainly by increasing pain and stiffness that last for approximately 6 months. The second stage is the “frozen” stage, which consists of steady stiffness for 4 to 20 months. The final stage is the “thawing” stage, which can be recognized by the relief of pain and stiffness lasting for 5 to 26 months. Several different definitions of frozen shoulder have been proposed by a number of authors according to their inclusion criteria. Huberty et al 13 defined the condition as the patients’ sense of disability due to restricted shoulder joint ROM. They reported that 24 of 489 patients (4.9%) who underwent arthroscopic RCT repair using their definition had postoperative frozen shoulder. However, their inclusion criteria for the diagnosis of frozen shoulder were somewhat vague.
Parsons et al 21 used precise data of ROM limitations in 2 directions: passive forward flexion <100° and external rotation <30°. They reported that 10 of 43 patients (23%) had frozen shoulder using their definition, which was more specific than that of Huberty et al 13 ; they also observed a higher incidence of frozen shoulder using their criteria. Using a somewhat different approach, Tauro 26 defined the limitation of ROM by total ROM in 3 directions: forward flexion and external and internal rotation in 90° of abduction. Recently, Chung et al 4 classified the condition as any ROM limitation that meets the specific criteria in any of 3 directions: forward flexion, external rotation at the side, and internal rotation at the back. They defined frozen shoulder as forward flexion <120°, external rotation <30°, or internal rotation lower than the L3 level, measuring with passive ROM. In their study, the prevalence of postoperative frozen shoulder was 2.8% to 18.6% at different time points in the follow-up period. In terms of classifying preoperative frozen shoulder, we used a definition similar to that of Chung et al. 4 We included another criterion, ROM compared with the unaffected side, to generate a definition of preoperative frozen shoulder that is more comprehensive and different from those used previously. Using these criteria, 27.4% of our patients had preoperative frozen shoulder, a higher rate than previous findings; for example, Oh et al 20 reported an incidence of preoperative frozen shoulder of 23.6%, while Hsu et al 12 reported a rate of 12.3%.
Oh et al 20 performed a cohort study that was somewhat similar to the present study. In patients undergoing RCT repair with manipulation performed under anesthesia and arthroscopic capsular release performed as the index procedure, the authors reported no significant difference in postoperative clinical outcomes between patients with and without frozen shoulder. 20 In our study, there was no significant intergroup difference in the PVAS score, a finding that is consistent with the results of the study by Oh et al; this result indicates that preexisting frozen shoulder does not influence postoperative pain outcomes. However, our FS group had worse clinical outcomes up to 1 year postoperatively compared with the NFS group, which is different from the previous study by Oh et al. A possible explanation for this discrepancy is that we implemented more comprehensive and different criteria for the classification of frozen shoulder.
McNamara et al 18 reported a relationship between postoperative frozen shoulder and RCT healing in a study of 1533 consecutive arthroscopic RCT repair procedures. In that study, the retear rates of patients with and without frozen shoulder at 6 weeks postoperatively were 7% and 15%, respectively. They reported that RCTs were more likely to heal in patients with postoperative stiffness. Conversely, our study reported a relationship between preoperative frozen shoulder and RCT healing. Our study also showed that preoperative frozen shoulder was associated with improved rotator cuff–bone healing. Unlike the abovementioned study, in which repair integrity was determined via ultrasound, we assessed all patients at 6 months postoperatively using higher accuracy MRI.
Because preoperative frozen shoulder is a risk factor for postoperative frozen shoulder, 7 we believe that there is a difference in the healing rate by limiting patient ROM in the early postoperative stage. We interpreted the outcomes shown 2 years after surgery as being in accordance with the natural history of frozen shoulder as shown by Neviaser and Hannafin, 19 who found that the condition of adhesive capsulitis resolves on its own.
The strengths of our study were its relatively large sample size and the inclusion of a control group. Additionally, all operative procedures were performed by a single experienced senior surgeon. We recorded the tear size, which could have significantly affected the clinical outcomes of patients with RCTs; thus, we were able to increase the reliability of the clinical data.
This study has several limitations. First, it was a retrospective review, so it carries the inherent limitations of this study design. Second, we had difficulty obtaining completely accurate data of 21 patients who did not visit the outpatient clinic during the follow-up period at 1 or 2 years postoperatively; their data were only obtained via telephone surveys. The outpatient follow-up rate was 90.1%. The results of this study might have been different with a higher data collection rate. Third, there was a large difference in the number of patients in the 2 study groups (n = 154 and 58); therefore, statistical validity could be questionable. More patients should be recruited, and a comparative study between 2 groups of similar sizes is needed. In addition, the subgroup analysis was limited by the small patient numbers in some subgroups. Fourth, we defined frozen shoulder using active assisted ROM. It is possible that active assisted ROM will be reduced compared with passive ROM. Finally, it was difficult to match groups because this was not a study with a very large sample size.
Conclusion
Preoperative frozen shoulder positively affected rotator cuff healing but negatively affected most functional outcomes, including ROM, at 6 months and 1 year postoperatively. At 2 years after surgery, there was no significant difference in active motion, but outcome scores remained lower in the FS group. For patients with preoperative frozen shoulder, a delay in surgery for additional physical therapy might not be necessary. The retear rate for the NFS group was more than double that for the FS group in the current study, which indicates that surgery for an RCT combined with frozen shoulder might offer better results in the long term because of an intact, healed rotator cuff.
Footnotes
Final revision submitted February 4, 2020; accepted February 17, 2020.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Samsung Medical Center Institutional Review Board (No. 2016-12-042).
