Abstract
Background:
Heat necrosis due to motorized drilling during anterior cruciate ligament (ACL) reconstruction could be a factor in delayed healing at the bone–tendon graft interface.
Hypothesis:
The process of osteointegration could be enhanced using manual drilling. It reduces the invasiveness of mechanical-thermal stress normally caused by the traditional motorized drill bit.
Study Design:
Controlled laboratory study.
Methods:
ACL reconstruction using semitendinosus tendon autografts was performed in 28 skeletally mature female New Zealand white rabbits, which were randomly divided into 3 groups. In group A (n = 12), the tunnels were drilled using a motorized device; in group B (n = 12), the tunnels were drilled using a manual drill bit; and group C (n = 4) served as a control with sham surgical procedures. The healing process in the tunnels was assessed histologically at 2, 4, 8, and 12 weeks and graded according to the Tendon–Bone Tunnel Healing (TBTH) scoring system. In addition, another 25 rabbits were used for biomechanical testing. The structural properties of the femur–ACL graft–tibia complex, from animals sacrificed at 8 weeks postoperatively, were determined using uniaxial tests. Stiffness (N/mm) and ultimate load to failure (N) were determined from the resulting load-elongation curves.
Results:
The time course investigation showed that manual drilling (group B) had a higher TBTH score and improved mechanical behavior, reflecting better organized collagen fiber continuity at the bone–fibrous tissue interface, better integration between the graft and bone, and early mineralized chondrocyte-like tissue formation at all the time points analyzed with a maximum difference at 4 weeks (TBTH score: 5.4 [group A] vs 12.3 [group B]; P < .001). Stiffness (23.1 ± 8.2 vs 17.8 ± 6.3 N/mm, respectively) and ultimate load to failure (91.8 ± 60.4 vs 55.0 ± 18.0 N, respectively) were significantly enhanced in the specimens treated with manual drilling compared with motorized drilling (P < .05 for both).
Conclusion:
The use of manual drilling during ACL reconstruction resulted in better tendon-to-bone healing during the crucial early weeks. Manual drilling was able to improve the biological and mechanical properties of bone–hamstring tendon graft healing and was able to restore postoperative graft function more quickly. Tunnel drilling results in bone loss and deficient tendon-bone healing, and heat necrosis after tunnel enlargement may cause mechanical stress, contributing to a delay in healing. Manual drilling preserved the bone stock inside the tunnel, reduced heat necrosis, and offered a better microenvironment for faster healing at the interface.
Clinical Relevance:
Based on study results, manual drilling could be used successfully in human ACL reconstruction, but further clinical studies are needed. A clinical alternative, called the original “all-inside” technique, has been developed for ACL reconstruction. In this technique, the femoral and tibial tunnels are manually drilled only halfway through the bone for graft fixation, reducing bone loss. Data from this study suggest that hamstring tendon–to–bone healing can be improved using a manual drilling technique to form femoral and tibial tunnels.
Successful anterior cruciate ligament (ACL) reconstruction using hamstring tendon grafts requires safe and fast healing of the tendons in the bone tunnels. 27 Nevertheless, the re-establishment of the soft tissue insertion at the bone still remains one of the greatest challenges in orthopaedics. 30,32 Graft-tunnel healing is a complex process influenced by many variables, including the type of graft used, method of graft fixation and tensioning, graft motion, and tunnel placement. 28,37 Fixation of the graft to bone remains the weakest link, as more than half of the failures occur at this site. 6,23 Better attachment of the tendon graft to bone could enable an earlier, faster, and safer return to sport activities as well as more aggressive rehabilitation programs. 5,33,36
Strategies in functional tissue engineering that combine cells, growth factors, and/or bioscaffolds as implants to mimic the natural enthesis are under investigation. 14,15,18 To date, none of these methods has led to a significant improvement in graft integration after ACL reconstruction. 10 However, no studies have examined the possible effects of different tunnel drilling methods on the graft healing process. 9,31 Studies on rat and canine models have shown that mechanical-thermal stress could delay bone healing as well as increase bone resorption, thus leading to graft integration failure. 24 Several other methods have been validated to study tendon graft–bone interface healing, such as analyzing gene expression levels 25 or evaluating the biomechanical behavior of the femur–ACL graft–tibia complex (FATC) through mechanical tests. 1,19,34
The research question of this in vivo study was whether the use of manual drilling for ACL reconstruction could enhance bone-graft osteointegration. The aim of our study was to histologically and biomechanically evaluate and compare the effects of manual drilling versus standard motorized drilling on the interface after ACL reconstruction. We hypothesized that the process of osteointegration could be enhanced by using manual drilling, as it reduces the invasiveness of mechanical-thermal stress normally caused by the traditional motorized drill bit. The use of manual drilling of the femoral and tibial bone tunnels during ACL reconstruction in a rabbit model may preserve the viability of the tissue by reducing bone necrosis, thus facilitating the integration of the graft at the interface and resulting in faster attachment healing.
Methods
This study was designed as a controlled laboratory study. The ethical review board for animal studies of our institution approved this study, and the experiments were performed according to the guidelines for the care and use of animals in research. A total of 53 skeletally mature female New Zealand white rabbits (age, 26 weeks; weight, 3.5-4 kg) were used in this study. For the histological arm of the study, 28 rabbits were randomly divided into 3 groups. ACL reconstruction with motorized drilling was performed in group A (n = 12). In group B (n = 12), ACLs were reconstructed using a custom-made manual drilling device. In group C (controls; n = 4), we performed a sham surgical procedure. The rabbits were euthanized at 2, 4, 8, and 12 weeks postoperatively: 7 rabbits at each time point (3 animals from the manual drilling group, 3 animals from the motorized drilling group, and 1 animal from the control group). For the biomechanical arm of the study, 25 rabbits were used and divided into 3 groups: 10 rabbits underwent ACL reconstruction with motorized drilling; 10 rabbits underwent ACL reconstruction with manual drilling; and 5 rabbits, used as controls, underwent a sham surgical procedure. The rabbits were euthanized at 8 weeks postoperatively.
Surgical Procedure
The animals were anesthetized with medetomidine subcutaneously and inhaled 3% isoflurane. The sterile field was prepared on the right stifle joint, properly shaved, and disinfected. Knee parapatellar arthrotomy was performed through a midline incision, and the ACL was completely dissected. Then, 2 tunnels were drilled at the center of the anatomic footprint. A special custom-made “T” drill bit was built with a 2.5-mm diameter compatible with both the motorized drill and the “T handle” for manual drilling (Figure 1). For the motorized drilling group, the tunnels were drilled using a surgical driller (System 5; Stryker) at an average speed of 1500 rpm, as per routine in clinical practice. The semitendinosus tendon was harvested, and the graft was prepared with a No. 2-0 Ethibond suture (Ethicon), passed through the bone tunnel, and attached to the neighboring periosteum with a metal washer at 30° of knee flexion. The wound was then sutured in anatomic layers. Postoperatively, all animals returned immediately to cage (52 cm in width, 35 cm in height, and 33 cm in depth) activity.

A custom-made “T” drill bit, compatible with both a motorized drill and a “T handle” manual drill.
Histological Study
At the specified time points during the healing phase, the 28 animals were euthanized, the distal femurs and proximal tibias were harvested, and the specimens were dissected free of all soft tissue. Except for the ACL graft being removed from the FATC, the specimens were transferred to 4% neutral-buffered formalin for 4 days and then decalcified in 10% EDTA (pH 7.4) for 3 weeks. The femur and tibia were then trimmed in an anterior-medial fashion to obtain a circular graft in 2 blocks of equal length along the perpendicular axis of the graft within the bone tunnel. The 2 blocks represent the proximal and distal regions of the tunnel. The samples were embedded in paraffin and sectioned into 3-µm slides. Hematoxylin and eosin staining was performed on selected slides. Slides were examined with a TE2000-U light microscope (Nikon Instruments).
Two blinded observers, a specialized pathologist subspecializing in bone diseases and sarcomas and a biologist (M.S. and R.T.), rated the integration of the tunnels according to the Tendon–Bone Tunnel Healing (TBTH) scoring system proposed by Lui et al (Table 1). 17 The TBTH score is a reliable, valid measurement for evaluating the histological outcomes of tendon graft–to–bone tunnel healing in ACL reconstruction. The reported intrarater and interrater reliabilities of the score are high, ranging from 87.6 to 95.5 and from 0.90 to 0.99 (all P < .001), respectively. 17 The TBTH scoring system analyzes 5 different parameters: graft degeneration, graft remodeling, percentage of fibrous tissue, collateral connection, and head-to-head connection. The highest possible score is 21 points.
Tendon–Bone Tunnel Healing Scoring System a

a According to Lui et al. 17
Biomechanical Study
At 8 weeks after surgery, the 25 animals were sacrificed and stored at −20°C until biomechanical testing was performed. For the tensile testing protocol, the specimens were dissected free of all soft tissue except the ACL graft, leaving the FATC. Uniaxial tensile testing was performed using a materials testing machine (Model 5965; Instron). The FATC was fixed to custom-made clamps and wrapped in saline-soaked gauze. Alignment of the FATC in the clamps was anatomic. 35 A preload of 1 N was applied. After cyclic preconditioning of the constructs between elongation limits of 0 and 0.75 mm (10 cycles at 5 mm/min), load-to-failure testing was performed at an elongation rate of 10 mm/min. Stiffness (N/mm) was calculated from the slope of the linear region of the load-elongation curve.
Statistical Analysis
Data were calculated as the mean ± SD. Statistical comparisons were made using 2-tailed t tests. P < .05 was considered significant.
Results
Surgery was successful in all the animals treated, and no postoperative infections or complications were observed. At the time of sacrifice, all of the reconstructed ACLs were intact. At gross observation, the graft appeared securely healed in the bone tunnel. At week 2 sacrifice and at all other time points, there was no difference between the manual and motorized drilling groups. No cartilage damage during the follow-up period between the time points analyzed was observed.
Histological Findings
Histological analysis of the healing interface demonstrated ingrowth of fibrovascular connective tissue, progressive collateral connection, head-to-head connection, and fibrocartilage developed in the 2 groups. The histological slices that are shown in Figure 2 are representative of and summarize the general trend that we observed for femoral and tibial tunnel healing at each time point analyzed.

Spatiotemporal differences in tendon-to-bone healing between the motorized drilling group and manual drilling group at different healing periods: (A, B) 2 weeks, (C, D) 4 weeks, (E, F) 8 weeks, and (G, H) 12 weeks. Group A shows a delay at the tendon-bone healing interface, while the interface is narrower and more organized in group B. Hematoxylin and eosin staining: ×40 original magnification. B, bone; IF, interface; T, tendon.
At 2 weeks of healing, we found in the bone wall a significantly greater amount of bone cellular necrosis (Figures 2 and 3) on the tissue around the tunnel in the motorized drilling group (group A) compared with the manual drilling group (group B) (Figure 4). The area at the interface showed highly vascularized fibrous tissue, with inflammatory lymphocyte cell and giant cell infiltrates (granulation tissue). The tissue appeared poorly organized; there was significant empty space and less head-to-head connection between the graft and bone (Figure 2) compared with the manual drilling group (Figure 2). At this time point, the graft in both groups showed signs of regeneration and degeneration. In the manual drilling group, we found more chondrocyte-like cells at the interface.

Histological evaluation of the motorized drilling group (group A) at 2 weeks. (A) Gap between the tunnel and graft. (B) Detail of bone cell necrosis at the tunnel wall. Hematoxylin and eosin staining: ×80 original magnification. IF, interface; T, tendon.
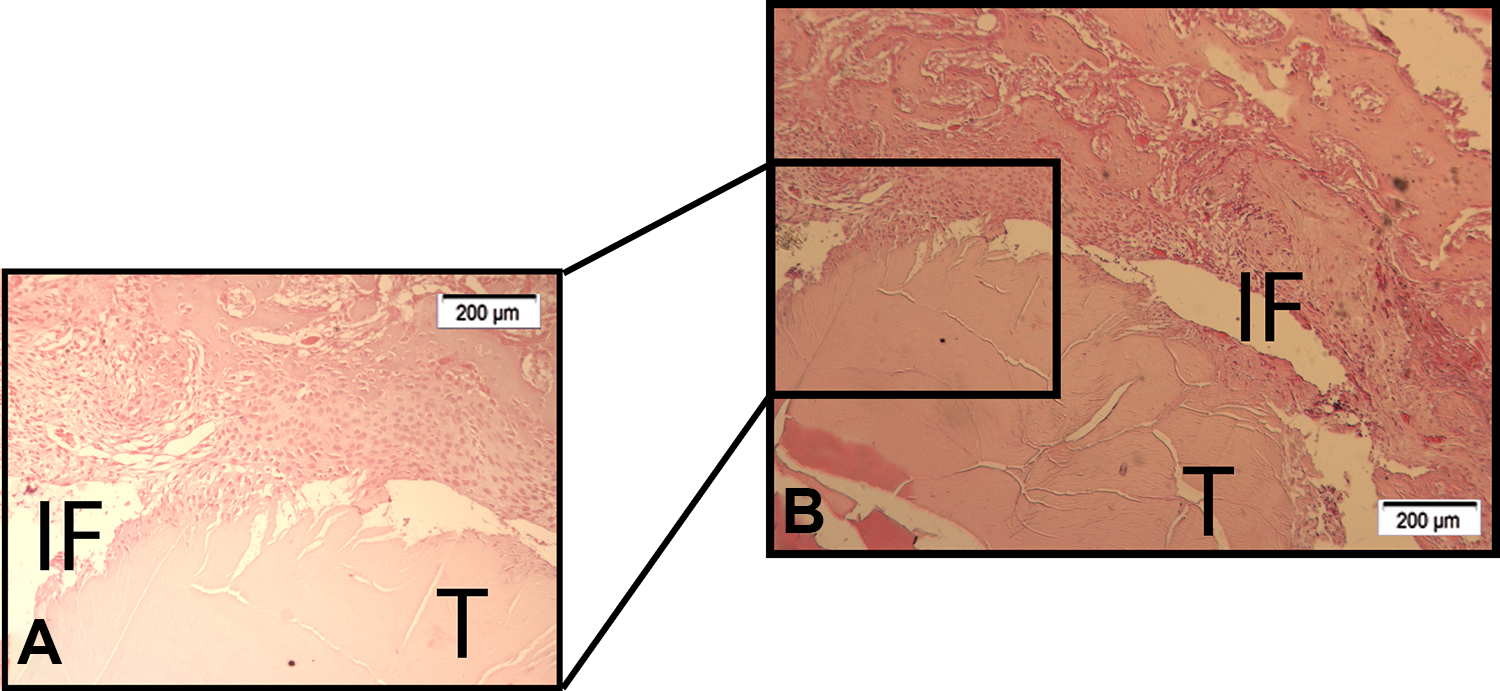
Histological evaluation of the manual drilling group (group B) at 2 weeks. (A) Detail of organized collagen fiber continuity at the bone–fibrous tissue interface. (B) Visible graft and bone with early mineralized chondrocyte-like tissue formation. Hematoxylin and eosin staining: ×80 original magnification. IF, interface; T, tendon.
At 4 weeks, the tendon in the tunnel remained viable with initial signs of remodeling in both groups. However, there was much more organized collagen fiber continuity at the bone–fibrous tissue interface in the manual drilling group (group B) (Figure 5). It was at this stage that we observed the greatest difference between the 2 groups, particularly in group B, in which there was better integration between the graft and bone with early mineralized chondrocyte-like tissue formation. Furthermore, a reduction in granulation tissue was observed with the formation of well-organized fibrous tissue (Figure 2). In group A, the graft presented greater signs of degeneration, which reflected negatively on the healing process (Figure 2).
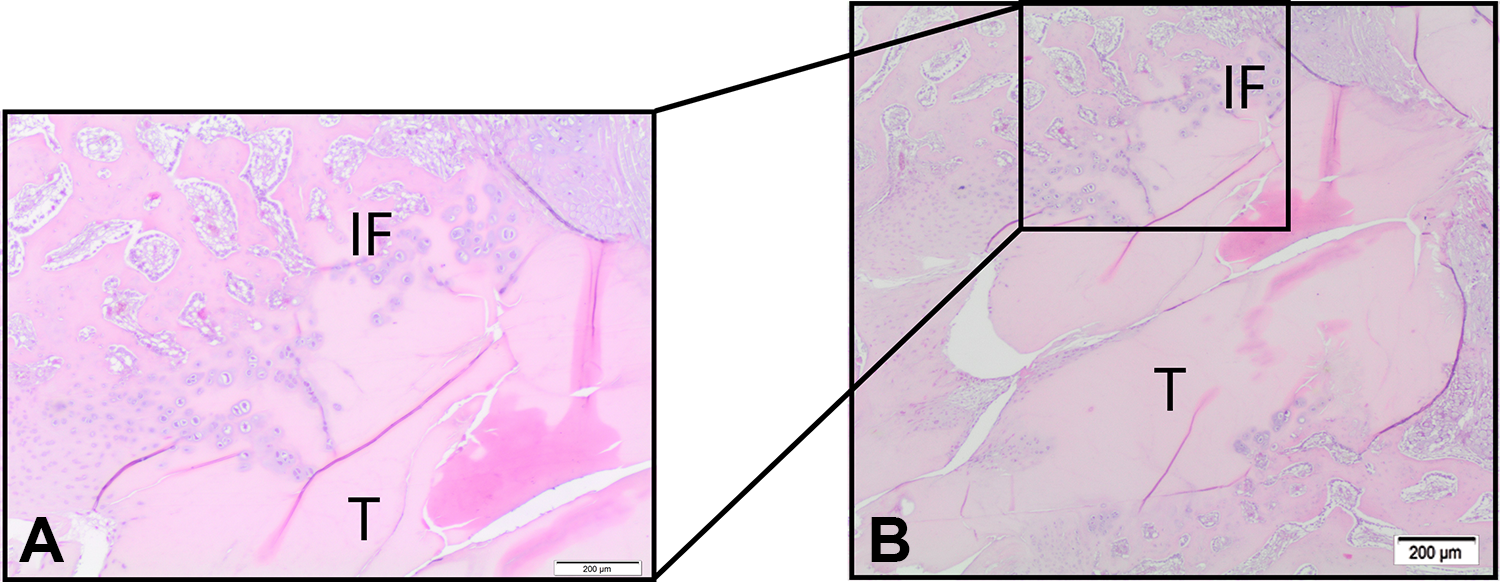
Histological evaluation of the manual drilling group (group B) at 4 weeks. (A) Detail of early mineralized chondrocyte-like tissue formation. (B) Signs of integration between the graft and bone. Hematoxylin and eosin staining: ×80 original magnification. IF, interface; T, tendon.
At 8 weeks after surgery, Sharpey fibers were mainly present in areas between the graft and fibrous tissue; a high number of fibers were present and bridged the newly formed fibrocartilage and graft in both groups (Figure 2). Remodeling of the trabecular bone around the tendon was also visible, but this was greater in the manual drilling group.
At 12 weeks of healing, there was increased organization and maturation of the fibrous tissue, with continuity of collagen fibers between the tunnel and tendon. Less fibrocartilage was found in the motorized drilling group, and there were large areas filled with partially mineralized chondrocyte-like tissue. The grafts in the manual drilling group showed increased density and organization, degeneration was less evident than in the motorized drilling group.
We analyzed all the slides using the previously mentioned TBTH scoring system and found statistically significant differences between the 2 groups at 2, 4, 8, and 12 weeks (P < .05 for all) (Table 2). Moreover, we analyzed the proximal and distal ends of the femoral and tibial tunnels and found the same trend, with better and faster healing in the manual drilling group for each time point analyzed (Figure 6).
Tendon–Bone Tunnel Healing Scores at Different Time Points

aP < .05 between groups.
bP < .001 between groups.

Time course of Tendon–Bone Tunnel Healing (TBTH) scores for proximal and distal areas of the femoral and tibial tunnels. (A) Proximal femoral tunnel, (B) distal femoral tunnel, (C) proximal tibial tunnel, and (D) distal tibial tunnel.
Biomechanical Findings
Biomechanical data of the specimens in the motorized drilling group (n = 10), manual drilling group (n = 10), and control group (n = 5) were obtained (Figure 7, A and B). Stiffness (23.1 ± 8.2 vs 17.8 ± 6.3 N/mm) and ultimate load to failure (91.8 ± 60.4 vs 55.0 ± 18.0 N) were significantly enhanced in the FATC treated with manual drilling compared with the values in the motorized drilling group (P < .05 for both). The control group showed significantly higher stiffness and ultimate load to failure (P < .05).

(A, B) Biomechanical proprieties of the femur–ACL graft–tibia complex after 8 weeks of healing. Values are presented as mean ± SD. *P < .05 compared with the motorized drilling group. ACL, anterior cruciate ligament.
Discussion
Heat necrosis due to motorized drilling during ACL reconstruction may contribute to a delay in healing of the bone–tendon graft interface. Manual drilling reduces the invasiveness of mechanical-thermal stress that is normally caused by a traditional motorized drill bit and may enhance the healing process. 7 Understanding the pathways of healing is fundamental to improve the outcomes of ACL reconstruction. In the present study, we demonstrated for the first time that manual drilling may enhance healing of the graft in ACL reconstruction in a rabbit model. Mechanical-thermal trauma from tunnel drilling could be a contributing factor to poor bone-to–tendon graft healing in ACL reconstruction. That thermal stress on bone occurs during drilling has long been accepted; it is clear that when the temperature reaches 47°C, it causes bone necrosis. 7,19 As reported by Wen et al, 32 the creation of tunnels could result in region-dependent stress shielding and subsequent bone loss, with deficient tendon-bone healing. Authors in clinical studies have suggested that heat necrosis is a cause of tunnel enlargement, blaming mechanical stress as a possible factor that delays healing. 12,22 A variety of improved drill bits and many precise parameters of drilling have been proposed to reduce thermal necrosis, without significant clinical improvements. Authors have mentioned that thermal necrosis could be a possible factor contributing to delayed healing, but they have not performed specific studies to clarify the topic. 9
Our results demonstrate that manual drilling could decrease bone necrosis and improve cell growth as well as biomechanical behavior. Tendon-bone healing still remains a complex and not completely understood process. It occurs with the production of new bone from the fibrovascular tissue present at the insertion site. As much preservation as possible of the insertion site is fundamental to obtain a better microenvironment. Moreover, the ACL enthesis is a complex structure. The morphology of ligament and tendon insertions to bone is among the most complex of all biological tissue. The transformation of soft to hard tissue requires a gradual transition of collagen fibers to nonmineralized fibrocartilage, then to mineralized fibrocartilage, and finally to bone. 2,30 Biological solutions to stimulate graft incorporation within the bone tunnel appear to have potential. 16,21 Only a few studies have analyzed the mechanobiological effects of drilling. 9,31 However, research in animals with various augmentation techniques is promising. 11
Several studies have evaluated the efficacy of stimulation with growth factors to enhance the healing process within the bone tunnels. 8,21 Other studies have investigated enveloping the tendon graft with the periosteum. 4 In an animal model, we investigated the histological changes that occur inside the femoral and tibial tunnels at 2, 4, 8, and 12 weeks after ACL reconstruction with manual and motorized drilling, and after 8 weeks, we tested the specimens to obtain the structural properties of the FATC. A group of control animals was used to compare the histological aspects of the ACL enthesis at the same time points in the same portion of the tunnel. Healing of the graft inside the bone tunnels proceeded as previously reported from the inflammatory to proliferative and finally reparative phases. 13,20,38 Healing occurred with the formation of dense fibrous tissue with occasional Sharpey fibers. 29 In the manual drilling group, the grafts healed in a zone of fibrocartilage, showing a gradual transition from bone to mineralized fibrocartilage, then to cartilage, and finally to the tendon graft. It has been reported that this type of healing is more physiological. This graft also looks like the normal ACL enthesis. 26 This was confirmed by our biomechanical data.
In this study, all the histological slides were evaluated using a simple, standardized, and validated instrument for the objective assessment of tendon graft–to–bone tunnel healing in ACL reconstruction: the TBTH scoring system. 17 The use of this tool allowed us to compare the histological data obtained, in an objective manner, overcoming the general limitation of histological studies. We are aware of the limitations of the present study: (1) this was an animal study, and (2) only decalcified slides were used, so it was not possible to view new bone formation or its quality and quantity.
The results of this study support our hypothesis that performing manual drilling leads to better graft-tunnel osteointegration during the early crucial weeks of healing. Our data suggest that developing an ACL reconstruction technique that uses manual drilling may be more effective than techniques that use motorized drilling.
Based on our results, manual drilling could be used successfully in human ACL reconstruction, but further clinical studies are needed. A clinical alternative, called the original “all-inside” technique, has been developed for ACL reconstruction. 3,39 In this technique, the femoral and tibial tunnels are manually drilled only halfway through the bone for graft fixation, reducing bone loss.
Conclusion
The data from this study suggest that hamstring tendon-to-bone healing can be improved using manual drilling to form femoral and tibial tunnels. Manual drilling may be used to mechanically enhance the healing process of hamstring tendon grafts within the bone tunnels by preserving the bone stock inside the tunnel, reducing heat necrosis and thus leading to a better microenvironment for faster healing.
Footnotes
Final revision submitted October 29, 2019; accepted November 8, 2019.
One or more of the authors has declared the following potential conflict of interest or source of funding: Financial support for this study was provided by Nicola’s Foundation Onlus. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Comitato Universitario di Bioetica, Università degli Studi di Perugia.
