Abstract
Background:
Articular cartilage damage is caused by traumatic sport accidents or age-related degeneration and might lead to osteoarthritis, which represents a socioeconomic burden to society. Cartilage damage in the knee is commonly treated surgically with microfracture (MFX) or matrix-associated autologous chondrocyte implantation (MACI).
Purpose:
To quantify the initial and follow-up costs associated with MFX and MACI treatments from the viewpoint of statutory health insurance in Germany.
Study Design:
Economic decision analysis; Level of evidence, 2.
Methods:
This comparative study was based on an anonymized representative claims data set of 4 million patients covered by statutory health insurance in Germany. Patients undergoing outpatient or inpatient treatment with MACI or MFX for cartilage damage in the knee between January 1, 2012, and December 31, 2013, were included and evaluated over 5 years. Groups (MACI and MFX) were adjusted via propensity score matching before initial treatment. The matched groups were compared regarding their outpatient, inpatient, pharmaceutical, and other costs during the 5-year period.
Results:
In total, 127 patients per group were analyzed (59.1% male, 40.9% female; mean age, 37 years). In the year of the initial surgical procedure, costs were €14,804.13 in the MACI group and €5458.59 in the MFX group. In years 2 and 3 after initial surgery, treatment costs were comparable between patients treated with MACI (€2897.97 and €2114.87, respectively) and MFX (€2842.66 and €1967.42, respectively), with slightly higher treatment costs for those treated with MACI. In years 4 and 5 after surgery, costs were less in patients treated with MACI (€2154.79 and €1478.08, respectively) than in those treated with MFX (€2232.57 and €2061.63, respectively). Costs related to revision surgery were, on average, €3732 for MACI and €3765 for MFX. Thus, additional costs in years with revision surgery were €1672 for MACI and €1915 for MFX.
Conclusion:
This was the first study to analyze a large representative population claims database with propensity score matching, and results indicated that follow-up costs of patients treated with MACI and MFX began to converge over time. We found that total costs for MACI were higher than for MFX but that additional costs for MACI were lower than previously reported. Perceived morbidity may have little to do with cost.
Keywords
Articular cartilage defects in the knee are, among other reasons, caused by accidents such as torsion or by age-related degeneration. They lead to the restriction of mobility 4,9,28 and represent a socioeconomic challenge to society. 21,30 If these cartilage defects remain untreated, premature osteoarthritis might occur. 1,2,6,8,17,23 Surgical procedures such as microfracture (MFX) and matrix-associated autologous chondrocyte implantation (MACI) are available to restore function, relieve pain, and reduce the probability of secondary osteoarthritis. 1 MFX, a marrow-stimulating procedure, is performed by drilling the subchondral bone lamella, leading to blood and stem cells entering the defect area. 24 –26 In past decades, MFX was increasingly performed because of its technical simplicity, minimal invasiveness of the procedure, and low costs. 1 MFX was proposed for smaller lesions and in patients with a lower level of activity. 22 However, besides the risk of weakening the subchondral structure, in some cases the quality of the regenerated tissue was insufficient, especially for larger lesions in patients with a higher level of activity. 14,27
Regarding associated health care costs, a 2017 review that created decision trees to examine the cost-effectiveness of MFX and autologous chondrocyte implantation (ACI) found that MFX treatment had lower costs and lower costs per point increase for the Hospital for Special Surgery knee score, 36-Item Short Form Health Survey, and Knee injury and Osteoarthritis Outcome Score (KOOS). 1 On the other hand, a study from 2010 pointed out that ACI treatment had higher costs compared with MFX treatment but that MACI can be considered a cost-effective treatment in the long run. 7 A National Institute for Health and Care Excellence (NICE) assessment from 2018 reported an incremental gain of 0.1131 quality-adjusted life years (QALYs) for MACI treatment compared with MFX treatment. 16
However, the existing studies obtained cost data mainly from calculations and assumptions. Thus, there is only limited information based on real-world cost data from the viewpoint of German statutory health insurance (SHI). Therefore, this study aimed to compare treatment costs from the perspective of the German health system based on claims data to evaluate the economic health burden of MACI and MFX in Germany during a 5-year period.
Methods
Study Design and Data Source
This cohort study was based on an anonymized German health claims database including 4 million insured people from German SHI. The data set included 5% of the population covered by SHI from January 1, 2012, to December 31, 2017. The data set was stratified by age and sex to the demographic structure of the German population. It contained information on patients’ diagnoses, treatment settings (ie, inpatient and outpatient claims), surgical and nonsurgical treatments, demographic data, and costs. Cost data were derived from actual claims in the SHI system and comprised outpatient costs, inpatient costs, pharmaceutical costs, and other costs (which included, for example, costs for medical remedies and medical devices and costs for rehabilitation and physical therapy). The study design was predefined by a detailed analysis protocol following the recommendation of the German Society for Epidemiology.
Patient Eligibility and Follow-up
Patients with at least 1 MACI or MFX procedure performed in the period between January 1, 2012, and December 31, 2013 (index period), in the outpatient or inpatient setting, were identified by the corresponding claims and included in the present study. If MACI and MFX were performed in the same patient within the index period, the patient was assigned to the treatment that was performed first.
MFX and MACI were defined by the following procedure codes, which are used in Germany in inpatient and outpatient claims: 5-801.kh and 5-812.hh for MACI and 5-812.fh and 5-801.hh for MFX. Each patient was evaluated for 5 years after his or her initial surgery. Patients without a 5-year observation period, for example, because of a change in insurance plans, were excluded from the study.
Outcome Assessment and Variables
The primary outcome of the present study was the costs associated with MFX and MACI treatment in a 5-year period after initial surgery. To identify relevant procedure codes for revision surgery, the German classification of medical procedures (OPS), an adaptation of the International Classification of Procedures in Medicine published by the World Health Organization, was searched for 4 search strings: (1) knee joint, (2) meniscus, (3) patella, and (4) prosthesis and knee replacement. Revision surgery was then assessed as the first claim of the following codes: surgery in the knee joint (27 procedure codes), meniscus (35 procedure codes), and patella (102 procedure codes) or the need for knee replacement (11 procedure codes).
Statistical Analysis
Costs were summed up on a patient level to obtain total costs per patient. Furthermore, these costs were stratified by each follow-up year and in years with and without a claim for revision surgery.
To reduce selection bias, both groups were risk adjusted to baseline by using a matched-pair approach. Risk adjustment was performed with a mix of direct matching and propensity score matching, as proposed by Rubin and Thomas, 20 to effectively reduce potential confounders. The propensity score was estimated by using multivariate logistic regression (logit model) with MACI serving as the modeled outcome. Thereby, the 20 most frequently prescribed concomitant medications (Anatomical Therapeutic Chemical Classification System) and comorbidities (International Classification of Diseases, 10th Revision–German Modification category) coded in the outpatient or inpatient (main and secondary diagnoses) setting within the index period were included as covariates in the logistic regression.
Risk factors used in direct matching, in addition to propensity score estimation, were defined as year of index treatment, age, and sex in the index period for the following reasons: (1) Age: In several reports, ACI is not recommended in patients aged between 40 and 50 years. Recommendations on older patients are not available. 11,18 Further studies have pointed out that surgery for cartilage defects is mostly performed in patients aged between 31 and 36 years 19 and younger than 39 years. 29 (2) Sex: Isolated patellofemoral defects are more commonly diagnosed in female versus male patients (24.3% vs 11.0%, respectively), whereas isolated medial defects are more commonly diagnosed in male versus female patients (21.2% vs 12.3%, respectively). 12 Because of hormonal changes, menopausal women older than 50 years have a higher risk of developing osteoarthritis, which is of importance because the majority of osteoarthritic patients are female (72.8% vs 27.2%). 10
Considering the subsequent matching process, year of index treatment and sex were specified to fit exactly, whereas a variance of ±5 years in age was permitted (caliper). Estimated propensity scores were allowed to vary by ±0.2 of the SD of propensity score estimation.
Data were stored and analyzed using Office Excel (2019; Microsoft) and SAS (version 9.3; SAS Institute). The GenMatch algorithm was performed using R, the free statistical software.
Results
Propensity Score Matching
The C-statistic value of propensity score matching was 0.85, indicating a good classification of the propensity scores. 31 Thus, patients of both groups, MFX and MACI, were well comparable with each other.
Demographic Characteristics
Before matching, 6425 patients met the inclusion criteria within the index year. Of these, 2.4% were initially treated with MACI (n = 152), and 97.6% were in the MFX group (n = 6273). The mean age was 35.98 ± 11.14 years for MACI-treated patients and 53.04 ± 14.00 years for MFX-treated patients. There were 60.53% male versus 39.47% female patients in the MACI group and 54.31% male versus 45.69% female patients in the MFX group. After matching, 127 patients, with a mean age of 36.84 ± 10.91 years in the MACI group and 36.94 ± 10.86 years in the MFX group, were identified for each group. Of these, 59.06% were male, and 40.94% were female (Table 1).
Patient Characteristics Before and After Matching

Costs per Treatment
Mean total costs over 5 years amounted to €23,449.83 (95% CI, €22,271-€24,628) in MACI-treated patients and €14,562.92 (95% CI, €13,491-€15,633) in MFX-treated patients (Δ€8886.91 [95% CI, €6637-€11,136]), and thus, total costs for MACI were higher than for MFX (Figure 1). Mean annual costs were €4689.97 per MACI-treated patient (95% CI, €3488.09-€5891.85) and €2912.58 per MFX-treated patient (95% CI, €2018.00-€3807.16). Thus, MACI treatment was associated with 1.6 times the cost of MFX treatment.

Total cost and cost per category over 5 years. MACI, matrix-associated autologous chondrocyte implantation; MFX, microfracture.
Higher costs for MACI versus MFX were seen in inpatient costs (€13,360.48 [95% CI, €12,533-€14,187] vs €5234.35 [95% CI, €4652-€5815], respectively) as well as other costs (€5104.33 [95% CI, €4713-€5495] vs €3186.32 [95% CI, €2729-€3643], respectively). Lower costs for MACI- versus MFX-treated patients were seen in outpatient costs (€3,347.71 [95% CI, €3166-€3528] vs €4206.97 [95% CI, €4004-€4409], respectively) and pharmaceutical costs (€1637.31 [95% CI, €1383-€1891] vs €1935.28 [95% CI, €1547-€2323], respectively).
In a longitudinal view, total costs within the year of initial surgery were €14,804.13 (95% CI, €13,768-€15,839) in the MACI group and €5458.59 (95% CI, €4549-€6368) in the MFX group. In years 2 to 5 after initial surgery, treatment costs were €8645.70 for MACI (95% CI, €7691-€9600) and €9104.33 for MFX (95% CI, €8293-€9914) (Figure 2).
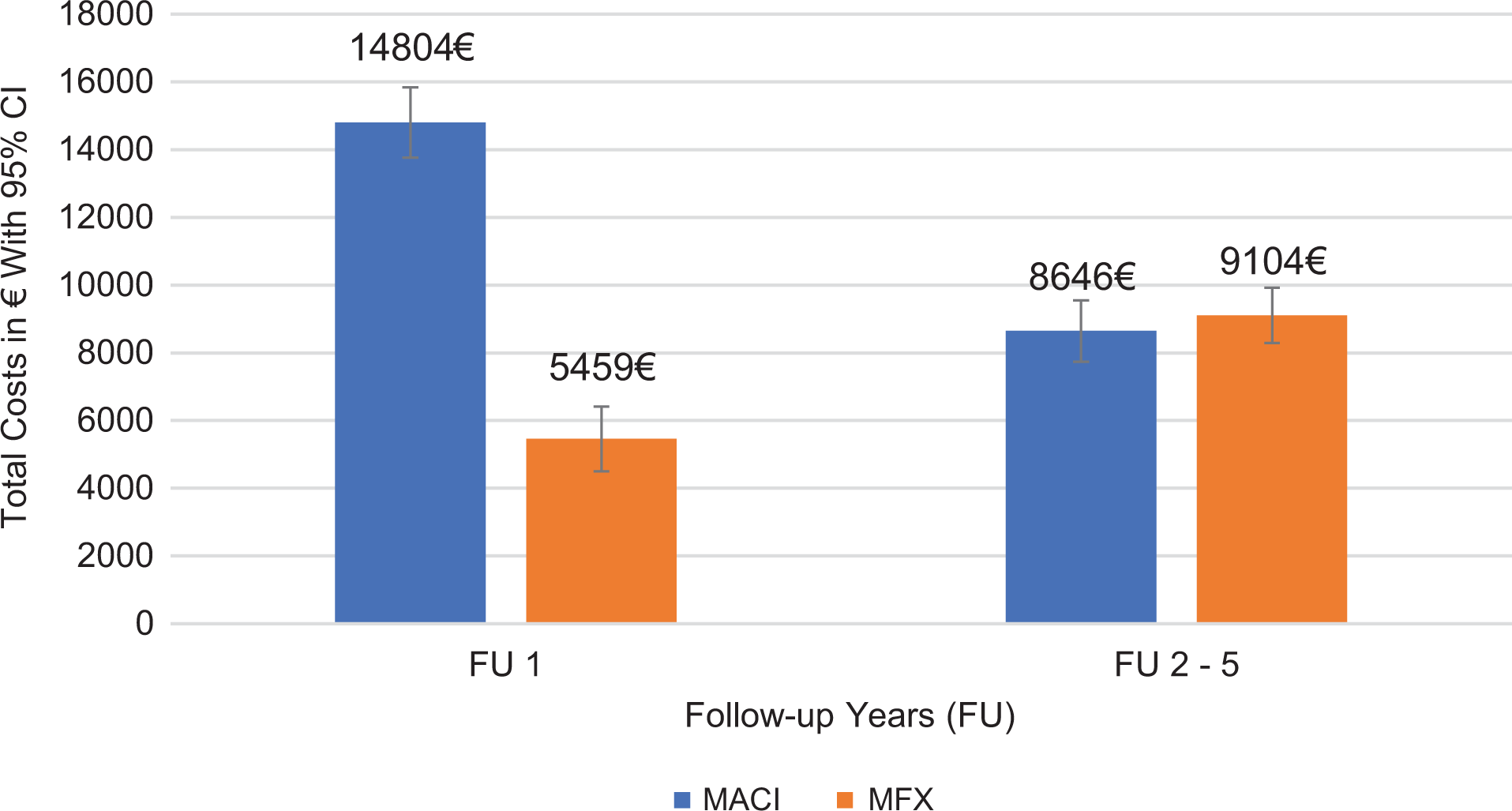
Total cost in the index year and follow-up years 2 to 4. FU, follow-up; MACI, matrix-associated autologous chondrocyte implantation; MFX, microfracture.
In years 4 and 5 after initial surgery, health care costs of MFX-treated patients were higher than those of MACI-treated patients (year 4: €2154.79 [95% CI, €1446-€2861] for MACI and €2232.57 [95% CI, €1506-€2957] for MFX; year 5: €1478.08 [95% CI, €1079-€1876] for MACI and €2061.63 [95% CI, €1459-€2662] for MFX) (Figure 3).

Total cost in follow-up years 2 to 5. FU, follow-up; MACI, matrix-associated autologous chondrocyte implantation; MFX, microfracture.
Mean total costs in years with the need for revision surgery were €3732 for MACI- and €3765 for MFX-treated patients. Thus, additional costs of €1672 for MACI and €1915 for MFX occurred in years with the need for revision surgery (Figure 4).

Comparison of health care costs in years with and without revision surgery. MACI, matrix-associated autologous chondrocyte implantation; MFX, microfracture.
Discussion
The present study is the first to analyze follow-up costs of MACI and MFX based on health claims data from a European SHI point of view. The results showed higher costs for MACI than for MFX in the year of initial treatment (€14,804.13 vs €5458.59, respectively), but costs of both treatments started to converge during a follow-up period of 5 years (follow-up costs of €8645.70 and €9104.33, respectively).
In 2016, about 3000 MACI and 60,000 MFX procedures were performed in Germany. 5 Multiplying the total costs after 5 years (€23,449.83 for MACI and €14,562.92 for MFX) with this number of treatments in 2016, total treatment costs of €70,349,490 for MACI and €873,775,200 for MFX would occur. Considering additional costs of €1672 for MACI and €1915 for MFX in years with revision surgery, the higher costs of MACI could partially be offset by savings in revision surgery in future years, although savings in these years are low compared with the total 5-year costs of both procedures.
In 2017, Aae et al
1
conducted a literature search on evidence level 1 and 2 studies with a follow-up period of at least 5 years to estimate the cost-effectiveness of ACI compared to MFX. Based on the medical results in the identified studies, a decision tree with associated service provisions and costs was designed. Cost data were extracted from a Norwegian orthopaedic hospital and acknowledged by other Norwegian orthopaedic hospitals. Comprising 319 patients (170 with MFX and 149 with ACI), the study showed total direct costs for MFX treatment of €3254 at baseline in the year of surgery, increasing to €3892 after 5 years (Δ€638) and €11,013 at baseline rising to €11,558 for ACI treatment (Δ€545).
1
Results were in line with our findings, which showed higher overall costs for MACI treatment (€23,449.83) compared with MFX treatment (€14,562.92) and also indicated that the majority of costs were incurred in the year of surgery. However, we found a downward cost trend in MACI treatment after the initial year of surgery (€14,804.13 in year 1 falling to €8645.70 in years 2-5), whereas MFX treatment increased within the follow-up years (€5458.59 in year 1 rising to €9104.33 in years 2-5). This deviation from the Aae et al study might be caused by limitations in their study due to assumptions concerning the length of hospital stay and hospital costs and the exclusion of outpatient costs (except assumed postoperative physical therapy costs) as well as the comparison with first-generation ACI. In addition, included revision surgery in the earlier study were restricted to magnetic resonance imaging and return visits, and there was no further definition of which costs were subsumed under the term
A US retrospective review from 2015 based on PearlDiver, a national private insurance database, examined the preoperative and postoperative costs for the evaluation as well as surgical treatment of symptomatic focal cartilage defects in the knee. 32 Costs included outpatient visits, imaging, rehabilitation, joint injections, and repeat procedures in case of postoperative complications. Among others, ACI (546 patients) and MFX treatments (38,444 patients) were analyzed, with costs amounting to $16,016.70 (∼€14,205) in MACI-treated patients and $7258.51 (∼€6402) in MFX-treated patients in a time horizon from 1 year preoperatively to 1-year postoperative follow-up. Mean costs for secondary procedures in cases of complications were $730.00 (∼€644) for MACI and $231.16 (∼€204) for MFX treatment. Regarding the trend of higher total costs of MACI treatment, these findings are in line with our results. Regarding costs associated with secondary procedures, the present study found in contrast that MACI was associated with lower costs than MFX treatment. The deviation in the latter aspect might be caused by the comparison with first-generation ACI, the shorter observational period of 2 years (1 preoperative year as well as 1 postoperative year), and the sparse information regarding the definition of secondary procedures in the Zhang et al 32 study.
A 2010 study by Gerlier et al 7 examined the cost-effectiveness of MFX treatment compared with ACI treatment in Belgium using clinical and quality-of-life data from a phase 3 randomized controlled trial. The authors combined these findings with a patient chart review to estimate local medical costs after knee surgery and a decision tree model to compare ACI with MFX in symptomatic cartilage lesions of the femoral condyles that had not yet developed osteoarthritis. The study covered a time horizon of 40 years from the point of view of a global health care payer; a short-term analysis after 5 years indicated total costs of €24,918 in ACI-treated patients and €2413 in MFX-treated patients. 7 Costs of MACI treatment were comparable with our findings, whereas costs of MFX treatment in the Gerlier et al study were underestimated. Deviating results might be due to the model assumptions, as follow-up costs for both treatment options were based on a calculation of a German and Belgian retrospective chart review comprising 82 patients. The subsequent calculation was conducted by the multiplication of these medical resources and the corresponding unit cost. In contrast, the current study used real claims data without referring to further calculation models.
Besides the difference in treatment costs, MACI has been compared with MFX in several studies regarding clinical results, including randomized controlled trials. In these clinical studies, the advantages of MACI over MFX were found with respect to better pain and functionality outcomes, 2 improvement on the KOOS, 3 and fewer adverse events. 13 In the 2018 NICE assessment in which MACI was found to have an incremental gain of 0.1131 QALYs in a 5-year period, incremental costs in the same time period were estimated to be £8524 (€9950). 16 The incremental cost-effectiveness ratio (ICER) per QALY after 5 years was thus £75,395 (€86,650; incremental costs of £8524/0.1131 QALYs) for MACI. 16
Based on the results of the current study, the ICER per QALY after 5 years would be €78,567 (incremental costs of €8886/0.1131 QALYs). Our results thus suggest that the long-term cost-effectiveness of MACI might be better than previously assessed, potentially lowering the lifetime ICER per QALY of £4360 (€5000) for MACI compared with MFX. 15
The main strengths of our study were the inclusion of unaltered cost data associated with the actual medical care situation, owing to representative health claims data from the German SHI system. The long observational period of 5 years enabled us to examine the costs not only of initial surgery but also 5 years after the initial treatment. Additionally, we minimized selection bias with a risk adjustment of patients by means of propensity score matching. Finally, the database used was a representative population sample, thus leading to high external validity of our results.
A limitation of the present study was that it was based on data from the German SHI system. Thus, patients with private health insurance (∼10% of the population) were not included in the present study. Furthermore, lesion size was not available in the database and could not be used in propensity score matching or to assess outcomes. Also, it is possible that both the size and the location of the lesions were different between groups and may have affected the results. Furthermore, this study was of a descriptive nature, so no statistical hypotheses were assessed.
Conclusion
We showed for the first time in a large representative European population and unselected claims database with propensity score matching that the follow-up costs of MACI- and MFX-treated patients begin to converge in a 5-year time period. We found that total costs for MACI were higher than for MFX, but the additional costs for MACI treatment were lower than previously reported, and therefore, the ICER per QALY gained over a lifetime could be even lower than the £4360 as assumed by the NICE. 15 As previously reported, 16 the cost-effectiveness of MACI is not demonstrated by direct cost savings but rather by its higher effectiveness. MACI might therefore be a more cost-effective treatment in a shorter follow-up period than previously thought.
Footnotes
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was sponsored by co.don. P.N. has received educational support from co.don. T.S. and A.H. have received consulting fees from co.don. M.G. is employed by co.don. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
