Abstract
Background:
Selection of optimal treatment for massive to irreparable rotator cuff tears (RCTs) entails a challenging decision-making process in which surgeons must consider several factors, including duration of symptoms, tear pattern, tear size, and muscle quality, as well as patient characteristics such as age, comorbidities, shoulder dominance, and activity level. Unfortunately, no clear consensus has been reached regarding optimal management.
Purpose:
To systematically review the published literature assessing outcomes after subacromial balloon spacer implantation for treatment of massive and irreparable RCTs.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
A comprehensive literature search was performed in September 2018 through use of MEDLINE and the Cochrane Library electronic databases. Studies were assessed for multiple outcomes of interest including Constant score, Oxford Shoulder Score (OSS), University of California Los Angeles (UCLA) Shoulder Score, complications, and patient satisfaction.
Results:
After applying the selection criteria, 12 clinical studies were included for data extraction and analysis. In total, 291 shoulders (in 284 patients) treated with subacromial balloon spacer implantation were pooled for evaluation, with a mean follow-up of 22.9 ± 14.9 months (range, 6-60 months). Constant scores were used as an outcome metric for 267 shoulders (91.7%; 11 studies), with improvements in mean Constant score ranging from 18.5 to 49.6 points. Patient satisfaction was assessed in 105 patients (37.0%; 5 studies), with rates of patients indicating they were satisfied or very satisfied with their treatment outcome ranging from 45.8% to 100%. A total of 6 patients (2.1%) experienced complications related to balloon spacer implantation, including transient neurapraxia of the lateral antebrachial cutaneous nerve, superficial wound infection, deep wound infection, and balloon migration. Of these, 3 patients (2 balloon migration, 1 deep wound infection) required subsequent surgeries for balloon removal.
Conclusion:
Placement of the subacromial balloon spacer is a minimally invasive, technically simple procedure with favorable patient-reported outcomes at limited short-term follow-up. However, inherent methodological limitations and patient heterogeneity between studies may impair our ability to fully characterize the longer term efficacy, particularly relative to other potential surgical options. Further prospective randomized or comparative studies are warranted to ascertain clinical outcomes of subacromial balloon spacer in the management of massive and irreparable RCTs.
Selection of optimal treatment for massive to irreparable rotator cuff tears (RCTs) requires a challenging decision-making process in which surgeons must consider several factors, including duration of symptoms, tear pattern, tear size, and muscle quality. Patient characteristics such as age, comorbidities, shoulder dominance, and activity level also must be considered. Cofield et al 12 classified massive tears as those greater than 5 cm in anteroposterior length. Gerber et al 24 defined massive tears as those involving 2 or more rotator cuff tendons. Surgical repair of RCTs with these characteristics is often technically challenging and is associated with higher rates of treatment failure or only modest clinical improvement. 1,12 Rockwood et al 43 described irreparable RCTs as those in which a direct repair of the native tendon to its insertion site is not possible. Signs of irreparability include fatty infiltration affecting 50% or more of the rotator cuff musculature, static superior migration of the humeral head, and a narrowed or absent acromiohumeral interval on an upright anteroposterior shoulder radiograph. 4
Treatment for such tears spans a spectrum that includes nonoperative treatment; surgical options such as subacromial decompression, biceps tenotomy or tenodesis, tuberoplasty, partial RTC repair; and more extensive and complex procedures such as superior capsular reconstruction, tendon transfers, and reverse total shoulder arthroplasty (RTSA). Unfortunately, no clear consensus has been reached regarding optimal management. When nonoperative treatment fails, operative management is often indicated to reduce pain, restore shoulder function, and decrease likelihood of cuff tear arthropathy that can cause progressive destruction of the glenohumeral joint. 15
A relatively new treatment modality for patients with massive to irreparable RCTs is the InSpace (Orthospace) biodegradable balloon system. The balloon is composed of a polylactide and ∊-caprolactone copolymer that is believed to biodegrade within 12 months. 10,33,45 The procedure can be performed under general or local anesthesia, and the balloon is most commonly inserted arthroscopically though a lateral portal. The device is designed to serve as a physical barrier to reduce subacromial friction, and it aims to restore proper shoulder biomechanics by lowering the humeral head closer to its anatomic position against the glenoid cavity during dynamic movements. 45 The technique was described by Savarese and Romeo 45 in 2012, and the earliest results of clinical outcomes were published by Senekovic et al 47 in 2013. Subsequent studies have used the balloon, both alone and in combination with other procedures such as subacromial decompression, biceps tenotomy, acromioplasty, bursectomy, and partial tendon repair. 52
The purpose of this study was to critically review the literature reporting clinical outcomes of subacromial balloon spacer implantation for the treatment of massive and irreparable RCT. We hypothesized that postoperative clinical outcomes will demonstrate pain reduction and improved shoulder function.
Methods
A systematic review of the literature was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines 35 to assess outcomes of subacromial balloon spacer implantation for massive and irreparable RCTs. This review was registered with the PROSPERO international prospective register of systematic reviews (PROSPERO registration identification: CRD42019123408).
Search Strategy
A comprehensive literature search was performed through use of the MEDLINE and Cochrane Library electronic databases. The following terms were used as keywords and appeared in the title, abstract, or keyword fields: (1) massive rotator cuff tear (n = 908); (2) irreparable rotator cuff tear (n = 464); (3) subacromial spacer (n = 30); subacromial balloon (n = 13); and balloon spacer (n = 40). The final search was performed on September 25, 2018.
Study Selection
Trials were eligible for inclusion if they met the following criteria: human participants with documented massive and/or irreparable RCTs, subacromial balloon spacer implantation, and patient outcome after at least 6 months of follow-up for clinical trials. Studies involving animals, cadaveric specimens, operative techniques, and partial RCT were excluded, as were duplicates and nonrelevant studies. Non–English language articles were excluded if direct translation was not possible. Study selection was determined by examining the title and/or abstract of all articles obtained from the database search. Full texts of these potentially relevant studies were then obtained and analyzed for eligibility. Additionally, all references within relevant studies were cross-referenced for inclusion if missed by the initial search. In cases of duplicate patient populations reported in separate studies, only the study with longer duration follow-up was retained for inclusion.
Data Extraction and Analysis
Data were collected and recorded in a Microsoft Excel spreadsheet (version 2016). Data regarding study design, level of evidence, sample size, patient age, patient sex, follow-up, indications, surgical techniques, rehabilitation protocols, and concomitant procedures performed were recorded. Studies were assessed for multiple outcomes of interest including the Constant score, Oxford Shoulder Score (OSS), University of California Los Angeles Shoulder Score, American Shoulder and Elbow Surgeons (ASES) score, and the abbreviated Disabilities of the Arm, Shoulder and Hand (QuickDASH) score, as well as complications, reoperation rates, and patient satisfaction.
Quality Assessment
The MINORS (Methodological Index for Non-Randomized Studies) checklist was used to assess the methodologic quality of included studies. 50 The checklist assigns a score of 0-2 for 8 items applicable to nonrandomized studies, with a maximum score of 16 indicating the highest possible score for an individual nonrandomized study. The items were scored 0 if not reported; 1 when reported but inadequate; and 2 when reported and adequate. A minimum of 18 months was deemed an appropriate length of follow-up, as the balloon spacer is believed to completely biodegrade within 12 months of insertion, thus allowing for assessment of primary endpoints and possible adverse effects. After thorough review of MINORS scoring guidelines, 2 authors (R.K.S., L.K.) independently reviewed and scored each included study. Any disagreements in scoring were resolved by consensus discussion with the senior author (B.R.W.). The κ (Fleiss) statistic was calculated to determine interrater reliability.
Statistical Analysis
Statistical analysis was used to describe data and assess outcomes. Descriptive statistics were calculated from each included study, with continuous variables reported as means ± SDs and categorical variables reported as frequencies with percentages. As all of the studies included in this review were nonrandomized and they exhibited significant variation in study design, patient selection criteria, and concomitant procedures performed, pooling of outcome measures was not performed. Instead, the results are presented in narrative summary fashion.
Results
Included Studies
The initial database search identified 1455 total studies. After application of the systematic screening strategy (Figure 1), a total of 12 clinical studies were included in this review. Of these, 9 (69%) reported no conflicts of interest. The systematic screening approach did not yield any non–English language studies that directly examined clinical results in patients treated with the subacromial balloon spacer; thus, none were excluded solely on the basis of inaccessible direct translation into English.
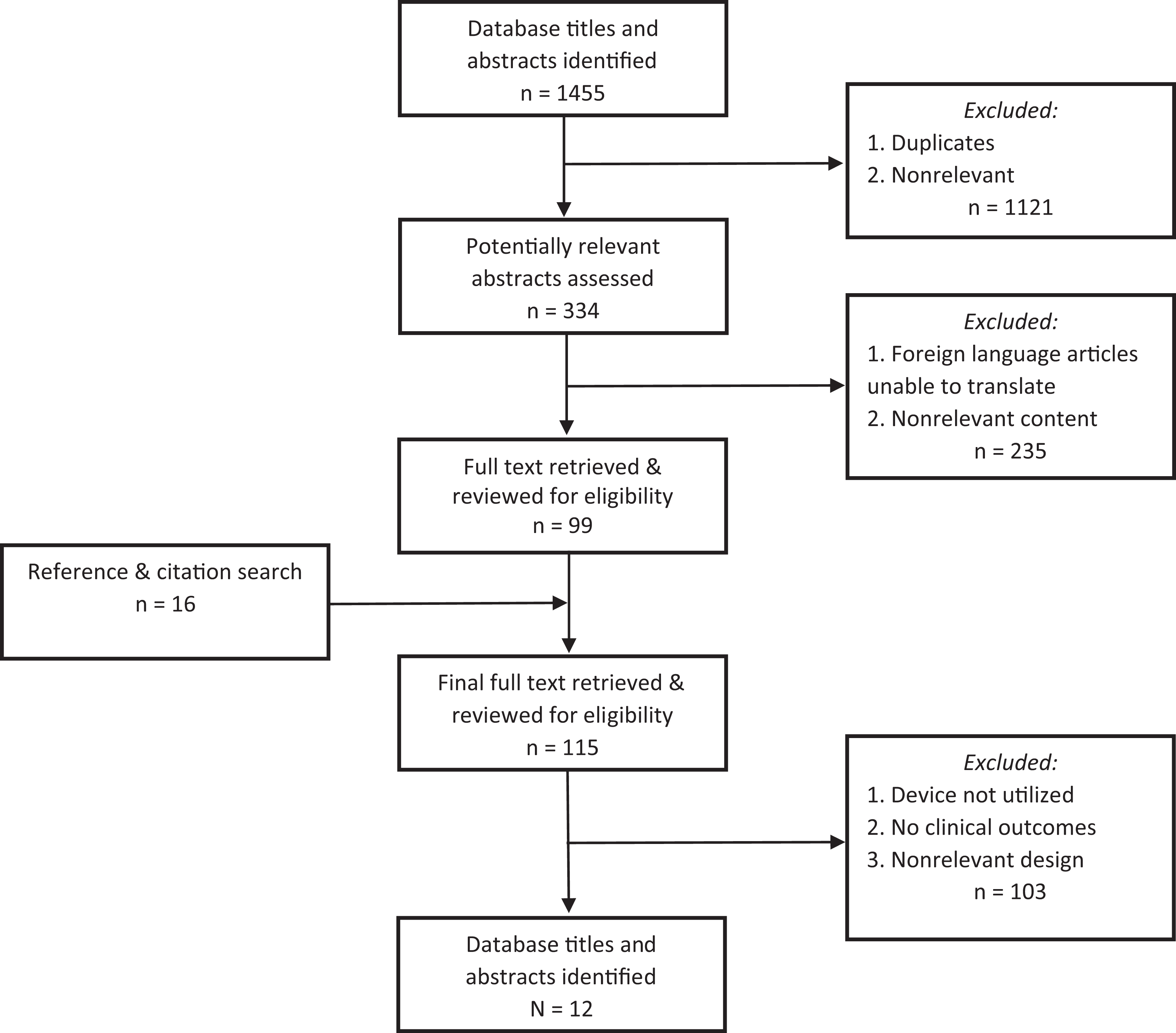
Literature selection process algorithm.
Study Quality
The included studies were composed of 10 case series, 1 prospective cohort study, and 1 retrospective cohort study. All studies were nonrandomized. The median MINORS score was 9.5 of 16 (Table 1). None of the studies used unbiased endpoint assessment, and no study prospectively calculated the target study size. Although 8 of the 12 studies (67%) had an appropriate mean follow-up time, only 3 of the 12 studies (25%) had a loss to follow-up rate less than 5%. In 2 studies, loss to follow-up rate could not be determined, as only patients with a minimum follow-up of 1 year were included. Substantial interrater reliability was noted in scoring between reviewers, with κ = 0.66.
Study Demographic Characteristics and Design a

a Dash (—) indicates that value was not reported. LOE, level of evidence; MINORS, Methodological Index for Non-Randomized Studies.
Patient Characteristics
The 12 studies reporting clinical outcomes, all with level 3 or 4 evidence, were included for data extraction and analysis. In total, 291 shoulders (in 284 patients) undergoing subacromial balloon spacer implantation were studied from all included articles. Patient age and sex were reported for 242 patients (85.2%; 11 studies), yielding a mean age of 68.5 ± 4.2 years (range, 62.4-76.2 years) and slight male predominance (51.2%). The mean duration of follow-up was 22.9 ± 14.9 months (range, 6-60 months) (Table 1).
Surgical Methods and Rehabilitation Protocol
In all studies, devices were implanted with the patient in either the beach-chair or lateral decubitus position. Standard arthroscopic portals were used for device deployment, with the exception of 1 case that was converted to a mini-open approach. 46 Device deployment time was assessed in 2 studies, each with a mean time of 10 minutes (range, 2-30 minutes). One additional study reported a mean operation time (including diagnostic arthroscopy and concomitant procedures) of 33 minutes. 3 Preoperative tear characteristics and concomitant procedures are reported in Table 2. Within the included studies, the use of a shoulder sling ranged from 1 to 3 weeks postoperatively. Active range of motion exercises were initiated between postoperative day 1 and postoperative week 3.
Study Features and Surgical Methods a
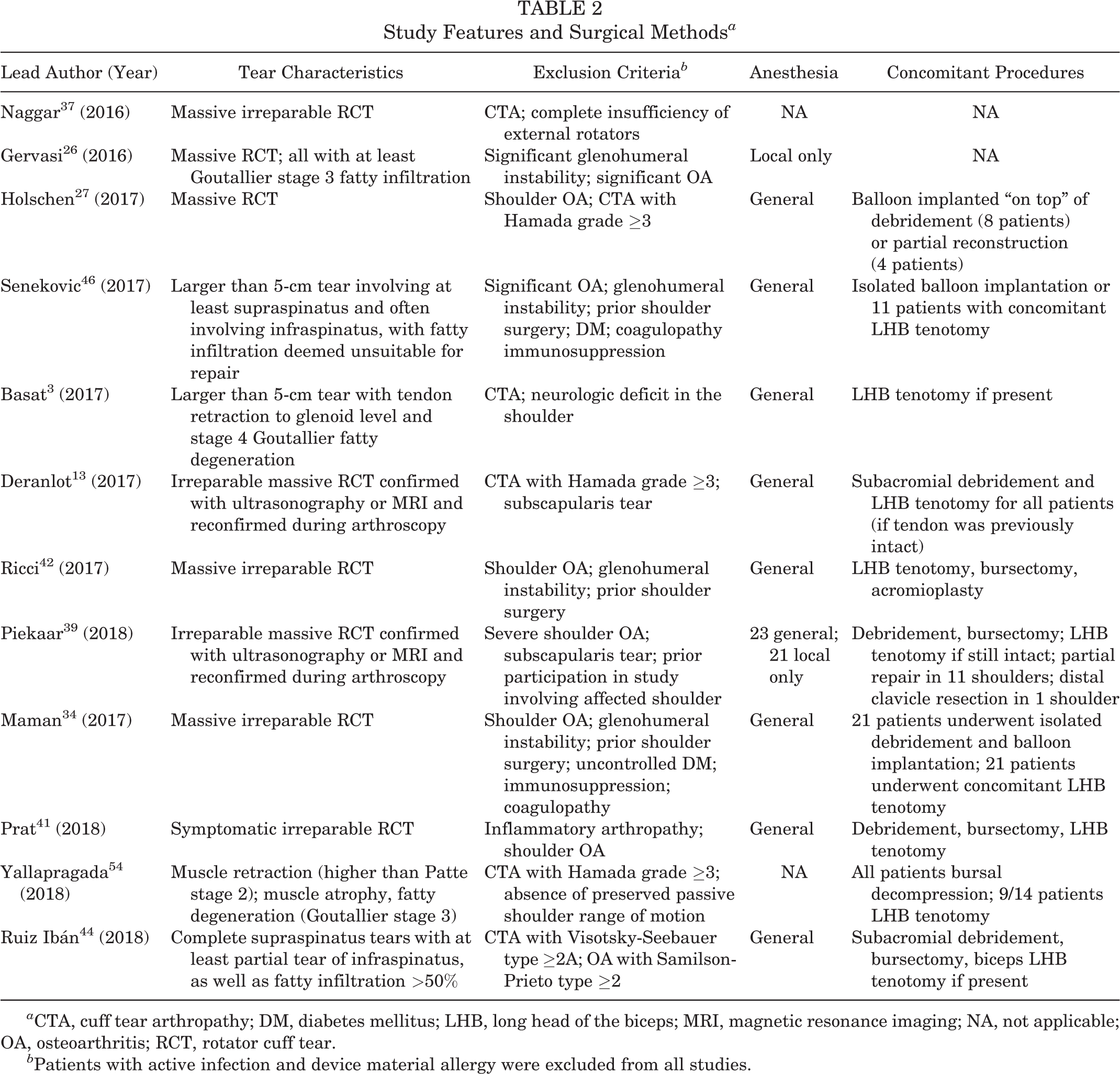
a CTA, cuff tear arthropathy; DM, diabetes mellitus; LHB, long head of the biceps; MRI, magnetic resonance imaging; NA, not applicable; OA, osteoarthritis; RCT, rotator cuff tear.
b Patients with active infection and device material allergy were excluded from all studies.
Patient Outcome Measures
Several functional outcome measures were reported in the included studies, with all 12 studies including at least 1 of the following: Constant score, OSS, ASES score, or short version of the DASH score (QuickDASH) (Table 3). The Constant score was the most frequently used outcome metric (11/12 studies), with improvements in mean Constant score ranging from 18.5 to 49.6 points.
Outcome Measures After Intervention a
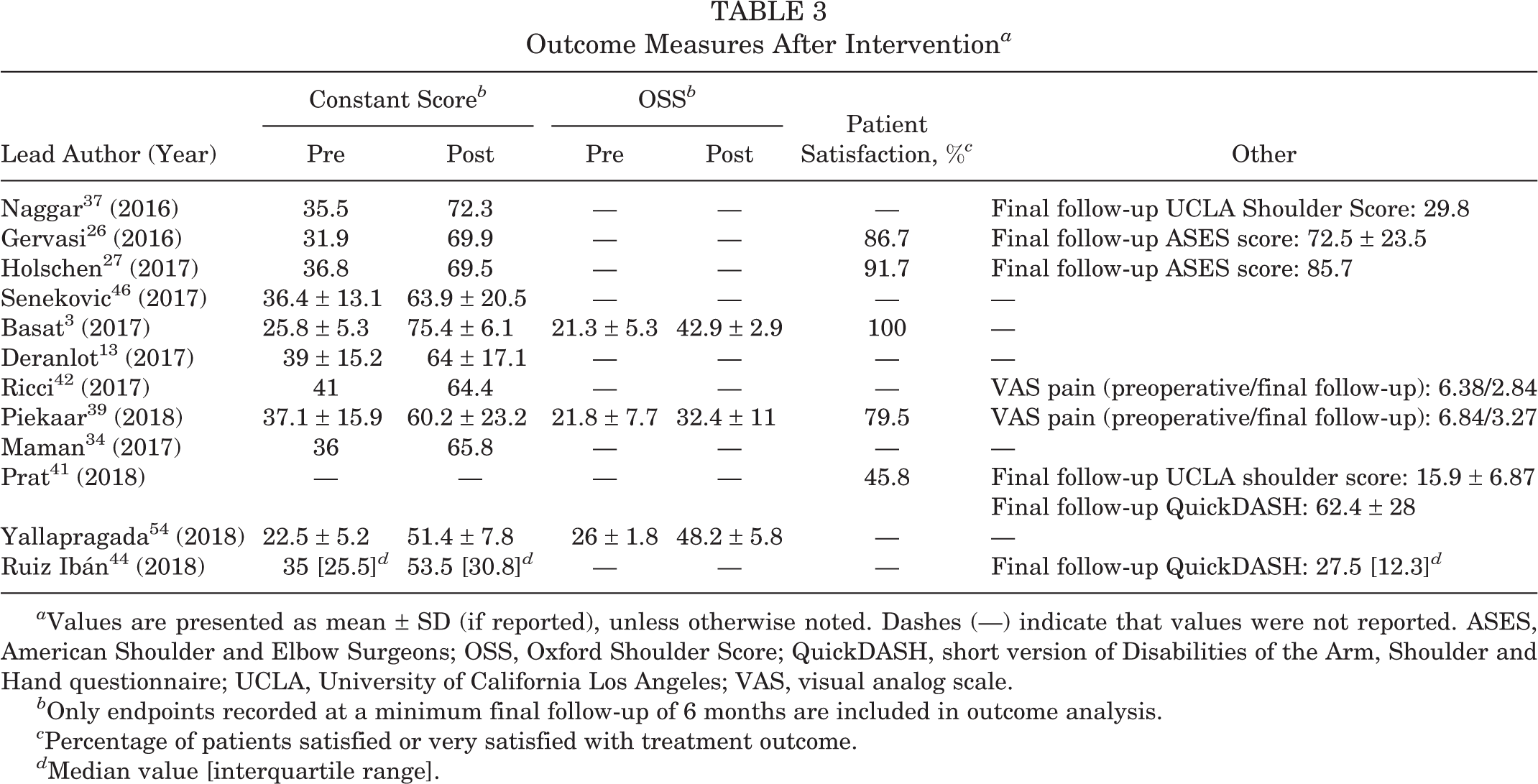
a Values are presented as mean ± SD (if reported), unless otherwise noted. Dashes (—) indicate that values were not reported. ASES, American Shoulder and Elbow Surgeons; OSS, Oxford Shoulder Score; QuickDASH, short version of Disabilities of the Arm, Shoulder and Hand questionnaire; UCLA, University of California Los Angeles; VAS, visual analog scale.
b Only endpoints recorded at a minimum final follow-up of 6 months are included in outcome analysis.
c Percentage of patients satisfied or very satisfied with treatment outcome.
d Median value [interquartile range].
Constant subscale scores were reported in 5 studies (145 shoulders). Improvements in pain, activities of daily living (ADL), range of motion, and strength (Figure 2) were observed in all 5 studies. In 2 studies, 34,42 strength was not statistically significant. In 1 study, 37 statistical significance was not reported for any subscale value. Among studies that assessed patient satisfaction (5 studies; 105 patients), rates of patients indicating they were satisfied or very satisfied with their treatment outcome ranged from 45.8% to 100%.
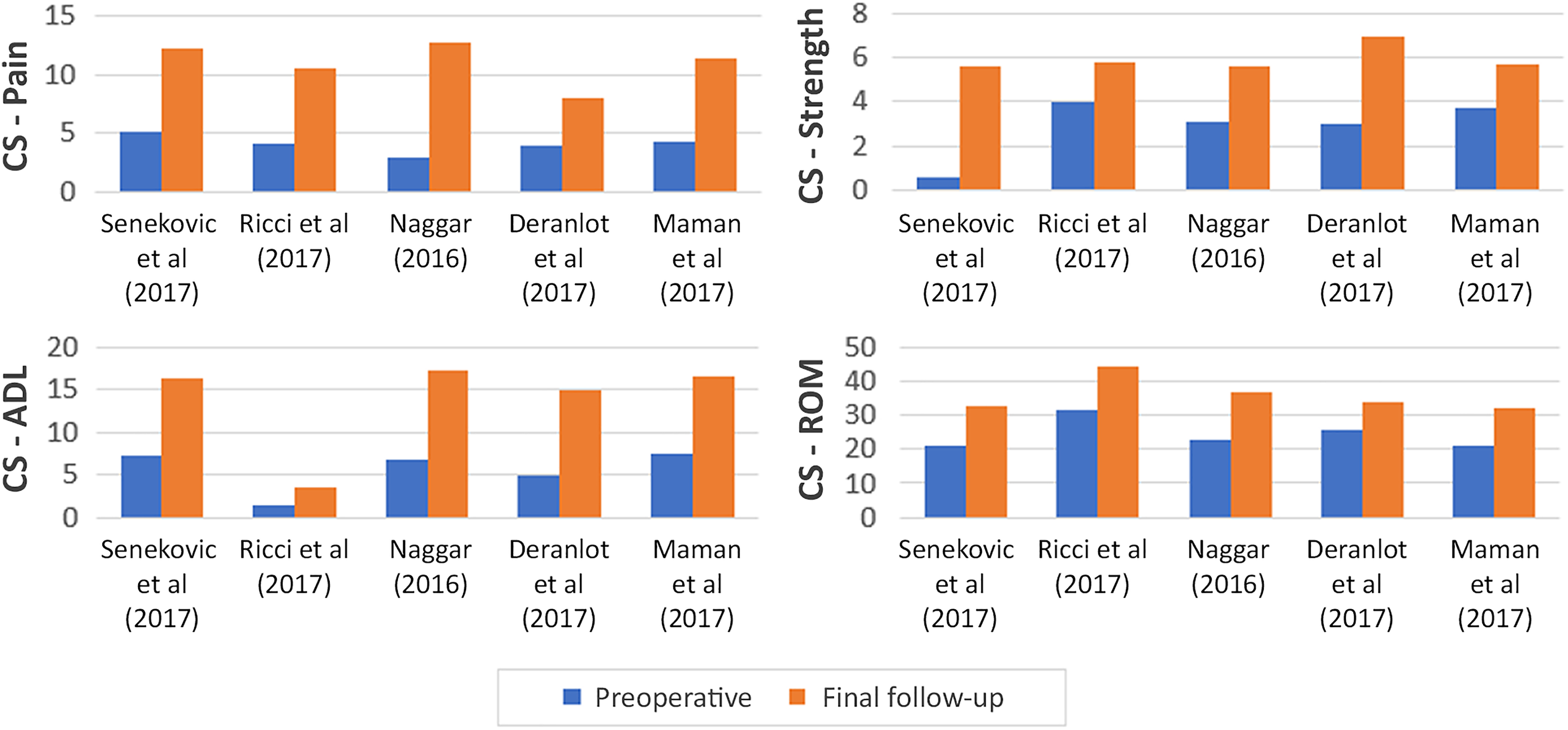
Comparison of Constant (CS) subscale scores, preoperatively to final follow-up. ADL, activities of daily living; ROM, range of motion.
We found that 5 studies assessed changes in active shoulder range of motion after subacromial balloon spacer implantation (104 patients). As well, 4 studies observed increases in active abduction; however, results from 1 of these studies were not statistically significant. 44 Of the 3 studies that reported active anterior elevation, all observed increases; however, only the results from 1 study were statistically significant (P = .00000001). 54 Similarly, although all 4 studies that assessed external rotation observed increased measurements at final follow-up, results from only 2 of these studies 13,54 were statistically significant (P = .001 and .0001, respectively).
Complications occurred in 2.1% of patients. These complications included transient neurapraxia of the lateral antebrachial cutaneous nerve in 1 patient, superficial wound infection in 1 patient, deep wound infection in 1 patient, and balloon migration in 3 patients. Of these, 3 patients (2 balloon migration, 1 deep wound infection) required subsequent surgeries for balloon removal. Balloon position was noted in 2 of the 3 cases of balloon migration, with both reporting anterior migration.
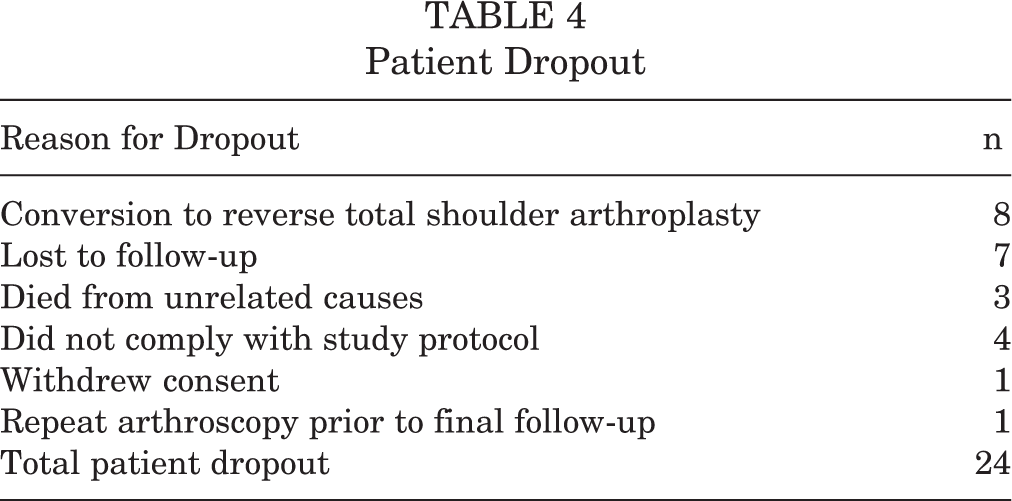
The included studies cited a dropout rate of 24 total patients. The reasons for patient dropout are shown in Table 4. Of note, 2 studies 13,54 included results only for patients who met an inclusion criterion of minimum 1-year follow-up. The number of patients lost to follow-up prior to 1 year postoperatively was not reported.
Patient Dropout
Discussion
We found considerable heterogeneity among the studies reviewed, with significant variations in study design, patient selection criteria, and concomitant procedures performed (Tables 2 and 3). Results from the currently available literature showed generally positive clinical outcomes, with balloon implantation demonstrating effectiveness in pain reduction and functional improvement for patients with massive and irreparable RCTs (Table 3). All 11 studies that used the Constant score as an outcome metric reported improvements in the mean score after balloon implantation (range of mean improvement, 18.5-49.6 points). Across the included studies, device implantation appeared to be a safe and reproducible procedure with a relatively low risk of complication (2.1%). RTSA was the most commonly cited salvage procedure, with 7 patients successfully converted to RTSA by final follow-up. This finding indicates that when balloon implantation fails, RTSA is a viable salvage option. Although the observed merits of this procedure are promising, the limitations of the included studies must be considered when these findings are applied to clinical practice.
Management of massive and irreparable RCTs remains a considerable challenge; a variety of treatments are offered, but we have no clear gold standard or definitive guidelines for optimal management. Proper patient selection is especially important in regard to treatment algorithms. 25,38 Compared with other, less complex treatment options such as arthroscopic debridement, subacromial decompression, and biceps tenotomy, the subacromial balloon spacer has demonstrated promising results on limited follow-up. Boileau et al 6 reported the results of 72 shoulders with massive and irreparable RCTs treated with either isolated biceps tenotomy or tenodesis. At mean final follow-up of 35 months, mean Constant scores increased by a mean of 20.2 points from 46.3 to 66.5 with no significant difference between groups. König and Braunstein 31 reported outcomes for 57 massive RCTs treated with arthroscopic debridement at short-term (2-4 years), midterm (5-6 years), and long-term (8-10 years) follow-up. Although outcomes showed substantial improvement in the short term (mean Constant score, 70.0), scores decreased to 51.0 and 42.3 at mid- and long-term follow-up, respectively. This trend demonstrates the limitations of many current options for palliative management of irreparable RCTs.
When compared with other minimally invasive, palliative treatment techniques such as subacromial decompression, biceps tenotomy or tenodesis, acromioplasty, and tuberoplasty, an important point of distinction with the subacromial balloon spacer system is that it more directly seeks to improve impaired shoulder biomechanics. Singh 48 performed a biomechanical study in 8 matched pairs of cadaveric shoulders to assess superior humeral head migration and functional abduction forces before and after implantation of a subacromial balloon. Initial testing of the intact shoulder state was performed to establish a baseline for each specimen. A 5-cm posterosuperior full-thickness tear involving both the supraspinatus and infraspinatus tendons was then created, and specimens were retested in the “torn state.” As expected, shoulders in the torn state experienced a significant decrease in functional abduction force (P = .009). When specimens were tested after balloon insertion, functional abduction forces were restored to values comparable with the intact state. Compared with the torn state, the subacromial balloon spacer was able to significantly lower the humeral head by a mean of 2.8 ± 1.9 mm at all abduction angles and deltoid loads (P = .006).
Other, less complex procedures, such as tuberoplasty and acromioplasty, have sought to improve shoulder biomechanics through reduced friction at the narrowed acromiohumeral interval. 20,32 However, recent studies have reported that these techniques may actually increase anterosuperior humeral translation and may thus contribute to shoulder instability in some patients. 9,51 For this reason, simple debridement and biceps treatments have largely supplanted acromioplasty and tuberoplasty when a minimally invasive, palliative technique is preferred; however, these techniques are primarily palliative, with little expected biomechanical benefit. Conversely, the balloon may offer an option of similar invasiveness and complexity that more directly seeks to improve impaired shoulder biomechanics in the patient with rotator cuff deficiency.
Although the subacromial balloon seeks to improve shoulder biomechanics, it does not directly correct the underlying problem—the rupture of rotator cuff tendons. As such, functional improvements cannot be expected to match outcomes after rotator cuff repair, reconstructive techniques, or shoulder arthroplasty. RTSA has been increasingly used for irreparable RCTs with and without glenohumeral osteoarthritis. Ernstbrunner et al 17 performed a systematic review that included 8 studies with a minimum of level 4 evidence and at least 5 years of follow-up to examine long-term results of RTSA for rotator cuff dysfunction. Mean absolute Constant scores improved significantly from a mean score of 24 preoperatively to a mean score of 59 at last follow-up, with significant improvements in active anterior elevation and abduction and no deterioration in active range of motion up to 20 years after the operation. Although similar superior outcomes after RTSA have been well-documented, 7,11,36,49 it is an extensive and technically complex surgery associated with relatively high complication rates and limited salvage options, especially among younger patients with high-demand jobs and activities. 2,36,56 Favard et al 19 reported a complication rate of 18%, and Ernstbrunner et al 18 found complication rates as high as 39% in patient cohorts younger than 60 years.
Latissimus dorsi transfer is a treatment option for irreparable RCTs among relatively young patients without glenohumeral osteoarthritis or subscapularis insufficiency. El-Azab et al 16 reported outcomes of 115 shoulders with irreparable RCT treated with latissimus dorsi transfer after a mean follow-up of 9.3 years. The authors found significant improvements in pain level and shoulder function, including a sustained increase in Constant score from 44 preoperatively to 71 at final follow-up. However, the procedure requires a long rehabilitation process, and high rates of tendon rupture have been reported in the literature. 29 Superior capsular reconstruction is a relatively new technique for treatment of irreparable RCTs. Although its indications continue to evolve, early outcomes are promising on short-term follow-up. As with RTSA and latissimus dorsi transfer however, superior capsular reconstruction is a technically challenging procedure that requires prolonged surgical time and a substantial rehabilitation period. 21
Beyond a shared indication of massive to irreparable RCT, the included studies exhibited variability in patient selection that likely affected reported outcomes. We found that 2 studies in this review included a total of 6 shoulders with preoperative pseudoparalysis. 27,44 Postoperative shoulder function remained poor in all cases, with 2 shoulders converted to RTSA by final follow-up. Variable definitions for pseudoparalysis have been reported in the literature, further complicating management decisions in these cases. 53 Because of poor early outcomes and limited data, balloon spacer implantation in shoulders with pseudoparalytic characteristics cannot be recommended at this time.
The included studies also demonstrated variability in patient selection regarding the role of partial repair in shoulders treated with the balloon spacer system. We found that 2 studies in this review included a total of 15 patients in whom some degree of partial repair was performed in addition to balloon spacer insertion. Subgroup analysis in one of the studies showed no statistical difference between outcomes of shoulders treated with balloon insertion alone compared with those treated with concomitant partial repair. 39 The other study did not report subgroup analysis. 27 Limited reporting and sample sizes in these studies restrict the utility of the findings, but several studies have shown that partial repair of a massive RCT is generally recommended if some degree of repair is possible. 5,23,28,30,38,40 The implantation of the balloon laid on top of a partial repair may protect fragile repairs from subacromial friction while supporting the forces exerted by the partially repaired tendon 8,52 ; however, further studies are needed to fully investigate this potential benefit.
Prior studies have demonstrated that patients with medical comorbidities tend to experience less improvement after both surgical and nonsurgical treatment. 22 Furthermore, in patients treated with primary rotator cuff repair, rates of retear are higher in patients older than 50 years. In a retrospective cohort study of 1600 patients who had undergone arthroscopic rotator cuff repair, Diebold et al 14 found retear rates of 10% in patients aged 50 to 59 years, 15% in those aged 60 to 69 years, 25% in those aged 70 to 79 years, and 34% in those 80 years or older. These rates are substantially higher than the 5% retear rate that was seen in patients younger than 50 years. In addition to age, several patient factors such as diabetes, chronic vascular disease, and smoking have potential to alter the wound-healing process. 55 This topic has generated great interest in biological augmentation with platelet concentrates and cell-based therapies after rotator cuff repair; however, little evidence is available to show that these strategies can help in the setting of the muscle atrophy, retraction, and fatty infiltration that are characteristic of chronic RCTs. For patients with massive to irreparable RCT and poor wound healing parameters, the balloon spacer may be a beneficial therapy that does not rely heavily on tissue healing processes.
This systematic review revealed that the beneficial effects of the subacromial balloon system may persist beyond the time of device deflation and degradation, although the exact reason for this effect remains unclear. The balloon copolymers (polylactide and ∊-caprolactone) are believed to biodegrade within 12 months. 10,33,45 Of the studies included in this review, 6 studies reported varying degrees of postoperative radiographic findings. Ricci et al 42 found that balloons progressively decreased in size from 6 to 12 months postoperatively, and the investigators observed complete degradation in all patients assessed by magnetic resonance imaging at final follow-up of 24 months. Using ultrasonography, Senekovic et al 46 found that 86% of balloons were inflated at 3-month follow-up, but the devices were detectable in only 54.5% of patients at 6 months. Despite this, shoulder function and pain level demonstrated continued improvement in the majority of patients. In another included study, repeat arthroscopy was performed in 1 patient at 7 months and showed near-complete balloon degradation. 39 Holschen et al 27 reported results for 1 patient examined by magnetic resonance imaging after 12 months. Findings showed remnants of the deflated balloon with formation of scar tissue in the subacromial space. Although continued improvement in outcomes appears to be consistent across studies, further investigations are needed to fully elucidate the mechanisms underlying these findings.
Limitations
As with any systematic review, this study has several potential limitations that warrant discussion. The primary limitation relates to the methodological quality of the included clinical studies, all of which were nonrandomized observational studies. The lack of randomization and control groups, as well as variations in exclusion criteria, could have introduced selection bias into the results. Additionally, because of the heterogeneity of the reported data, included studies varied in the degree of detail offered regarding presurgical RCT characteristics, concomitant procedures performed, and outcome measures reported. Therefore, the potential for confounding cannot be discounted. Furthermore, most of the included articles were of level 4 evidence, with modest sample size and limited follow-up.
Conclusion
This systematic review of the existing literature suggests that subacromial balloon spacer placement is a minimally invasive, technically simple procedure with low rates of perioperative complications and favorable patient-reported outcomes at limited short-term follow-up. This emerging technology may be most appropriately indicated for lower demand patients with or without advanced medical comorbidities in whom complete repair or more prolonged rehabilitation is not feasible. The inherent methodological limitations and patient heterogeneity between the studies we reviewed may have impaired our ability to fully characterize the longer term efficacy, particularly relative to other potential surgical options. Further prospective randomized or comparative studies are warranted to ascertain clinical outcomes of subacromial balloon spacer in the management of massive and irreparable RCTs.
Footnotes
One or more of the authors has declared the following potential conflict of interest or source of funding: S.A.P. has received research support from Exactech, consultant fees from Arthrex and Exactech, and hospitality payments from Smith & Nephew and Stryker. B.R.G. has received research support from Southtech Orthopedics, educational support and consultant fees from Mitek, and hospitality payments from Exactech. N.N.V. has received research support from Arthrex, Breg, Ossur, Smith & Nephew, and Wright Medical; consultant fees from Medacta, Minivasive, and Orthospace; royalties from Smith & Nephew and Vindico Medical-Orthopedics Hyperguide; educational support from Medwest; and nonconsulting fees from Pacira Pharmaceuticals and has stock/stock options in Cymedica, Minivasive, and Omeros. B.R.W. has received research support from Arthrex, royalties from Elsevier, speaking fees from Genzyme, educational support from Smith & Nephew, and hospitality payments from Medical Device Business Systems. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
