Abstract
Background:
The results of arthroscopic anterior labral repair have demonstrated high failure rates in patients with significant glenoid bone loss. Several reconstruction procedures using a bone graft have been developed to overcome bone loss.
Purpose:
The primary objective of this study was to generate a safety profile for arthroscopic anatomic glenoid reconstruction using a distal tibial allograft. The secondary objective was to evaluate the radiological outcomes of patients who underwent this procedure.
Study Design:
Case series; Level of evidence, 4.
Methods:
This retrospective review included the medical charts and diagnostic images of 42 consecutive patients who underwent arthroscopic shoulder stabilization by means of capsule-labral reattachment and bony augmentation with a distal tibial allograft. The safety profile was measured by detecting intraoperative or postoperative complications, including neurovascular (nerves and blood vessels) injuries, bleeding, infections, and dislocations. A radiological evaluation was conducted by assessing computed tomography (CT) scans obtained preoperatively and at approximately 6 months postoperatively.
Results:
A total of 42 patients (29 male, 13 female) with a mean age of 26.73 ± 9.01 years were included. An excellent safety profile was observed, with no intraoperative complications, neurovascular injuries, adverse events, bleeding, or infections. CT bone scans were obtained for 31 patients, and the mean follow-up for CT scanning (to measure resorption and union) was 6.31 ± 1.20 months (range, 6-7.5 months). There were no cases of nonunion or partial union. Thirteen patients (42%) had no resorption, whereas 13 (42%) and 5 (16%) patents had <50% and ≥50% resorption, respectively.
Conclusion:
Arthroscopic shoulder stabilization with distal tibial allograft reconstruction is a safe operative procedure with a minimal risk to neurovascular structures. Most patients had a healed allograft, but 16% of patients had ≥50% resorption on CT at 6 months. Studies with a longer follow-up are recommended for better assessment of the safety profile.
Glenohumeral instability encompasses various soft tissue and osseous injuries. The most common soft tissue injury is to the capsular ligament. Labral damage (Bankart lesion and variants) is present in nearly all cases of recurrent anterior shoulder instability 24 and is commonly treated with arthroscopic repair. 16
Burkhart and De Beer 4 reported a 67% recurrence rate of redislocations or subluxations in patients with bone deficits (either inverted-pear glenoid or Hill-Sachs lesion of the humeral head) after arthroscopic Bankart repair, which is significantly higher compared with patients without bone deficits (4%). Moreover, patients with risk factors for failure, such as young age, hyperlaxity, competitive contact sport participation, and in particular, glenoid loss and/or humeral bone defects, have been shown to have significantly higher failure rates after Bankart repair. 1,3,4,6,8,15,18 Several glenoid reconstruction procedures using a bone graft have been described for patients with anterior glenoid defects or other risk factors for shoulder dislocation recurrence, including autogenous coracoid transfer to the anterior glenoid (Latarjet procedure) 19 as well as iliac crest autografts 22 and distal tibial allografts.2
In recent years, trends toward minimally invasive shoulder surgery and improvements in technology and surgical techniques have led surgeons to expand the application of arthroscopic treatment in anatomic glenoid reconstruction. As in other joints, arthroscopic procedures offer improved cosmetic results and postoperative advantages including less pain, earlier mobility, and faster rehabilitation and return to sports. 13 Arthroscopic techniques have been developed to treat severe instability with or without associated bone loss using a coracoid autograft (arthroscopic Latarjet), 13,14 autologous iliac crest bone graft, 12,25 and distal tibial allograft. 23
Wong and Urquhart 23 developed and published an all-arthroscopic anatomic glenoid technique using a distal tibial allograft, which avoids damaging the subscapularis muscle and allows for repair of the soft tissue. The technique is based on arthroscopic Bankart repair performed in a lateral decubitus position. It requires only one additional medial portal that is created from an inside-out technique to avoid injuring neurovascular structures. Because it is an inside-out portal created parallel to the glenoid surface, it is reproducible for safe passage of the graft.
The primary objective of this study was to generate a safety profile for arthroscopic anatomic glenoid reconstruction using a distal tibial allograft as described by Wong and Urquhart. 23 The secondary objective was to evaluate the short-term radiological outcomes of patients who underwent this procedure.
Methods
Design
This study was a retrospective analysis of a consecutive series of patients who underwent arthroscopic shoulder stabilization using a distal tibial allograft, performed by the senior author (I.H.W.) between 2012 and 2016. This study was approved by the Nova Scotia Health Authority Research Ethics Board.
Patients
From 2012 to 2016, a total of 184 patients underwent anterior shoulder stabilization, including the arthroscopic Latarjet procedure (n = 65), arthroscopic anatomic glenoid reconstruction (n = 49), and arthroscopic Bankart repair (n = 70), for recurrent anterior shoulder dislocations. The indications for both arthroscopic anatomic glenoid reconstruction and the arthroscopic Latarjet procedure were bone loss or previous failed surgery. However, the Latarjet procedure was only performed when a tibial allograft was not accessible from the bone bank.
Among the 49 patients who underwent arthroscopic anatomic glenoid reconstruction, only 42 were included in this study. Seven patients were excluded based on the exclusion criteria of patients with rotator cuff injuries, those without a preoperative Western Ontario Shoulder Instability Index (WOSI) score, or those lost to follow-up (Figure 1). All the included patients had glenoid bone loss (15%-45%) as diagnosed by en face computed tomography (CT) (with 3-dimensional reconstruction). The participants included both patients undergoing a primary procedure for anterior instability and patients undergoing revision for a failed procedure.

Flowchart of the selection of patients for the study.
Surgical Technique
All procedures were performed arthroscopically with the patient in the lateral decubitus position under general anesthesia (see the online Video Supplement for this technique). The surgical technique and portal placement were performed according to the methods previously described by Wong and Urquhart in 2015. 23 After performing diagnostic arthroscopic surgery, the rotator interval was excised to insert the graft, and the conjoint tendon and coracoacromial ligament were exposed for inside-out creation of a medial portal (referred to as the Halifax portal) (Moga I, Konstantinidis G, Wong I. “Safety of a Far Medial Arthroscopic Portal for Anatomic Glenoid Reconstruction.” Presented at National Association for Canadian Orthopaedic Resident Doctors, 2017). This was created using a switching stick, which was placed through the posterior portal, superior to the subscapularis and lateral to the conjoint tendon and through the deltopectoral interval.
The next step included an arthroscopic measurement of glenoid bone loss and preparation of the tibial bone allograft. The grafts were fresh-frozen and 2 to 3 mm larger than the estimated defect. The usual measurement of the graft was 1 × 1.5 × 2 cm. The graft was inserted by a double-barrel cannula into the shoulder from the Halifax portal and was fixed to the glenoid by using 2 cannulated screws (3.5-mm screw and 1.1-mm guide wire from DePuy Synthes–Mitek) through the Halifax portal. 23 Finally, the capsule and labrum were fixed to the native anterior glenoid using anchors, as performed in Bankart repair (Figure 2).

Surgical technique of arthroscopic anatomic glenoid reconstruction.
Postoperative Rehabilitation
All patients were discharged with a shoulder brace in neutral rotation for 6 weeks. The physical therapy protocol consisted of passive mobilization exercises up to neutral shoulder rotation for the first 2 weeks, gradually increasing to active assisted and active range of motion (ROM) exercises and aiming for full active ROM at 6 to 8 weeks. After achieving full ROM, attention was given to strengthening the surrounding muscles and to scapular control. Return to sports was allowed once bony union was radiologically confirmed with a CT scan (usually 6-9 months after surgery).
Data Collection
Demographics and clinical characteristics of the eligible participants were collected from their medical charts. Demographic variables included age at surgery, sex, and side. Clinical characteristics such as the presence of glenoid bone loss and/or a humeral Hill-Sachs lesion, prior fractures, and surgical history were extracted. Surgical recordings of initial diagnostic arthroscopic surgery were used to confirm the diagnosis taken from a chart review, and the extent of bone loss was arthroscopically measured using a calibrated probe. The surgeons used a bare area to measure glenoid bone loss, following the method mentioned by Burkhart et al. 5
Medical charts of the 42 patients were reviewed retrospectively by the authors to generate a safety profile as the primary outcome of the study. The authors reviewed the charts to assess intraoperative complications such as neurovascular injuries (nerves and blood vessels) and bleeding as well as postoperative adverse events including infections and dislocations.
The secondary outcome measures included postoperative diagnostic imaging to assess union and resorption. Anteroposterior, lateral, and axial radiographs were obtained routinely at baseline, at 2 weeks to rule out fractures and screw malplacement, and again at approximately 1 year to evaluate bony union or resorption of the allograft. CT was also conducted before surgery and at a postoperative time point of roughly 6 months. The rationale for performing CT during the early postoperative period (at around 6 months) was to assess bone healing before return to sports activities. Postoperative CT scans of all participants were assessed to evaluate bone graft location (inferior, middle, or superior third of the anterior glenoid face); medial, flush, or lateral step-off of the graft in relation to the native glenoid surface; bony union; or graft resorption. CT scans were also used to measure the preoperative and postoperative sagittal dimensions of the glenoid as well as to confirm radiographic findings of bony union and resorption.
Statistical Analysis
Statistical analysis of the data was carried out using Stata software (version 14; StataCorp). Frequencies and percentages were reported for categorical variables, and means with SDs were reported for continuous variables. Multiple 1-sided and 2-sided and paired and unpaired t tests were conducted, with a significance level of 95% (α = 0.05).
RESULTS
Forty-two patients met the inclusion criteria. Among them, 29 were male, and 13 were female. The mean age of the patients at the time of surgery was 26.73 ± 9.01 years (range, 16-51 years). The mean bone loss was 30.32% ± 7.90% (range, 15%-45%). Twenty-two patients (52.4%) underwent surgery for recurrent shoulder instability due to glenoid bone loss, whereas 20 patients (47.6%) had bone loss with previous failed surgery (Table 1).
Demographics and Clinical Characteristics of the Study Population (N = 42)

Safety Profile
In this case series, there were no intraoperative complications, neurovascular injuries, adverse events, readmissions to the hospital, major bleeding, postoperative infections, or implant failure.
Radiographic Outcomes
Thirty-one patients (73.8%) underwent postoperative CT, with a mean of 6.31 months (range, 6-7.5 months) between the surgical intervention and CT scanning. CT was performed to assess bone healing and the progress of rehabilitation for ensuring early return to sports activities. There was no incidence of nonunion or partial union. The mean sagittal dimension of the glenoid (measured by preoperative CT) was 24.03 ± 2.96 mm. The mean width of the articular surface of the graft was 10.11 ± 4.03 mm (Figure 3 and Table 2).

(A) Preoperative 3-dimensional (3D) computed tomography (CT) scan showing the loss of anterior glenoid bone from recurrent dislocations. (B) Postoperative 3D CT scan showing reconstituted, healed distal tibial allograft bone held with 2 cannulated screws. (C) Preoperative axial CT scan at the inferior third of the glenoid compared with (D) postoperative axial CT scan at the same level showing healed allograft bone with screw fixation.
Radiographic Findings From Preoperative and Postoperative CT a

CT, computed tomography.
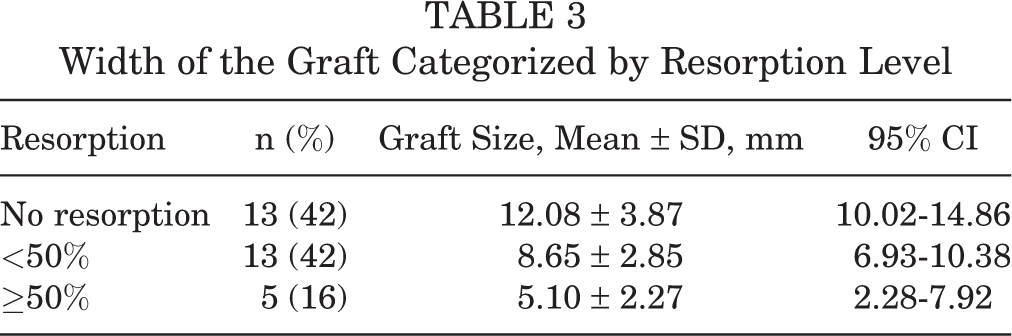
No resorption was seen in 13 patients (42%); however, 13 (42%) and 5 (16%) patients had <50% and ≥50% resorption of the initial volume of the allograft, respectively. Although 5 patients (16%) in our study had ≥50% resorption, the sagittal dimension of the remaining allograft postoperatively was 5.10 ± 2.27 mm, indicating that there was still a bone graft present and that there was no complete resorption (Table 3).
Width of the Graft Categorized by Resorption Level
Discussion
This study presents the safety profile and short-term radiological results of the arthroscopic anatomic glenoid reconstruction technique without subscapular split for the treatment of anterior shoulder instability using a distal tibial allograft. The study population had a mean glenoid bone loss of more than 30%, with a high risk of Bankart repair failure and recurrence of shoulder instability. Considering this population, the distal tibial allograft technique showed good short-term results in terms of the study’s primary outcome, as no safety concerns arose during or after surgery. This includes no intraoperative or postoperative complications. There were no neurovascular injuries, admissions to the hospital, major bleeding, postoperative infections, or implant failures. In addition, there were no dislocations at follow-up, but 1 patient had a positive shoulder apprehension test result on clinical examination. However, it is too early to comment on clinical safety based on 16-month follow-up results, as postoperative instability mostly presents at around 2 years after the Latarjet procedure, as mentioned by several studies. 7,20
Shah et al 21 recorded 5 cases (10%) that resulted in a neurological injury after the Latarjet procedure, including 2 axillary nerve injuries, 2 musculocutaneous nerve injuries, and 1 radial nerve injury. Delaney et al 7 demonstrated that 7 (21%) of 34 patients presented a clinically detectable nerve deficit postoperatively. Lafosse and Boyle 13 described a 12.5% rate of postoperative complications, including hematoma, graft fractures, and nerve injuries. In comparison to these studies, our study results indicate an excellent safety profile for arthroscopic anatomic glenoid reconstruction using a distal tibial allograft. 13
The technique seems to reduce injuries to the musculocutaneous and axillary nerves because the conjoint and subscapularis tendons remain intact. The establishment of the Halifax portal by an inside-out technique allows the identification of the above-mentioned 2 structures, which protects the neurovascular structures from injuries. In this technique, the Halifax portal has been found to be, on average, 4 cm away from any neurovascular structures because the subscapularis muscle and conjoint tendon protect the musculocutaneous and axillary nerves (Moga I, Konstantinidis G, Wong I. “Safety of a Far Medial Arthroscopic Portal for Anatomic Glenoid Reconstruction.” Presented at National Association for Canadian Orthopaedic Resident Doctors, 2017).
Allograft resorption has been reported to be a problem for bone graft procedures, as presented by several studies. Iannotti and Frangiamore 11 reported on large structural allografts in which 6 (31%) of 19 patients experienced resorption. Likewise, Phipatanakul and Norris 17 described 20 patients who underwent glenoid bone allografting in which 10 patients (50%) reported having graft resorption. A retrospective review of 11 patients who underwent bone grafting because of severe glenoid defects mentioned graft resorption in 100% of patients. 20 Hoffelner et al 10 described 11 patients who underwent revision shoulder arthroplasty with bone grafting, with all patients (100%) experiencing bone resorption.
In our study, 18 patients (58%) presented with either <50% or ≥50% resorption. However, none of these patients with documented bone resorption developed shoulder instability. Most importantly, even in the case of ≥50% resorption, there was an increase in the sagittal diameter of the glenoid by a mean of 5.10 mm that was proven to be enough to offer clinical stability of the shoulder. This may be because we had put in a graft that was larger than its physiological size. There was physiological resorption, but this can be explained by the Wolff law, where the graft was remodeled into its native bony anatomy to help support its structure in the glenoid. 9 However, CT at a longer follow-up period would be useful to assess if resorption continues.
The arthroscopic anatomic glenoid reconstruction technique reported in this study contributes to the safety profile of this surgical procedure and demonstrates several advantages. Perhaps the described surgical technique does not require repositioning of the conjoint tendon or splitting of the subscapularis muscle. Therefore, it is more anatomic in the sense of conserving the original anatomy and biomechanics of the shoulder. Furthermore, with this technique, the axillary and musculocutaneous nerves are protected by preserving the native conjoint and subscapularis muscles throughout the procedure. Moreover, using an allograft obviates donor site morbidity, although there are small risks of disease transmission and the potential for rejection of the allograft in patients. In addition, there is the potential added benefit of restoring the articular surface of the glenoid and providing an anatomic fit by customizing the graft size for the defect. Using an iliac crest or coracoid autograft, both offer bony reconstruction options of the glenoid, although they lack the ability to conform to the glenoid and are limited by the amount of bone available for grafting. Last but not least, this procedure is likely to reproduce the normal anatomy of the shoulder compared with a nonanatomic reconstruction such as the Latarjet procedure. 2,15 A subscapularis sling was not used in this procedure, as a normal anatomic shoulder does not need any sling.
The strengths of this study include the use of both clinical and radiological evaluations. These allow for the effective measurement of clinical outcomes (WOSI score) and radiological findings at different time points, providing a better understanding and comparison between preoperative and postoperative measurements.
The major limitation of our study is its retrospective nature, which includes retrospective analysis of patients’ charts for assessing intraoperative and postoperative complications. This might be a potential source of errors because of limited information available in the clinic charts. Moreover, we do not have clinical results for follow-up beyond 2 years. This is a major limitation, as the most common complication of an anterior stabilization procedure is recurrent instability, which typically occurs later. Other limitations include nonrandomization and the size of the study population. The size of the study group constitutes a limitation because it makes our analysis vulnerable to bias. We do not have serial CT scans to assess resorption at a longer follow-up period.
Conclusion
The study findings demonstrated a good safety profile for arthroscopic anatomic glenoid reconstruction with a distal tibial allograft for the treatment of anterior shoulder instability due to glenoid bone loss. Although there are some concerns for allograft resorption, this did not seem to affect shoulder stability at short-term follow up. A longer follow-up study is needed for better assessment of the safety profile and outcomes of the patients who undergo this procedure.
Footnotes
Acknowledgment
The authors acknowledge Swagata Ghosh, MBBS, MPH, for his assistance with the statistical analysis and overall formatting.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution.
Ethical approval for this study was obtained from the Nova Scotia Health Authority Research Ethics Board.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
