Abstract
Background:
Disproportionate end-range plantar flexion weakness, decreased passive stiffness, and inability to perform a heel rise on a decline after Achilles tendon repair are thought to reflect increased tendon compliance or tendon lengthening. Since this was first noted, we have performed stronger repairs and avoided stretching into dorsiflexion for the first 12 weeks after surgery.
Hypothesis:
Using stronger repairs and avoiding stretching into dorsiflexion would eliminate end-range plantar flexion weakness and normalize passive stiffness.
Study Design:
Case series; Level of evidence, 4.
Methods:
Achilles repairs with epitendinous augmentation were performed on 18 patients. Plantar flexion torque, dorsiflexion range of motion (ROM), passive joint stiffness, and standing single-legged heel rise on a decline were assessed at 43 ± 24 months after surgery (range, 9 months to 8 years). Maximum isometric plantar flexion torque was measured at 20° and 10° of dorsiflexion, neutral position, and 10° and 20° of plantar flexion. Passive dorsiflexion ROM was measured with a goniometer. Passive joint stiffness was computed from the increase in passive torque from 10° to 20° of dorsiflexion. Tendon thickness was measured by use of digital calipers. Plantar flexion electromyographic (EMG) data were recorded during strength and functional tests. Analysis of variance and chi-square tests were used to assess weakness and function.
Results:
Marked weakness was evident on the involved side at 20° of plantar flexion (deficit, 26% ± 18%; P < .001), with no weakness at 20° of dorsiflexion (deficit, 6% ± 17%; P = .390). Dorsiflexion ROM was decreased 5.5° ± 8° (P = .015), and tendon width was 8 ± 3 mm greater on the involved side (P < .001). Passive joint stiffness was similar between the involved and noninvolved sides. Only 2 of 18 patients could perform a decline heel rise on the involved side compared with 18 of 18 on the noninvolved side (P = .01). No difference in EMG amplitude was found between the involved and noninvolved sides during the strength or heel rise tests.
Conclusion:
The use of stronger repair techniques and attempts to limit tendon elongation by avoiding dorsiflexion stretching did not eliminate weakness in end-range plantar flexion. EMG data confirmed that end-range weakness was not due to neural inhibition. Physiological changes that alter the force transmission capability of the healing tendon may be responsible for this continued impairment. This weakness has implications for high-demand jumping and sprinting after Achilles tendon repair.
Separation of the tendon ends has been shown to increase for the first 12 weeks following Achilles tendon repair surgery. 12,13 Although side-to-side plantar flexion strength deficits and functional impairments have been documented in patients following surgical repair of Achilles tendon ruptures, 2,10,11,12,15,19 the impact of tendon elongation on strength seems to be equivocal. Using radiographic measurements (ie, tantalum bead implantation), both Kangas et al 7 and Heikkinen et al 3 found no correlation between tendon elongation and isokinetic plantar flexion strength deficits at mean follow-ups of 5 and 14 years, respectively. In contrast, Pajala et al, 17 using the same measurement techniques, found a significant correlation between tendon elongation and isokinetic strength deficits at 1-year follow-up. Heikkinen et al 4 reported increasing strength deficits as the ankle moved into end-range plantar flexion (ie, 10°-30° of plantar flexion). Additionally, using magnetic resonance imaging (MRI) to measure tendon elongation, these authors also found that the most significant correlations between tendon elongation and strength deficits occurred at end-range plantar flexion.
Mullaney et al 14 observed weakness in end-range plantar flexion using an isometric strength assessment. These authors also found decreased passive stiffness in dorsiflexion and the inability to perform a heel rise on a decline in patients who had completed rehabilitation and returned to their desired activities. 14 These subtle impairments were thought to reflect increased tendon compliance or anatomic tendon lengthening. Two strategies proposed by Mullaney et al 14 to reduce or prevent tendon lengthening, and thereby possibly avoid weakness in end-range plantar flexion, were to (1) use stronger surgical repair techniques and (2) reduce the stress through the repaired Achilles tendon during rehabilitation. 14
Since weakness in end-range plantar flexion was first noted, we have performed Achilles tendon repairs using epitendinous augmentation, as this modification has been shown to be 68% stronger than a nonaugmented repair, and it prevents gapping at the site of the repair. 8,9 Additionally, previous research has shown that the passive tension in the Achilles tendon with the ankle in 20° of dorsiflexion and the knee extended is very close to the failure strength of some commonly used repair techniques. 16 Therefore, avoiding this excessive force on the repair site during a period when lengthening may naturally occur may reduce elongation. Based on this information, the rehabilitation protocols were also revised to avoid any stretching of the Achilles or gastrocnemius-soleus complex for joint mobilization during the first 12 weeks after surgery. As the origin of the postsurgical strength deficits could include neural inhibition as well as the previously hypothesized mechanical sources, a side-to-side comparison of calf muscle activation would help to provide a clear assessment of the aforementioned changes in treatment.
The purpose of this study was to evaluate the effects of these treatment changes on end-range plantar flexion strength, passive joint stiffness, and neuromuscular activation. It was hypothesized that these treatment changes would reduce end-range plantar flexion weakness and normalize passive stiffness.
Methods
A total of 55 patients who underwent surgery between 2006 and 2014 to repair an acute, full rupture of the Achilles tendon were identified and contacted. Eighteen of these patients (mean ± SD age, 39 ± 9 years; 16 men, 2 women) volunteered to be tested. The mean ± SD follow-up for this group was 43 ± 24 months, with a minimum period of 9 months and a maximum of 8 years. Suture repairs using a 4-strand Krackow technique with No. 2 Ethibond sutures (Ethicon Inc) were performed on all patients. The repaired tendons were then augmented with an epitendinous cross-stitch weave using No. 0 PDS sutures (Ethicon Inc). All patients were nonweightbearing for 6 weeks, at which point partial weightbearing in a controlled ankle motion (CAM) walker boot (Ossur) commenced.
Patients were progressed to full weightbearing in a CAM walker boot with heel lifts and then to walking without a boot by 9 weeks. Active and passive ankle range of motion (ROM) exercises from plantar flexion to neutral position were performed daily from week 1 to provide mobilization of the tendon. However, neither active nor passive stretching of Achilles or gastrocnemius-soleus complex past the initial tendon tension was permitted for the first 12 weeks following surgery. Low-intensity isometrics began at 6 weeks, followed by isotonics at 9 weeks. Jogging and return to sports were milestone-dependent and typically started around 13 to 20 weeks and 20 weeks to 1 year, respectively. To be included in this study, patients had to have completed the rehabilitation protocol with no postoperative complications and have returned to their desired activities.
Postoperative testing consisted of passive plantar flexion and dorsiflexion ROM, Achilles tendon width measurement, calf girth measurement, isometric plantar flexion strength evaluation, passive joint stiffness, toe walking, and standing single-legged heel rise (on an incline, decline, and level surface) on each leg. Electromyographic (EMG) activities of the plantar flexors (medial gastrocnemius, lateral gastrocnemius, and soleus) were recorded during the isometric strength tests and the toe walking and heel rise tests. All patients gave informed consent prior to participation, and the study protocol was approved by an institutional review board.
Range of Motion, Calf Girth, and Tendon Width
Passive plantar flexion and dorsiflexion ROM was measured manually on each leg with a goniometer, with the patient in the prone position and the knee flexed to 90°. Calf girth, in centimeters, was measured with a tape measure placed around the thickest portion of the lower leg (ie, over the muscle bellies of the medial and lateral gastrocnemius). Achilles tendon width, in millimeters, was measured at the level of the medial malleolus by use of calipers.
Isometric Strength Testing and Passive Joint Stiffness
Maximum isometric plantar flexion torque was measured at 20° and 10° of dorsiflexion, neutral position, and 10° and 20° of plantar flexion (Biodex System 2; Biodex Medical Systems). Patient positioning was similar to that described by Mullaney et al 14 : Patients were seated with the knee in full extension and the foot placed in the ankle testing attachment. 8 Patients performed 3 maximal isometric plantar flexion contractions at each angle. Each contraction lasted 5 seconds, with 10 seconds of rest between contractions. Patients were given a 1-minute rest between each test angle. At each test angle, the average of the peak torque generated during each of the 3 contractions was calculated. All torque measures were corrected for the contributions of passive torque due to limb mass and passive muscle tension at each test angle. The percentage strength deficit at each test angle between the involved and noninvolved limbs was determined using the following formula: (Noninvolved – Involved)/Noninvolved. Passive joint stiffness (N·m/deg) was calculated from the increase in passive torque from 10° of dorsiflexion to 20° of dorsiflexion on each limb, replicating the methods and calculations used by Mullaney et al. 14
Functional Tests
As described previously by Mullaney et al, 14 toe walking consisted of walking in a plantar flexed position for 40 feet. If patients could maintain a plantar flexed foot, similar to the noninvolved side, the result was noted as a success. If the heel was noticeably lower on the involved side or touched the floor, the result was noted as a failure. For the neutral single-legged heel rise, the patient was instructed to perform a heel rise while standing on the floor without using any manual support. If the patient was unable to elevate the involved heel from the ground to a similar height to the noninvolved side, then the result was noted as a failure. The same criteria were used for grading incline and decline single-legged heel rises. For the incline heel rise test, the patient stood on an incline board at an angle of 21° (of dorsiflexion) and was instructed to perform a heel rise with the noninvolved lower extremity, followed by the involved lower extremity. For the decline single-legged heel rise test, the patient stood on the same board facing the opposite direction (21° of plantar flexion). The patient was instructed to perform a heel rise from the plantar flexed position.
Electromyography
Surface EMG data were acquired from each leg during all isometric and functional tests (common mode rejection ratio, >110 dB at 50-60 Hz; input impedance, >10 GΩ) (BTS FREEEMG 1000; BTS Bioengineering). After the skin of each patient was shaved, cleaned, and lightly abraded, muscle activity was sampled at 1000 Hz by use of disposable Ag/AgCl passive dual electrodes (2.0-cm interelectrode distance) (Noraxon). Electrodes were placed over the muscle bellies of the medial gastrocnemius, lateral gastrocnemius, and soleus muscles in accordance with the Anatomical Guide for the Electromyographer. 18
EMG data were high-pass filtered at 10 Hz to eliminate motion artifact and then full-wave rectified and smoothed using a root mean square calculation with a window of 100 milliseconds. The peak EMG activation level for each muscle during each repetition of each activity was then identified. The mean activation level, in millivolts, of each muscle from 250 milliseconds before the peak to 250 milliseconds after the peak was calculated and averaged for each test. Since there was no effect of joint angle on the EMG signals recorded during the isometric tests (see Results), the peak activation levels for each of the plantar flexor muscles during the functional tests were normalized to the peak activation level of the corresponding muscle during the isometric tests, regardless of the position at which they occurred.
Statistics
Plantar flexion ROM, dorsiflexion ROM, calf girth, Achilles tendon thickness, and passive joint stiffness were compared between the involved and noninvolved limbs by use of paired t tests. Single-factor, repeated-measures analysis of variance (ANOVA) (angle) was used to assess isometric plantar flexion strength deficits. When an effect of angle was revealed, Bonferroni-corrected paired t tests were used to compare strength differences between the involved and noninvolved limbs at each test angle. Pearson correlation was performed to assess the relationship between the side-to-side differences in dorsiflexion ROM and isometric strength deficits at each ankle position. Additionally, due to the wide range in length of follow-up, Pearson correlation was performed between the length of follow-up and isometric strength deficits at each ankle position. Functional test results were compared between the involved and noninvolved sides using chi-square tests. Additionally, paired t tests were performed to assess differences in length of follow-up between patients who could successfully perform the functional tests and those who could not. EMG data recorded during the isometric testing were assessed through use of a Side × Muscle × Angle (2 × 3 × 5) repeated-measures ANOVA. EMG data recorded during each of the functional tests (toe walking and heel rise tests) were assessed using a Side × Muscle (2 × 3) repeated-measures ANOVA.
Results
Range of Motion, Calf Girth, and Tendon Width
Passive dorsiflexion ROM was decreased on the involved side (mean difference, –5.5° ± 8.0°; P = .015). Passive plantar flexion ROM was not significantly different between the involved and noninvolved sides (P = .115). Calf girth was also decreased on the involved side (mean difference, –1.1 ± 1.3 cm; P = .002). Achilles tendon width was greater on the involved side compared with the noninvolved side (mean difference, 8.0 ± 3.0 mm; P < .001) (Table 1).
Side-to-Side Differences in Calf Girth, Tendon Width, and ROM a
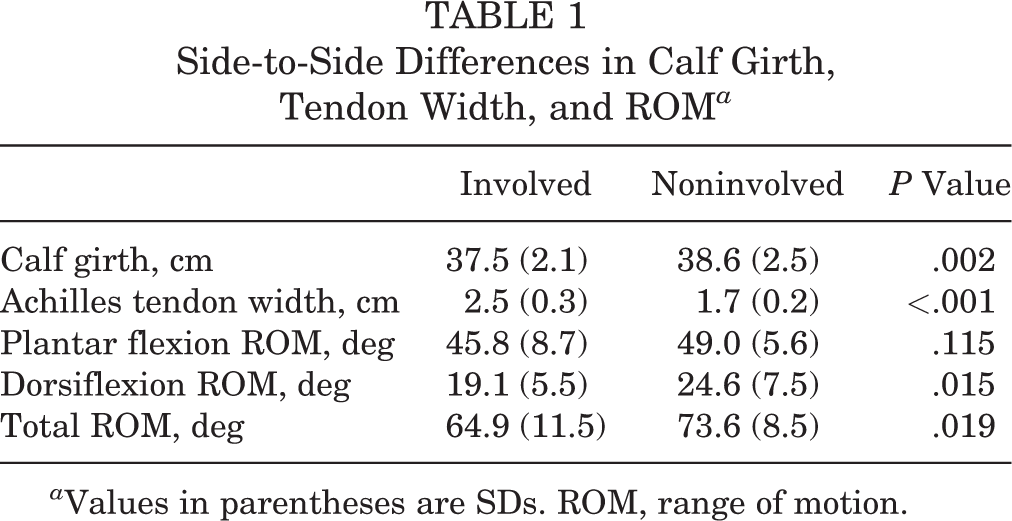
a Values in parentheses are SDs. ROM, range of motion.
Isometric Strength Testing and Passive Joint Stiffness
Despite the stronger repairs and avoidance of stretching, disproportionate end-range plantar flexion weakness was evident on the involved side. At 20° of plantar flexion, the strength deficit between the involved and noninvolved limbs was 27% ± 18% (P < .001), whereas that at 20° of dorsiflexion was 6% ± 17% (P = .390). Similarly, the strength deficit was 16.8% ± 16.7% (P = .005) at 10° of plantar flexion versus 7.5% ± 15.5% at 10° of dorsiflexion (P = .390) (Figure 1). Passive joint stiffness was not different between sides (involved side, 0.51 ± 0.29 N·m/deg; noninvolved side, 0.52 ± 0.33 N·m/deg; P = .945). Neither the side-to-side difference in dorsiflexion ROM nor the length of follow-up was correlated with the isometric strength deficits at each ankle angle (P > .05).

Isometric strength deficits at each test position. *Significant strength deficits were observed at 10° of plantar flexion (P = .001) and 20° of plantar flexion (P < .001). DF, dorsiflexion; PF, plantar flexion.
Functional Tests
The number of patients with failed heel rise tests increased as the starting position was changed to a more plantar flexed position (ie, from an incline to a flat surface to a decline) (linear effect, P < .001; Table 2). On an incline, 13 of 18 patients were able to perform a heel rise on an incline on the involved side, compared with all 18 on the noninvolved side. On a flat surface, 7 of 18 patients were able to perform a heel rise on a flat surface on the involved side, whereas all 18 patients were able to perform this test on the noninvolved side. On a decline, only 2 of 18 patients were able to perform a heel rise on the involved side, while all 18 patients could perform this test on the noninvolved side. Conversely, 14 of 18 patients were able to maintain similar heel height to the noninvolved side during the toe walk test (P = .104). Paired t tests indicated that there was no difference in the length of follow-up between those patients who successfully performed the functional tests and those patients who could not (P > .05).
Number of Patients With Impaired and Normal Functional Tests on the Involved Side a

a Values are expressed as n (%).
Electromyography
EMG activation was not different between the involved and noninvolved sides during the strength testing (P > .05) (Figure 2). Similarly, no side-to-side differences were found in EMG activation levels during the heel rise tests (P > .05) (Figure 3). Plantar flexor activity was greater on the involved side compared with the noninvolved side during the toe walk test (96% maximal voluntary contraction [MVC] on the involved side vs 75% MVC on the noninvolved side; main effect of side, P < .001) (Figure 3).

Combined average of the peak activities of the medial gastrocnemius, lateral gastrocnemius, and soleus in millivolts (mean ± SE) at each ankle position during the isometric strength tests. No significant difference was found between the involved and noninvolved limbs at any of the test positions (P > .05). DF, dorsiflexion; PF, plantar flexion.

Combined average of the peak normalized activities (% maximal voluntary contraction [MVC]) of the medial gastrocnemius, lateral gastrocnemius, and soleus (mean ± SE) during the functional tests. *A significant difference in activation was found between the involved and noninvolved limbs during the toe walk test (P < .001).
Discussion
Although Achilles repairs were augmented with an epitendinous cross-stitch weave and stretching was avoided for 12 weeks, patients still had marked weakness in end-range plantar flexion. Isometric strength deficits at 10° and 20° of plantar flexion were 17% and 26%, respectively, compared with 7.5% and 6% at 10° and 20° of dorsiflexion, respectively. Additionally, the heel rise tests indicated increasing impairment with increasing plantar flexion, as 28% of patients (5/18) had difficulty performing a heel rise on an incline, 61% (11/18) had impairment on a flat surface, and 89% (16/18) could not perform a heel rise on a decline. Based on these results and those of Mullaney et al, 14 a heel rise performed on a decline seems to be an effective clinical test to identify limitations in plantar flexion strength at end-range in the absence of strength testing.
Although weakness in end-range plantar flexion persisted following these treatment modifications, passive joint stiffness was not significantly different between the involved and noninvolved sides. This is in contrast to the findings of Borges et al 2 and Mullaney et al, 14 both of whom observed reduced stiffness in the repaired Achilles tendons at a mean follow-up of 40 and 21 months, respectively. Passive joint stiffness represents the resistance of all muscle tendon units and ligaments crossing the ankle joint to passive dorsiflexion. Herbert et al 5 demonstrated that 72% of the strain on the medial gastrocnemius muscle tendon unit imposed by passive dorsiflexion is absorbed by the tendon. Therefore, the passive stiffness measurement used in the current study can be interpreted to be a surrogate for tendon stiffness.
Additionally, in the current study, passive dorsiflexion ROM was decreased on the involved side compared with the contralateral side. Previous research has suggested that increased dorsiflexion ROM on the involved side may be indicative of tendon elongation. 10,12 Therefore, although we did not directly measure tendon length in the current study, the decreased dorsiflexion ROM on the involved side combined with the fact that there was no difference in passive joint stiffness between sides seems to indicate that significant separation of the tendon ends had not occurred. Both the normalization of passive joint stiffness and the reduction in passive dorsiflexion ROM were most likely due to the use of a stronger repair technique and avoidance of dorsiflexion stretching early in rehabilitation. Ankle joint capsule stiffness, which is a component of the overall passive joint stiffness measurement, also may have increased due to the lack of stretching for 12 weeks. This may have contributed to the decreased dorsiflexion ROM and normalization of the overall passive joint stiffness.
The fact that no correlation was found between the difference in passive dorsiflexion ROM and the isometric strength deficits suggests that elongation of the repaired tendon may not be a major contributor to end-range plantar flexor weakness following surgery. Similar results have been shown using direct measurements of tendon length. Using radiographic measurements (ie, tantalum bead implantation), both Kangas et al 7 and Heikkinen et al 3 found no correlation between tendon elongation and isokinetic plantar flexion strength. Agres et al 1 used ultrasonography to measure tendon length in patients following Achilles tendon repair and found that tendon elongation was not a contributing factor to asymmetries in plantar flexion moments generated during gait. The effect of tendon elongation on strength deficits, however, remains controversial, as both Heikkinen et al 4 and Pajala et al 17 documented a significant correlation between strength and tendon length.
Despite these positive changes, calf circumference was significantly decreased on the involved side, indicating persistent muscle atrophy. Although this may partially explain the overall weakness on the involved side, the disproportionate weakness with the plantar flexors in a shortened position may be due to inefficient transmission of contractile forces through the repaired tendon. When the muscle is in a shortened position, the tendon is responsible for a greater change in length in order to generate force. With the ankle in a plantar flexed position, the baseline passive tension in the plantar flexors and the repaired Achilles tendon is quite small and not enough to produce the change in length necessary to generate force equivalent to the noninjured side. In addition, although both ends of the tendon are sewn together, the individual fibers may not be directly reconnected. Thus, force transmission through the tendon may be disrupted and inefficient.
Previous studies have shown altered EMG activity of the calf muscles on the involved side compared with the contralateral side following Achilles tendon repair. 6,20,21 At 1 year after surgery, Wang et al 21 found reductions of 16% to 30% in calf muscle activation on the involved side compared with the noninvolved side during a maximal isometric plantar flexion task. This decrease in muscle activation was attributed to reduced neuromechanical response and function. In the current study, calf muscle activation was not found to be different between the involved and noninvolved sides during the isometric strength tests and the heel rise tests. Therefore, these EMG findings seem to indicate that the observed plantar flexion weakness was not due to neural inhibition.
The only difference in EMG activity of the calf muscles between the involved and noninvolved limbs was observed during the toe walk test. Suydam et al, 20 examining patients at 1-year follow-up, found a correlation between the increased triceps surae muscle activity during gait and the increased resting Achilles tendon length on the involved side. These authors concluded that the increase in muscle activity compensated for the increased tendon length by facilitating the removal of the additional slack in the tendon before movement was produced in the ankle joint. Considering the limited ROM at the ankle during the toe walk test and the decreased calf circumference on the involved side, it is possible that the increase in EMG activity of the calf muscles on the involved side could be compensating for the decrease in muscle mass rather than removing the slack in the tendon.
A few limitations of this study warrant mention. The length of the Achilles tendon was not measured directly. While ultrasonography or MRI imaging would have allowed us to quantify the amount of elongation that had occurred during the follow-up period and to correlate this measurement with the strength deficits throughout the ROM, these modes are not very practical to use in a clinical setting. Previous research has shown that increased dorsiflexion ROM on the involved side can indicate tendon lengthening. 10,12 In the current study, we found a decrease in dorsiflexion ROM on the involved side and no difference in passive ankle joint stiffness between sides. Taken together and in the context of previous research, these simpler measurements seem to indicate that tendon elongation in this patient group was most likely minimal.
Another limitation of this study was the low rate of follow-up. As only 18 of the 55 eligible patients volunteered for testing, our results may not be completely representative of the outcomes in the whole group. Additionally, the level of function of these patients was not objectively assessed. Therefore, it is not known whether this study group had high, low, or normal function. However, as per our inclusion criteria, each patient successfully completed rehabilitation, was satisfied with his or her surgery, and was able to return to his or her desired level of activity. Future research is needed to further examine any connections between weakness in end-range plantar flexion and objective measures of function. Finally, because this study was a within-participant comparison (involved vs noninvolved limb), it is not known whether our results can be generalized to patients with Achilles tendon repair who are treated with other surgical or nonsurgical techniques.
Conclusion
The use of stronger repair techniques and attempts to limit tendon elongation by avoiding dorsiflexion stretching did not eliminate weakness in end-range plantar flexion following Achilles tendon repair. Our data indicate that this continued weakness was not due to neural inhibition. Muscle atrophy or physiological changes that alter the force transmission capability of the healing tendon may be responsible for this continued impairment. This weakness has implications for high-demand jumping and sprinting after Achilles repair. Future research should focus on determining whether reducing atrophy and specifically strengthening the plantar flexors in end-range can reduce this weakness.
Footnotes
One or more of the authors has declared the following potential conflict of interest or source of funding: B.B.B. is a paid speaker/presenter for Arthrex and Stryker and receives educational support from Arthrex and Stryker. S.J.L. is a consultant for, receives royalties from, is a paid speaker/presenter for, and receives educational support from Arthrex and Peerless Surgical. S.J.N. is a consultant for Arthrex, receives royalties from Arthrex, and receives educational support from Arthrex.
Ethical approval for this study was obtained from the Office of the Human Research Protection Program at North Shore–Long Island Jewish Health System (IRB No. 14-263A).
