Abstract
Background:
The use of autologous osteochondral grafts has become popular in the treatment of small, isolated, well-contained articular cartilage defects. However, donor site morbidity is a problem, and few reports are available of donor site morbidity after mosaicplasty.
Purpose:
To examine the clinical outcomes of donor sites after osteochondral grafts from healthy knees.
Study Design:
Case series; Level of evidence, 4.
Methods:
Between September 1997 and September 2011, there were 40 patients (40 knees; 32 men, 8 women; 31 right knees, 9 left knees) with asymptomatic osteochondral graft donor sites used for autologous transfer; all had a follow-up period of >2 years. The mean patient age at surgery was 21.0 years (range, 12-58 years). The recipient sites included the elbow (n = 28), contralateral knee (n = 5), and ankle (n = 7). The mean diameter of the grafted plugs was 7.5 mm (range, 4.5-9 mm), and the mean number of grafted plugs was 2.2 (range, 1-3). At a mean follow-up of 43.1 months (range, 24-177 months), knee symptoms, return to sport, ability to sit straight in Japanese style, and radiological changes of the patellofemoral joint were evaluated. Whether operative age, follow-up period, and diameter or number of the grafted plugs were risk factors was analyzed statistically. Significance was defined as P < .05.
Results:
Thirty-four patients had no knee symptoms, and 4 patients had occasional mild knee pain. Two patients underwent reoperation for arthrofibrosis and not for cartilage defect. Twenty-seven patients had complete return to sports, and 32 patients could sit straight; donor site morbidity was not the cause of failure to return to sports or inability to sit straight. The radiological changes became worse in 3 patients, and the risk factor for degenerative change was older operative age.
Conclusion:
When osteochondral plugs were obtained from healthy knees, 34 patients (85%) were asymptomatic at follow-up. No donor site defects required surgical intervention due to persistent symptoms.
More than 20 years have passed since Matsusue et al 18 first reported autologous osteochondral grafts of the knee. The use of autologous osteochondral grafts has recently become common for the treatment of small, isolated, well-contained, articular cartilage defects. 3,11,16,18,32 In osteochondral diseases such as osteonecrosis and osteochondritis dissecans, the technique, termed mosaicplasty, has been reported to have good clinical outcomes. 20 –22 The long-term clinical outcome after mosaicplasty for a symptomatic osteochondral defect in the knee is good, but it varies greatly depending on patient age and sex and the size and cause of the lesion. 8,29
Donor site morbidity after mosaicplasty is an issue. However, few reports of donor site morbidity have been published. 13,15 If the recipient site is an elbow 20 or ankle, 23 a healthy knee must be the donor site. If recipient sites in the knee joint are large, the contralateral knee joint may be the donor site if it is healthy. Therefore, when we have performed mosaicplasty, the asymptomatic knee joint serves as the donor site for osteochondral plugs transferred to either the symptomatic knee or the joints. The best way to determine donor site morbidity after mosaicplasty is subjective evaluation of symptoms and clinical examination of the donor knee. Thus, the objective of this study was to determine the clinical outcomes after osteochondral graft harvest from healthy knees.
Methods
Patients with autologous osteochondral grafts that were harvested from asymptomatic knees from September 1997 to September 2011 were initially examined. Of 58 patients, 40 had more than 2-year follow-up (69% follow-up rate) and were selected for this study. The mean age of the patients at the time of operation was 21 years (range, 12-58 years); there were 32 men and 8 women. The mean follow-up was 43.1 months (range, 24-177 months). All procedures were reviewed and approved by the research ethics committee of our hospital.
The diagnosis for which the osteochondral transfer was performed is shown in Table 1. The recipient sites were 28 elbows, 5 contralateral knees, and 7 ankle joints. Of the 18 patients with less than 2 years of follow-up, the recipient sites were 11 elbows, 6 ankles, and 1 metatarsal bone. In 39 of the 40 study patients, the donor site was the lateral side of the femoral trochlea. The mean diameter and number of grafted plugs are shown in Table 2. The mean diameter of the grafted plugs was 7.5 mm (range, 4.5-9 mm), and the mean number of grafted plugs was 2.2 (range, 1-3). In general, the donor defects were filled with bone chips from the recipient sites to subchondral bone level, not to the articular cartilage surface (Figure 1). A drain tube was inserted into the donor knee for 2 days after mosaicplasty. After removal of the drain tube, range of motion exercise of the knee and weightbearing were started.
Diagnosis Leading to Osteochondral Transfer

Diameter and Number of Implanted Plugs


The donor site of an 18-year-old woman who had osteochondritis dissecans of her right talus was the lateral edge of her right trochlea. Two plugs, 7 mm in diameter, were harvested, and the donor defects were filled with bone chips from the recipient site to the level of the subchondral bone.
At follow-up, knee symptoms, return to sport, the ability to sit straight in Japanese style (seiza in Japanese), and radiological changes of the patellofemoral joints were evaluated. The presence of donor site symptoms was defined as knee symptoms persisting more than 1 month after mosaicplasty. The Kellgren-Lawrence classification was used for radiological changes. Whether operative age, follow-up period, and diameter or number of grafted plugs were risk factors for knee symptoms was analyzed statistically by use of the Mann-Whitney U test; a significant P value was defined as less than .05.
Results
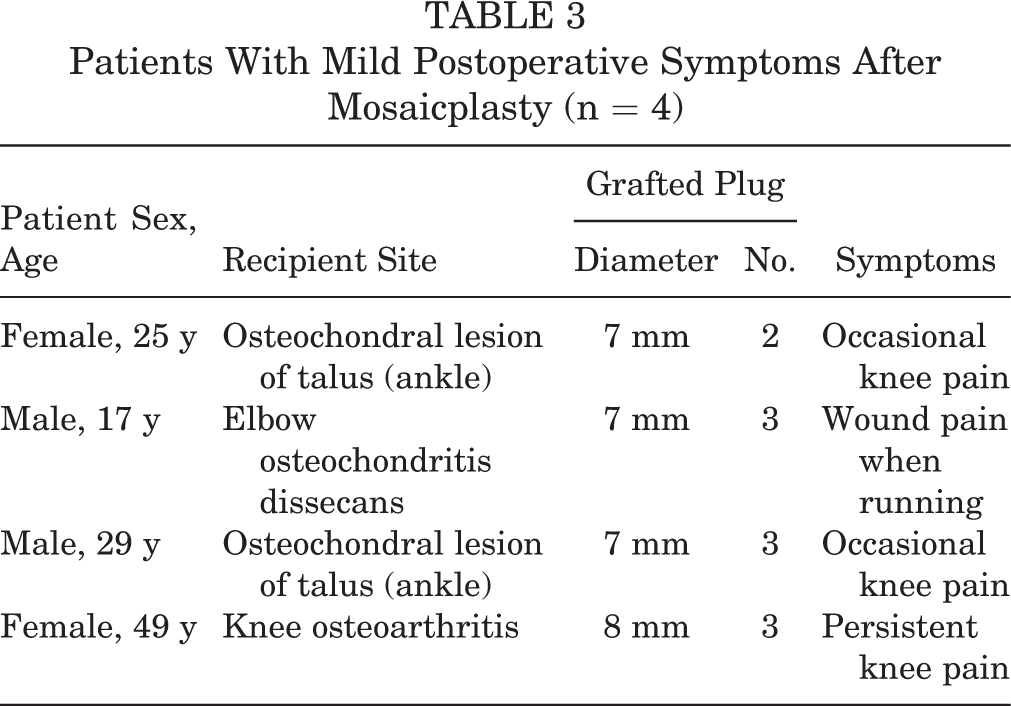
Thirty-four patients (85%) had no symptoms, and 4 patients had occasional mild knee pain 1 month after mosaicplasty. The details are shown in Table 3. These 4 patients did not need any additional treatment.
Patients With Mild Postoperative Symptoms After Mosaicplasty (n = 4)
Two patients had reoperations for arthrofibrosis of the knee that was secondary to the arthrotomy used to harvest the donor plugs. One was a 44-year-old man who had occasional knee pain and catching 16 months after mosaicplasty for osteoarthritis of the right knee. At the time of reoperation, the medial donor site of his left knee was filled with fibrocartilage-like tissue (Figure 2A). A tight fibrous tissue band was seen in his medial capsule (Figure 2B). The cartilage injury of his left medial condylar edge was probably due to this tight fibrous scar band (Figure 2C). The patient underwent resection of the fibrous band (Figure 2D) and had no symptoms in the donor knee after reoperation. The other patient was a 14-year-old boy. He could not return to sport due to donor knee pain for 1 year after mosaicplasty. He underwent resection of capsular scar tissue, experienced no subsequent donor symptoms, and made a full return to sport after reoperation.

(A) At 16 months after the mosaicplasty, the medial donor site of the left knee is filled with fibrocartilage-like tissue. (B) A tight fibrous scar band is shown in the medial capsule. (C) A cartilage injury at the left medial condylar edge was probably caused by the tight fibrous tissue band. (D) After resection of the fibrous scar tissue.
A summary of the risk factors comparing the group without symptoms (34 patients; 85%) and the symptomatic group (6 patients; 15%) is shown in Table 4. We found that 12.5% of male and 25% of female patients were symptomatic. None of the analyzed risk factors were significantly associated with symptoms.
Comparison of Patients With and Without Symptoms a

a Values are expressed as mean (range).
Return to Sport
With respect to return to sport, 10 patients did not participate in sports preoperatively; of the remaining 30 patients, 27 returned to sports, including 25 patients who had undergone elbow treatment and 2 patients who had undergone ankle treatment. The sport performance of 3 patients, all receiving elbow treatment, was less than 50% of the preoperative level due to recipient site symptoms. All patients had negative patellar compression tests in their donor knees. With respect to seiza, 32 patients could sit straight in Japanese style: 100% of elbow patients, 20% of knee patients, and 43% of ankle patients. An inability to sit straight was attributed to problems with the recipient site, not the donor knee.
Radiological Changes
A total of 37 patients showed no radiological changes of the patellofemoral joint from mosaicplasty to the end of follow-up, including 36 patients with grade 0 and 1 patient with grade 1 Kellgren-Lawrence changes of the knee. Radiological changes became worse in 3 patients: 1 patient (without symptoms) progressed from grade 1 to 2, and 2 patients (with symptoms) progressed from grade 0 to 2 (Figure 3). The risk factors for radiological changes were older operative age (P = .0170) and the area of the recipient site (P = .0102). The mean operative age and area of the recipient site in the patients with no radiological changes were 18.9 years and 186.1 mm2, respectively, while in those with radiological changes they were 47.0 years and 583.3 mm2.

(A) Preoperative skyline view of the right knee of a 49-year-old woman with osteoarthritis of the left knee. (B) At 73 months after mosaicplasty, an osteophyte of the lateral patella is shown in her skyline view of the right knee. She had 3 donor sites in the lateral trochlea of her right knee at mosaicplasty.
Case Reports
The following are 2 case reports. Patient 1 had right elbow osteochondritis dissecans and mosaicplasty at 13 years of age. 20 Fifteen years after mosaicplasty, his knee radiographs were normal (Figure 4). He had no symptoms in his donor knee joint. Patient 2 had an osteochondral lesion of the right talus and mosaicplasty at 29 years of age. 23 Because he developed recurrent right ankle pain, he underwent surgery of his right ankle and a second-look arthroscopy of his right knee. The second-look at 77 months after mosaicplasty showed that the donor site was filled with fibrocartilage-like tissue, and his patellar cartilage was normal (Figure 5). He had no symptoms in his donor knee joint.

Case presentation 1. The patient had elbow osteochondritis dissecans and underwent mosaicplasty at 13 years of age. Fifteen years after mosaicplasty, the knee radiographs are normal.

Case presentation 2. The patient had an osteochondral lesion of the talus and underwent mosaicplasty at 29 years of age. A second-look arthroscopy at 77 months after mosaicplasty shows that the donor site is filled with fibrocartilage-like tissue, and the patellar cartilage is normal.
Discussion
Reports are available that indicate donor site morbidity after mosaicplasty but not when the grafts are harvested from the healthy knee. Hangody et al 8 reported that patellofemoral pain related to graft harvest was observed in 5% of patients and that second-look arthroscopies showed acceptable fibrocartilage coverage of donor sites in 16 patients. Another study of 652 patients treated with mosaicplasty reported that 6 patients had mild or moderate patellofemoral pain with strenuous physical activity during the first postoperative year only, 34 patients had painful hemarthroses after surgery, and 4 patients developed deep infections. 10 Reverte-Vinaixa et al 28 reported that 1 of 17 patients had osteoarthritis at the graft donor site. Fifteen percent of patients in the present study had some donor site morbidity, and fibrocartilage coverage of the donor site was seen at second-look (Figures 2A and 5). Three patients showed worsening of the patellofemoral joint on radiographs. However, no postoperative deep infections or hemarthroses occurred, because a drain tube was inserted into the donor knee for 2 days after mosaicplasty.
Case series have reported harvesting of grafts from asymptomatic knees. In studies in which elbow joints were the recipient sites, Iwasaki et al 13 –15 reported that all knees were graded as excellent and normal based on the Lysholm knee score and the International Knee Documentation Committee evaluation form. Magnetic resonance imaging showed 50% to 100% defect fill in 6 of 9 patients and normal or nearly normal signals in 4 patients at the donor sites. One patient had mild anterior knee pain with stair-climbing. Nishimura et al 25 reported that strength testing of the knee extensors showed that 8 of 12 patients had reduced muscle strength at 3 months compared with the preoperative level. With ankle joints as the recipient sites, Hangody et al 9 reported that the symptoms in the donor knee were resolved at 1 year in 98% of patients. Reddy et al 27 reported a mean postoperative Lysholm score of 81 and noted that osteochondral harvest from asymptomatic knees for the treatment of talar osteochondral lesions led to decreased knee function. Valderrabano et al 31 reported knee pain in 6 patients (50%). Paul et al 26 reported that the mean Lysholm score was 89 and that the operative age, diameter, and number of grafted plugs were not risk factors, but high body mass index was a risk factor. In the present study, 15% of patients (n = 6) had donor symptoms, including minor symptoms that did not require treatment, in the donor knee after mosaicplasty. This rate was small compared with the above-mentioned studies. The risk factors in the symptomatic group were not clear in the present study.
Investigators have evaluated methods to decrease donor site morbidity. Nishida et al 24 found that the proximal lateral trochlea of the distal femur had the lowest bone density of the patellofemoral joint and that the proximal lateral trochlea was the optimal site for harvesting osteochondral grafts. We harvested grafts mainly from the proximal lateral side of the trochlea. Bartha et al 1 filled the donor sites with artificial materials to reduce donor site morbidity and postoperative bleeding after mosaicplasty. Using a rabbit model, Intzoglou et al 12 reported that the donor site area was better revised with osteochondral graft than with a bone graft substitute implant. In the present study, the donor defects were filled to the level of the subchondral bone with bone chips from the recipient site. Feczko et al 4 reported that larger grafts (diameter >8 mm) were associated with greater donor site morbidity. Guettler et al 7 indicated that the creation of 5-mm donor defects in the lateral aspect of the lateral femoral condyle did not lead to significant alterations in local contact pressure, and the investigators noted that donor site morbidity might be minimized if donor site defects were limited to 5 mm or smaller. In the present study, the diameter of the donor plug was not associated with increased symptoms; therefore, we allow harvesting of a 10-mm-diameter donor plug in men and an 8-mm-diameter donor plug in women, and we have not found donor symptoms due to the larger diameter of the donor plugs.
Some investigators have examined radiological changes at the recipient site after mosaicplasty. Gudas et al 6 found no arthritic changes on plain radiographs either before surgery or at 3-year follow-up. At 10-year follow-up, 7 (25%) patients had Kellgren-Lawrence grade 1 osteoarthritis, but these findings did not affect the final International Cartilage Repair Society scores. 5 However, few reports are available regarding radiological changes at donor sites after mosaicplasty. 19 Mukai et al 19 reported that patients older than 40 years had significant progression of radiological changes at the donor site more than 5 years after mosaicplasty. The cause of the progression in radiological changes may have been aging. 19 In the present study, 3 patients developed worse radiological changes at the donor site (see Figure 3). The risk factors were older operative age and the area of the recipient site. However, aging may also have contributed to these changes.
Reoperation after mosaicplasty for recipient site pathological change has been reported several times. Lim et al 17 reported a subsequent arthroscopy for a patient with limited range of motion due to prominence of the osteochondral plug. Ulstein et al 30 reported 5 reoperations, and Bentley et al 2 reported 23 reoperations at long-term follow-up. Nakagawa et al 23 reported reoperation of mosaicplasty in the talus for osteonecrosis of the grafted plug. Only 1 report is available regarding reoperation after mosaicplasty due to donor site pathological change. 27 One patient had persistent pain at the donor site that required a subsequent arthroscopy and lateral retinacular release 8 months after ankle mosaicplasty. The condition of the articular cartilage was normal at the time of arthroscopy, and the donor sites were well healed. In addition, the patient required a tibial tubercle realignment 10 months after lateral release. 27 In the present study, 2 patients underwent reoperation, 1 at 12 months and the other 16 months after mosaicplasty. The donor sites were fully filled with fibrocartilage-like tissue, and the patellar cartilages were normal. The cause of donor site pain in these cases was capsular scar tissue, not problems associated with the donor cartilage. After resection of the scar tissue, the donor pain resolved, unlike the result reported by Reddy et al. 27
Regarding limitations, the present study was retrospective and had a moderate number of patients (N = 40) and short follow-up (43.1 months). A greater number of patients with long-term follow-up is needed.
In conclusion, the current study presents 40 patients who underwent mosaicplasty in which the healthy knee was used as the donor site. At a mean follow-up period of 43.1 months, there were no symptoms in the donor knee in 34 patients (85%). No patients had reoperation due to cartilage defects in the donor site. Therefore, we recommend that a healthy knee be used as the donor site if the potential benefits of mosaicplasty are thought to be substantial.
Footnotes
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution.
Ethical approval for this study was obtained from the National Hospital Organization Kyoto Medical Center, Kyoto, Japan.
