Abstract
Rotator cuff tears are common and result in considerable morbidity. Tears within the tendon substance or at its insertion into the humeral head represent a considerable clinical challenge because of the hostile local environment that precludes healing. Tears often progress without intervention, and current surgical treatments are inadequate. Although surgical implants, instrumentation, and techniques have improved, healing rates have not improved, and a high failure rate remains for large and massive rotator cuff tears. The use of biologic adjuvants that contribute to a regenerative microenvironment have great potential for improving healing rates and function after surgery. This article presents a review of current and emerging biologic approaches to augment rotator cuff tendon and muscle regeneration focusing on the scientific rationale, preclinical, and clinical evidence for efficacy, areas for future research, and current barriers to advancement and implementation.
Keywords
Rotator cuff disease accounts for over 4.5 million physician visits per year, with over 250,000 repairs performed annually in the United States. 30,139 Cadaveric studies have revealed a 6% incidence of full-thickness rotator cuff tears in those aged <60 years, which rises to 30% in those aged >60 years. 77 For many patients, these injuries result in considerable disability, pain, and a diminished quality of life. 38 Current standard surgical treatment involves open or arthroscopic repair of the torn tendon(s) frequently using suture anchors in a variety of configurations. 86 However, considerable variation in surgical procedures exists, confirming the lack of a single reliable technique. 86 Despite improvements in pain and function after rotator cuff repair, the rate of persistent tears has been reported from 11% to 94%, and it is recognized that current surgical treatments need improvement. 40,43 In this review article, we outline the pathogenesis of rotator cuff tears outlining current and emerging biological approaches that aim to improve healing and function after surgical repair. In addition, we discuss critical areas for future research and barriers to the advancement and implementation of biologic therapies for the rotator cuff, identified at the 2015 American Orthopaedic Society for Sports Medicine (AOSSM) Biologics II Think Tank. 1
Pathogenesis of Rotator Cuff Tears
The pathogenesis of rotator cuff tendon tears is multifactorial but can be broadly considered as resulting from chronic degeneration, trauma, or a combination of these factors. Chronic degeneration can occur through extrinsic impingement from bony structures surrounding the cuff and degeneration within the tendon itself where tensile loads exceed intrinsic healing and adaptive responses. Repetitive microtrauma is commonly seen in overhead or throwing athletes while acute avulsions of the rotator cuff are associated with contact sports or trauma. 14
Biologic approaches target a particular aspect of the injury microenvironment. An appreciation of the normal healing response allows clinicians to identify which strategy may be of greatest benefit for individual patients. The rotator cuff tendon is composed primarily of type I collagen produced by tissue-specific fibroblasts called tenocytes. 125 Rotator cuff muscle is highly vascular but the size and number of vessels progressively decrease within the tendinous region and in proximity to the humeral head insertion. 17 At the site of insertion into the proximal humerus, there is a transition into 4 distinct tissue zones: tendon, nonmineralized fibrocartilage, mineralized fibrocartilage, and bone. 6 This arrangement is critical to the ability of the rotator cuff to withstand force. Tendon heals in 3 overlapping phases, namely an inflammatory, proliferative (or fibroblastic), and remodeling phase. 125 Cells from both within and outside the tendon contribute at each stage. The inflammatory phase occurs over the 7 days after injury and is characterized by fibrin and fibronectin deposition by platelets. 34,125 Platelets also release a host of growth factors, including insulin-like growth factor–1 (IGF1), platelet-derived growth factor (PDGF), and transforming growth factor–β (TGFβ), that attract macrophages and neutrophils. 34 Macrophages secrete further TGFβ, promoting activation of myofibroblasts and the formation of scar tissue. Mesenchymal stem cells (MSCs) capable of differentiating into tenocytes and myofibroblasts are recruited to the area. 142 The proliferative stage begins within 48 hours and lasts up to 6 weeks. 125 In the remodeling phase, type III collagen laid down initially is remodeled to type I collagen resulting in a more organized matrix. 34 Full-thickness tears of the rotator cuff initiate a cascade of alterations that compromise the entire muscle-tendon unit. 45,46 In both clinical and animal studies, changes in muscle architecture and structure have been reported. 45,46 After a rotator cuff tear, fat accumulates within the muscle, which itself undergoes atrophy. 69 Tendomuscular retraction and muscle fibrosis result, creating tension over the site of injury, further compromising healing potential. 46
Animal studies have confirmed that the healing tendon-bone insertion after rotator cuff repair does not resemble the native enthesis. 19 Instead of the 4 transitioning zones, the repaired tendon forms fibrovascular scar tissue with a large proportion of type III collagen rather than the biomechanically superior type I collagen. 74 In particular, the zone of calcified cartilage does not reform, and this scar tissue is biomechanically weaker and more prone to failure than the native insertion. 74
A Hostile Environment for Healing
Studies have identified biological factors that are key determinants of failure after rotator cuff repair, including intrinsic tendon degeneration, muscle degeneration, fatty infiltration, and a lack of vascularity. These studies highlight the unique challenges of tissue healing and regeneration at the rotator cuff tendon and its enthesis.
Limited Blood Supply
The vascularity of rotator cuff tendon at the anterolateral rotator cuff is significantly lower than that at the bony insertion, limiting a meaningful inflammatory response and access to extrinsic progenitors. 13 Although no studies have directly proven a causal relationship between increased vascularity and improved rotator cuff healing, emerging data suggest that stimulating angiogenesis may improve tensile strength of Achilles tendons after repair. 150
Absence of Appropriate Signals Directing MSC Recruitment and Differentiation
An absence of appropriate molecular signals to direct cell differentiation at the zone of injury has been theorized to contribute to the formation of biomechanically inferior fibrotic tissue, although this is not currently substantiated by robust evidence. 50,74,111 This is the rationale for strategies that aim to direct healing away from scar formation toward the regeneration of a native tendon-bone insertion site through the application of growth factors that stimulate tenocyte and bone differentiation.
Local MSC Population Depleted in Patients With Rotator Cuff Tears
In a large study of 200 patients, Hernigou et al 59 demonstrated that the number of MSCs present in the greater tuberosity of patients with a rotator cuff tear decreases as a function of a number of clinical factors, including lag time from tear onset to treatment, tear size, chronicity, and degree of fatty infiltration, among others. Therefore, patients in the greatest need of successful repair are the ones who are poorest equipped to heal.
Intra-articular Location of Rotator Cuff Limits Inflammatory and Proliferative Response
Synovial fluid with its fibrinolytic enzymes prohibits the formation of a blood clot, which subsequently leads to a lack of provisional scaffold bridging the wound site. 101
Tension Over the Healing Site
Tension over the rotator cuff repair prevents healing and increases the risk of rerupture. 40 Tear size and tenovascular retraction secondary to muscle atrophy and fibrosis both contribute to this phenomenon. 40
Biologic Options to Augment Healing
Biologic adjuvants have great potential to improve rotator cuff healing and reduce rates of reinjury. The ultimate goal after rotator cuff injury is to re-create the native tendon insertion site formed during embryologic development, and as such, improve rotator cuff tendon healing while at the same time diminishing, halting, or reversing fatty degeneration of the muscle. Current and emerging biological approaches include the application of growth factors, platelet-rich plasma (PRP), stem cells, and patches.
Growth Factors
Numerous cytokines and growth factors can be found in the rotator cuff in predictable expression patterns early in the healing process. These factors function primarily to promote the inflammatory response/angiogenesis (vascular endothelial growth factor [VEGF], basic fibroblast growth factor [bFGF], PDGF); increase cellular proliferation, differentiation, and matrix synthesis (TGFβ); promote bony incorporation of tendon (bone morphogentic protein [BMP]); and remodel extracellular matrix (matrix metalloproteinase [MMP]). While no human study investigating recombinant growth factors in rotator cuff tendon healing has yet been published, the results of animal studies indicate that delivery or modulation of growth factors may provide a useful way of improving the biology of rotator cuff healing. 61,134
Proinflammatory and Angiogenic Cytokines
Tendon degeneration and tears frequently affect the most distal hypovascular portions of the supraspinatus and infraspinatus tendons, close to their insertion sites, prompting some investigators to suggest that the establishment of vascularity at the tendon-bone interface may promote healing. 133 However, there still remains a paucity of data to support or refute this concept.
VEGF describes a family of signaling proteins that stimulate vasculogenesis through tyrosine kinase receptor–mediated signaling cascades. 103 No specific study has evaluated VEGF augmentation of rotator cuff repairs, although conflicting results have been achieved in animal models of Achilles tendon healing and anterior cruciate ligament (ACL) reconstruction. 148,150 Application of VEGF improved tendon tensile strength in a rat model of Achilles tendon repair 150 while ACL grafts soaked in VEGF prior to implantation in a sheep model of ACL repair resulted in increased graft laxity. 148
Fibroblast growth factors (FGFs) act by stimulating proliferation of capillary endothelial cells. In addition, they are recognized to have a mitogenic effect on mesenchymal cells. 103 While both FGF1 and FGF2 (bFGF) are reported to be present early within the rotator cuff repair process, basic FGF is considered the more potent mitogen 72,144 for mesenchymal stem cells. Ide et al 61 reported that FGF2, when applied to repaired supraspinatus tears in rats, accelerated bone ingrowth resulting in improved biomechanical properties and better histologic scores at 2 weeks compared with controls. However, no differences were seen between the control and experimental groups at 4 and 6 weeks, suggesting that FGF2 accelerated ingrowth but not the final result. 61 In a study examining the effects of FGF2 applied in an acellular dermal matrix, Ide et al 62 demonstrated that FGF2 improved histologic and strength scores compared with controls. These studies support a possible role for FGF2 in accelerating and remodeling the rotator cuff tendon repair site. 61
PDGF is a proinflammatory cytokine important in rotator cuff healing that peaks between 7 and 14 days after injury. 72 PDGF exists in 3 dimeric isoforms: PDGF-AA, -BB, and -AB. These isoforms function as chemotactic agents for inflammatory cells and promote type I collagen synthesis. Uggen et al 134 examined the effects of recombinant human PDGF-BB–coated sutures on rotator cuff healing in a sheep model. They reported enhanced histological scores in animals receiving PDGF over controls at 6 weeks, although there was no significant difference between the groups in terms of ultimate load to failure. Hee at al 57 reported that addition of recombinant human PDGF-BB implanted in a type I collagen graft for rotator cuff repair in sheep improved the biomechanical strength and anatomic appearance of healing tendons compared with controls. Importantly, the response was dependent on the dose, timing, and the PDGF delivery vehicle used. PDGF augmentation holds promise for rotator cuff repair but further studies are required to determine its efficacy.
Cytokines to Increase Cellular Proliferation, Differentiation, and Extracellular Matrix Synthesis
The TGFβ superfamily of cytokines includes 3 isoforms (TGFβ1, TGFβ2, and TGFβ3) that have diverse physiologic effects, including cellular proliferation, differentiation, and matrix synthesis within the tendon. 4 Postnatally, wound healing is characterized by extensive scar tissue formation with high levels of expressed TGFβ1, while the wound microenvironment in scarless fetal healing is characterized by high levels of TGFβ3. 28 It has therefore been proposed that the delivery of exogenous TGFβ3 could promote regenerative healing of a torn rotator cuff at the tendon-bone insertion site. 73 With this in mind, 3 recent studies have examined the use of TGFβ3 in a rat rotator cuff model. Kim et al 70 investigated the role of TGFβ1 and TGFβ3 at the tendon-bone insertion of repaired rat supraspinatus tendons. Animals receiving TGFβ1 through an osmotic pump had increased type III collagen production, consistent with a scar-mediated healing response, and demonstrated a trend toward reduced mechanical properties compared with controls. TGFβ3 delivery resulted in no histological or biomechanical advantage. Using a system that delivers TGFβ3 directly to the tendon-bone insertion, the same authors demonstrated accelerated healing in experimental animals with increased inflammation, cellularity, vascularity, and cell proliferation at early time points. 82 In addition, significant improvements in structural and mechanical properties were demonstrated. Kovacevic et al 73 examined the delivery of TGFβ3 in an injectable calcium-phosphate matrix to the tendon-bone interface of repaired rat supraspinatus tendons. The addition of TGFβ3 to the matrix significantly improved strength at the repair site 4 weeks postoperatively. Overall, these findings suggest a potential therapeutic role for TGFβ3 in improving tendon healing after surgical repair.
Osteoinductive Factors
Healing of the rotator cuff onto the humerus is dependent on bone ingrowth into the repaired tendon. 115 It has therefore been hypothesized that osteoinductive growth factors would improve rotator cuff tendon healing. The BMPs belong to the TGFβ superfamily and are best known for their ability to stimulate bone formation, although they also regulate fibrocartilage, neotendon, and ligament formation. 78 Certain BMPs, namely BMP1, BMP12, BMP13, and BMP14, are expressed in the acute phase of healing after rotator cuff tears 144 while a number of BMPs (including BMP2 and BMP7) have been reported to induce collagen production when added to cultured tenocyte-like cells derived from samples of human rotator cuff. 107 For these reasons, the ability for BMPs to augment rotator cuff healing is currently under investigation in preclinical studies.
Studies in sheep and rats have demonstrated that BMP therapy is beneficial in rotator cuff repair. 95,123 Murray et al 95 reported that delivery of BMP13 on a collagen sponge improved the maximum loads to failure at 6 weeks after surgery when compared with controls in a rat model of supraspinatus repair. At 6 weeks, BMP13-treated repairs had recovered to 80% of the “strength to failure” of uninjured controls. Application of recombinant human BMP12 alone in a collagen or hyaluronic acid sponge strengthened the repair and accelerated healing in an ovine model of rotator cuff repair. 123 In these studies, BMP12 and BMP13 improved the strength and histological appearance of repaired tendons with improved cellular organization and fiber alignment. Dines et al 31 reported that coating of sutures with recombinant BMP14 accelerated tendon healing and enhanced strength. However, no biomechanical or histologic difference was found when compared with controls at 6 weeks. Rodeo et al 115 reported that a combination of growth factors (BMP2 to BMP7, TGFβ1 to TGFβ3, and FGF) within an osteoinductive bone protein extract placed on the tendon-bone interface resulted in stronger repairs, illustrated by greater load to failure at 12 weeks. However, when the loads were normalized for tissue volume, no differences between treated and untreated shoulders were found, which may suggest that the bovine cortical bone extract may have accelerated the healing process without improving repair quality. Similar results were observed with addition of recombinant human BMP12 to acute full-thickness tears in sheep. 123 These findings suggest a potential therapeutic role for osteoinductive factors in improving tendon-bone healing after surgical rotator cuff repair.
Regulators of Extracellular Matrix
A dynamic balance between MMPs and their inhibitors (tissue inhibitors of metalloproteinases [TIMPs]) maintains the integrity of tissue extracellular matrix. 16 Dysregulation of this homeostasis with elevated levels of MMPs is associated with tendon degeneration and rupture. 113 Elevated levels of MMP1 and MMP3 have been demonstrated in the synovial fluid of patients with massive rotator cuff tears. 147 Furthermore, McDowell et al 85 demonstrated that MMP-mediated tissue degradation within the immediate postoperative period after a surgical repair leads to weakening of the rotator cuff tendon at the repair site. As such, the possibility that selective inhibition of MMPs may have a positive effect on tendon healing has recently been explored. In addition to its antibiotic effect, doxycycline is a potent MMP inhibitor that acts by competitive binding of the zinc binding site of MMP enzyme and by reducing MMP gene transcription. 106 Using a rat model, Bedi et al 10 demonstrated that doxycycline-mediated inhibition of MMP13 activity after acute rotator cuff repair was associated with improved biomechanical and histological parameters of healing at the tendon-bone interface.
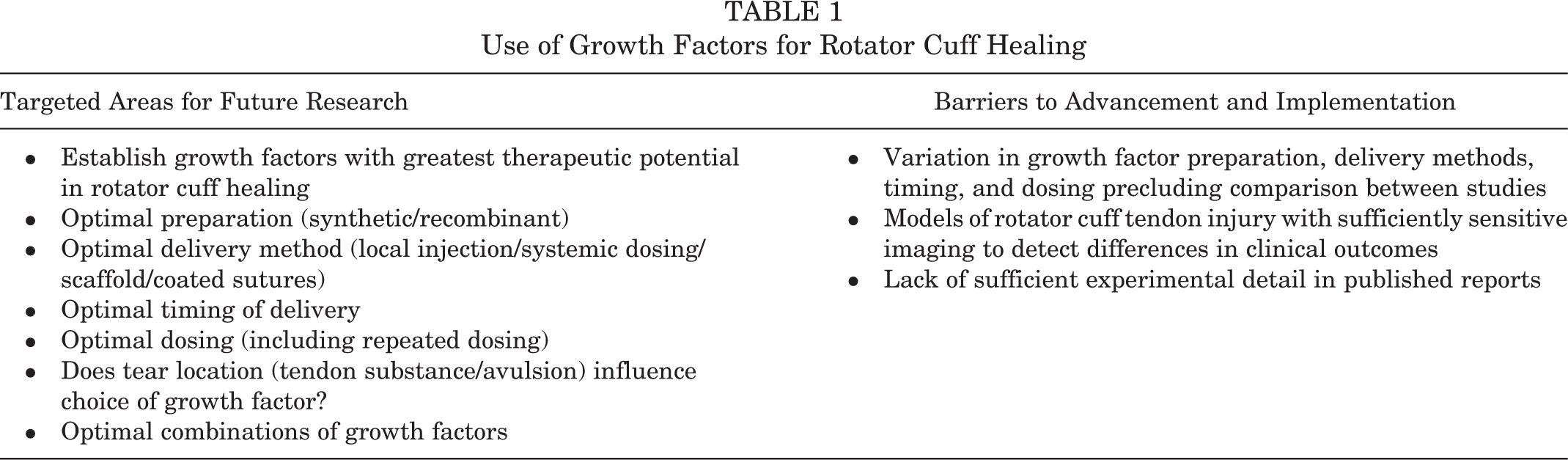
Targeted Areas for Future Research and Barriers to Advancement and Implementation
There is currently strong rationale and preliminary evidence to support a potential therapeutic role for growth factors after rotator cuff repair. However, within reported studies there is considerable variation in the preparation of growth factors (recombinant, synthetic), delivery methods (local injection, systemic dosing, within scaffold, coated sutures), timing (preoperatively, perioperatively, postoperative, and repeated dosing), and dose precluding comparison between studies. This is further complicated by potential interactions of these factors; for example, growth factors are rapidly cleared after local injection, but their persistence may be prolonged using scaffolds or coated suture material. 32 The optimal delivery method, timing, and dose will be different for each growth factor given the unique expression patterns of factors in normal healing and has not been explored in these studies. There is a danger that conclusions on the potential benefit of each growth factor are not supported by studies that explore each of these specific factors.
In vivo, growth factors work in concert and synergistically to create appropriate bone-tendon healing, and it is therefore intuitive that the most striking improvements in rotator cuff repair will be achieved through the delivery of multiple growth factors rather than by single growth factors in isolation. A number of preliminary preclinical studies support this notion. 56,121 In an in vitro model using rabbit flexor digitorum tenocytes, the greatest cell proliferation rates were achieved by combining bFGF, IGF1, and PDGF. This combination was more effective at lower doses than when the growth factors were delivered singly. 27 This is likely to vary depending on whether the repair involves tendon-bone healing or only tendon-tendon. Finally, the combination of growth factors may need to be individualized to the characteristics of each tear; for example, a tear with considerable bony avulsion may benefit from more osteoinductive cytokines than a midsubstance tendon tear. The design of studies that reflect the complexities of rotator cuff healing is an ongoing challenge. Critical areas for future research and barriers to advancement of growth factor–based therapies to treat rotator cuff injuries are summarized in Table 1.
Use of Growth Factors for Rotator Cuff Healing
Platelet-Rich Plasma
The use of PRP as a biological adjuvant to improve rotator cuff tendon healing has recently gained popularity. 89 Physiological tendon healing involves a complex interaction of cytokines, with some authors suggesting that no single growth factor is likely to be effective when delivered in isolation. PRP is a term used to describe preparations of whole blood enriched for platelets that, once activated, release a host of growth factors that may contribute to tissue repair. 3 Growth factors released by platelets are recognized to perform a wide range of regenerative functions including the proliferation and recruitment of stem cells, modulation of local inflammatory responses, and stimulation of new blood vessel formation. 55 A number of these growth factors have been shown to increase rotator cuff–derived tenocyte proliferation and promote the production of key extracellular matrix proteins including collagen types I, II, and X; decorin; aggrecan; and biglycan. 65 In addition, PRP provides protection against oxidative stress that can lead to cell apoptosis after injury 118 and inhibits the inflammatory effects of interleukin-1β (IL1β), which contributes to rotator cuff tendon degeneration. Conversely, TGFβ present in PRP positively contributes to rotator cuff tendon repair strength. 100 Similarly, neotendon formation may be induced by cytokines present within PRP including VEGF, PDGF, FGF2, TGFβ, IGF1, and BMP2. 2 PRP may enhance the self-healing potential of tissues by recruiting and activating resident progenitors. 63
Unfortunately, the use of PRP as an aid in arthroscopic repair of the rotator cuff has not met the expectations raised by in vitro studies. 65 Clinical trials using different autologous PRP formulations to augment rotator cuff tear repairs have yielded controversial results. 5,110,114,117 Appraisal of the literature is complicated by the range of treated injuries, varying from small single-tendon tears to massive multitendon tears; concomitant use of acromioplasty or not; and the formulation of PRP used, which ranges from intratendon injection, 67 spray application, 110 or incorporation of a platelet-rich fibrin matrix into the tendon repair site. 20 A summary of level 1 studies evaluating the role of PRP to augment surgical repair is shown in Table 2. Recent analyses evaluating all but 1 of the randomized controlled trials to date have reported that there was no significant difference in the rate of persistent tears between a PRP and control group. 21,111 However, stratified analysis of small or medium lesions showed a significantly lower persistent tear rate in the PRP group. Importantly, the use of PRP appears safe, with only 2 cases of infection and no complications reported in this meta-analysis. 111 Further prospective randomized controlled trials (level 1 evidence) are necessary to establish whether PRP has a role in the management of rotator cuff tears.
Summary of Level 1 Studies With Minimum 6-Month Follow-up Evaluating the Role of PRP to Augment Surgical Rotator Cuff Repair a

a ASES, American Shoulder and Elbow Surgeons; CTA, computed tomography arthrography; L-PRP, leucocyte-rich platelet-rich plasma; MRI, magnetic resonance imaging; PRP, platelet-rich plasma; P-PRP, pure platelet-rich plasma; QDASH, quick Disability of the Arm, Shoulder, and Hand; ROM, range of motion; SER, strength in external rotation; SF12, Short Form–12; SST, simple shoulder test; UCLA, University of California, Los Angeles; US, ultrasound; VAS, visual analog scale.
Targeted Areas for Future Research and Barriers to Advancement and Implementation
There is growing interest in establishing the optimal PRP formulation for rotator cuff healing. The most effective platelet and leukocyte counts and the balance between these counts, as well as the ratio with respect to plasma proteins, is not known. In particular, there is debate about which factor(s) are central to the regenerative effects of each formulation. While studies have indicated optimum platelet concentrations for bone regeneration (1,000,000 platelets/mm3), 143 there is currently no consensus about the best platelet concentration for rotator cuff healing and no comparative clinical study. In an in vitro study with tenocytes, Jo et al 65 showed that a concentration of 4,000,000 to 8,000,000 platelets/mm3 promotes higher cell proliferation. Different platelet-leukocyte ratios show a plateau effect of platelet concentrations, with increasing platelet concentrations being detrimental to extracellular matrix synthesis. 15 There is evidence to support that growth factors and cytokine concentrations are influenced by the cellular composition of PRP, with leukocytes increasing catabolic signaling molecules. 131 Increasing the platelet concentration within leukocyte-rich PRP preparations results in the delivery of more anabolic growth factors and decreased proinflammatory cytokines, but the biological effect on tendons is diminished metabolism, as indicated by a decrease in the synthesis of both collagen types I and III. 15 Together, this information suggests that minimizing leukocytes in PRP is more important than maximizing platelet numbers with respect to decreasing inflammation and enhancing anabolic signaling. However, other studies have pointed out the positive role of leukocytes in PRP as anti-infectious and immune regulatory agents. 92 Dragoo et al 33 reported that leukocyte-rich PRP led to an inflammatory response, although the clinical implications of this remains unclear. Three studies used leukocyte-rich PRP, with better results, 110 worse results, 53 or no difference in the PRP group. 23 Most clinical studies have used numerous different PRP formulations. The obtained results have never been analyzed to establish the influence of variable leukocyte content. Further randomized controlled trials comparing the effectiveness of leukocyte-rich PRP versus pure PRP will help in defining the optimal PRP formulation to manage rotator cuff injuries.
Similarly, no comparative study has evaluated the use of thrombin to activate the coagulation cascade in humans despite authors setting out arguments for 137 and against 120 this option. Virchenko et al 137 reported that the use of thrombin leads to improved outcomes in a biomechanical study in rat calcaneus tendons, while Scherer et al 120 demonstrated that PRP without thrombin achieved better results in the repair of skin defects.
A further variable affecting the efficacy of PRP is the timing of delivery. 119 The vast majority of previous PRP-augmented rotator cuff studies have delivered PRP at the time of surgical repair. 20,114 After activation, platelets release growth factors almost immediately with total elution within 1 hour, and the half-life of growth factors is a matter of minutes to hours. 93 Furthermore, each cytokine will contribute differently to the repair process at different time points after injury or repair. For example, PDGFs may have a more pronounced effect on tendon cellular maturation and biomechanical strength when delivered at 1 week compared with earlier delivery. 22 Hence, the timing for PRP delivery remains an important consideration for optimizing tendon healing and should be considered in future studies. To enable meaningful assessment of the effectiveness of PRP across studies, researchers must take care to characterize the composition of biologic compounds and standardize delivery protocols. Key areas for future research and challenges facing widespread implementation of PRP preparations to treat rotator cuff injuries are summarized in Table 3.
Use of Platelet-Rich Plasma for Rotator Cuff Healing a

a GFs, growth factors; MSCs, mesenchymal stem cells; PRP, platelet-rich plasma; RCT, randomized controlled trial.
Mesenchymal Stem Cells
MSCs hold great promise for rotator cuff tissue engineering because of their capacity to differentiate into bone, cartilage, and tendon cells and their ability to support a regenerative microenvironment through the release of trophic factors while tempering immune responses. 98 As such, MSC-based therapies may facilitate rotator cuff healing by differentiating into tenocytes or osteoblasts, by recruiting and stimulating progenitors, and by reducing inflammation. Studies demonstrating decreased resident MSC numbers in patients with rotator cuff tears provide further rationale for supplementing healing rotator cuff tendons with exogenous MSCs. 59 It is not clear whether patients with low resident progenitors are predisposed to rotator cuff tears or whether this reflects an exhaustion of the local progenitor pool after attempts at healing.
Human MSCs have been reported to be manipulated to differentiate into a tenogenic lineage and produce tendon tissue when exposed to the appropriate stimuli in culture. 83 Factors known to regulate the differentiation of MSCs into tenocytes include TGFβ3, scleraxis, BMPs, prostaglandin E, and the stimulus of a mechanical load. 24 Cells can be used alone or in combination with biocompatible scaffolds and growth factors, which are delivered intraoperatively to the site of tissue damage. A number of studies in animal models have evaluated the use of MSCs to improve healing after rotator cuff repair. 51,71,146 Although a number of clinical studies have evaluated MSCs in the context of rotator cuff repair, no level 1 studies have yet been performed. 35,58
MSC Sources and Purification
Numerous sources of MSCs, methods of cell purification, and formats of delivery are now available and have been summarized in part 1 of this series. 76 Although the ideal preparation and method of delivery of MSCs is not clear, it is important to appreciate the potential advantages and pitfalls of each.
Bone marrow and fat remain the most popular source of MSCs in studies evaluating rotator cuff regeneration. However, a number of studies have explored sources of MSCs within the shoulder in an attempt to avoid the requirement for a distant surgical procedure (eg, pelvic bone marrow aspirate or lipoaspirate). Beitzel et al 11 demonstrated that arthroscopic bone marrow aspiration from the proximal humerus consistently yields reliable concentrations of MSCs. Utsunomiya et al 136 isolated and characterized MSCs from 4 shoulder tissues during arthroscopic rotator cuff repair: synovium of glenohumeral joint, subacromial bursa, rotator cuff tendon, and enthesis at greater tuberosity. They concluded that the subacromial bursa may be a good candidate for the source of MSCs in rotator cuff surgery. Song et al 129 isolated MSCs from bursa tissue associated with rotator cuff tendons and confirmed their capacity for multilineage differentiation in vitro and in vivo. Unfortunately, the limited number of cells available from these sources has so far restricted their application in a nonexpanded format. 96
Preclinical Studies
The use of MSCs to enhance tendon regeneration has been examined in several animal models of tendon injury, although the number specifically evaluating their role in rotator cuff tendon repair is limited. 50,146 In a case-control study on 80 rats, Gulotta et al 50 reported that the injection of bone marrow–derived MSCs (BMSCs) at the time of rotator cuff repair did not improve the histologic or biomechanical properties of the tendon attachment site. The authors concluded that concomitant delivery of cellular and/or molecular signals to induce appropriate differentiation of the transplanted cells was probably required. Kim et al 71 compared the use of a tridimensional open-cell polylactic acid scaffold with and without BMSCs as an augment to rotator cuff repair. They reported the persistence of implanted cells to at least 6 weeks, with BMSC implanted repairs having greater type I collagen expression when compared with acellular controls. Yokoya et al 146 reported improved mechanical strength and an increase in type I collagen production at 4 months in rat rotator cuff defects managed with a polyglycolic acid scaffold seeded with BMSCs also in a rat model.
Clinical Studies
Only 2 studies have evaluated the application of MSCs in rotator cuff surgery. Ellera Gomes et al 35 investigated the effects of bone marrow mononuclear cells (BMMCs) on rotator cuff healing in 14 patients with complete tears. Although no control group was used in this study, the authors concluded that delivery of BMMCs was safe and had potential to enhance rotator cuff regeneration. In a case-control study of 45 patients, Hernigou et al 58 reported significant improvement in healing outcomes at 10 years in patients receiving MSCs as an adjunct therapy in standard of care rotator cuff repair. In this study, injection of MSC as an adjunctive therapy enhanced the healing rate and improved the quality of the repaired surface as determined by ultrasound and magnetic resonance imaging (MRI).
Microfracture
Microfracture of the greater tuberosity, just lateral to the site of rotator cuff repair, results in a large clot containing MSCs, platelets, and growth factors that has hence been referred to as a “crimson duvet.” 127 Microfracture awls are utilized to perforate into the cancellous bone enabling marrow to ooze out to cover the rotator cuff repair. This clot acts a reservoir of growth factors and a surface to guide the migration and proliferation of progenitor cells. 84 This method is inexpensive and technically straightforward, but as with nonpurified MSC preparations, its efficacy may be limited by cell heterogeneity inhibiting MSC differentiation. 97
In a randomized controlled trial of 57 patients undergoing arthroscopic rotator cuff repair, Osti et al 104 reported that microfracture at the rotator cuff footprint resulted in reduced short-term pain. However, augmentation with microfracture did not result in significantly different longer term outcomes, clinically or at imaging, compared with traditional rotator cuff repair. 104 In a further prospective randomized study of 80 patients evaluating greater tuberosity microfracture augmentation of rotator cuff repair, Milano et al 88 found no significant difference between groups on MRI analysis of structural integrity. However, patients receiving microfracture for large tears demonstrated significantly improved healing rates.
Gene Therapy
Gene therapy involves the insertion of genetic material into a target cell so as to manipulate the behavior of that cell. 79 Understanding the expression profile of growth factors central to the development of the normal enthesis in development might reveal candidate cytokines that could be used to augment the tendon repair site. 39 The majority of growth factors are short-lived within the site of injury, thus requiring repeated dosing if direct delivery is to be effective. 79 A key advantage of gene therapy is the potential for continuous production of the growth factor or cytokine by cells that become established within the wound. 79 In ex vivo gene therapy, cells are harvested, cultured, and transduced with the vector in culture prior to reimplantation. 138 Gene therapy approaches in rotator cuff repair have been used to manipulate expression of scleraxis, BMP13, and platelet-derived growth factor receptor–β (PDGFRβ). 51,52,135
Scleraxis is a transcription factor necessary for tenogenesis within the developing enthesis. 68 In a rat model of rotator cuff repair, BMSCs transduced with adenoviral-mediated scleraxis were applied to the tendon-bone repair site. 51 Animals receiving MSCs transduced with scleraxis had greater stiffness and stress to failure and a significantly increased load to failure at 4 weeks after repair. 51 Although there were no histological differences between experimental and control groups at 2 weeks after repair, the insertion site of the scleraxis group had more fibrocartilage that more closely resembled the native tendon-bone insertion site. Also in a rat model, the same group examined the ability of BMSCs transduced with human BMP13 to improve rotator cuff healing in a rat model. 52 There were no differences in new cartilage formation, collagen fiber organization, or biomechanical strength at the repair site between experimental and control groups at 4 weeks after repair.
Uggen et al 135 transduced rat tendon fibroblasts with IGF1 or PDGFRβ via retroviral vectors. These cells were expanded in culture and delivered on a bioabsorbable scaffold over a supraspinatus repair. Animals that received the PDGFRβ overexpressing cells showed near complete restoration of the tendon, significantly better than suture repair alone. 135 These data indicate that it may be possible to manipulate and deliver growth factors to promote a regenerative response.
Targeted Areas for Future Research and Barriers to Advancement and Implementation
A key area of future research must be to identify the mechanisms by which the native cells within the rotator cuff tendon and surrounding tissues contribute to rotator cuff healing and the formation of scar tissue. There has been an increasing appreciation that MSCs can become activated myofibroblasts, contributing to tissue fibrosis after severe or repetitive injury. 75 The identity of cells contributing to repair of normal enthesis prenatally and the identity of cells contributing to scar formation after injury have yet to be identified. Modern mouse genetic tools now facilitate cellular fate mapping in a diverse range of biological processes. 112 The Cre/loxP system, which is widely used for this purpose, employs the gene for bacterial Cre recombinase (causes recombination: an enzyme that can perform site-specific knockdown of genes), which is linked to a cell- or lineage-specific promoter prior to incorporation in the genome of a transgenic mouse. 49 Studies are urgently required that utilize fate mapping to establish the role of resident cells in the response to rotator cuff injury and repair.
The vast majority of studies exploring muscle, bone, and cartilage regeneration with MSCs in which the fate of delivered MSCs have been assessed have reported that only a fraction of the delivered cells can be found at the site of repair beyond the acute phase. 41,94 The majority of authors have concluded that one of the many trophic factors released by MSCs must be responsible for their therapeutic effect, but no definitive studies have identified this elusive factor. 41,94 It is not clear whether the presence of MSCs is required for the therapeutic effect or whether the delivery of the responsible factor(s) in isolation could be a further therapy. Approaches to address this question may include the delivery of fluorescently labeled cells to delineate their fate and the delivery of MSCs with the systematic knockdown of potential target molecules.
For therapies involving the application of MSCs; the ideal stem cell source, preparation, and number; and the optimal timing of delivery remains largely unexplored but is critical to the translation of this technology. One potential advantage of allogeneic over autologous cells is the possibility of their use as an “off-the-shelf” therapeutic agent, avoiding the need for bone marrow aspiration and tissue culture delays before treatment. Furthermore, some studies indicate that the function of MSCs in patients with advanced age or significant comorbidities is impaired (including impaired differentiation potential and senescence). 66 Allogeneic cells may therefore be of particular benefit in the elderly or those with multiple comorbidities. Further studies are required to establish the most favorable site of MSC harvest and whether allogeneic or autologous cells are favorable.
It is not clear from the limited studies to date what the optimum number of cells for each indication is or whether there is a critical number of cells to exert therapeutic benefit. 32 It is intuitive that the optimal number of cells will vary relative to the size of the rotator cuff defect, the cell type used, and the particular region in need of repair. MSCs can be exposed to differentiation factors in culture to begin the process of tenocyte differentiation prior to implantation. 80 Future studies should establish whether this is beneficial to the delivery of naive MSCs. Similarly, growth factors that induce directed differentiation or promote a regenerative microenvironment can be delivered simultaneously. The optimal combinations of MSC and growth factors for rotator cuff repair should be a focus of future research efforts. Potential off-target effects of these factors on local cell populations should be explored, as well as adverse systemic effects.
Having established that a reservoir of MSCs resides in almost every tissue, 29,97 the question of whether therapeutic strategies could be employed to “recruit” or accelerate the regeneration capacity of resident MSCs after injury should be considered. The “activation” of adult stem cells is known to be regulated, in part, by their neighboring cells; manipulation of this communication through the local infiltration of growth factors or molecules integral to this communication may represent such a therapeutic strategy. 96
In the future, it may be possible to use alternative sources of stem cells such as induced pluripotent stem (iPS) cells, embryonic stem cells (ESCs), or other recently identified adult stem cell populations. MSCs have been generated from both iPS cells and ESCs, raising the possibility of direct delivery of these more primitive populations. 26 Tendon-derived stem/progenitor cells (TSPCs) can differentiate, form tendon-like tissue, and have the ability to synthesize tendon extracellular matrix. 12 However, no studies using TSPCs to augment rotator cuff repair have so far been reported. The native origin of these cells is interesting given the presumed perivascular origin of mesenchymal progenitors in organs and the avascular nature of tendons. 29 A population of muscle-derived stem cells (MDSCs) has been derived using a modification of the preplate technique that uses the adhesion characteristics of the cells to collagen-coated flasks. 47 In vivo studies have demonstrated that MDSCs differentiate into multiple lineages, self-renew, and regenerate bone, cartilage, muscle, blood, and cardiac tissue. 18 MDSCs injected into supraspinatus tendons of athymic rats incorporated into host tissue with a morphology comparable to resident tendon fibers. 108 However, the ability of MDSCs to regenerate tendon after injury remains unclear. Keys areas for future research and critical barriers to advancement of MSCs for rotator cuff healing are summarized in Table 4.
Use of Mesenchymal Stem Cells for Rotator Cuff Healing a

a FACS, fluorescence-activated cell sorting; GFs, growth factors; MSCs, mesenchymal stem cells.
Patches
Augmentation of rotator cuff repair with patches can result in improved clinical outcomes compared with nonaugmented repairs. 8,36 Patches can be nondegradable, based on xenogeneic or allogeneic extracellular matrix (ECM) constructs, or degradable scaffolds that can be combined with stem cells, matrix proteins, and growth factors. There is currently no clear consensus as to what is the most efficacious patch strategy and whether their use is safe in the long term. Augmentation with nondegradable structures aims to provide permanent mechanical support to facilitate self-healing. Nondegradable patches require good strength and tissue compatibility and have been made from materials, including polycarbonate polyurethane, polytetrafluoroethylene, and polyester. To encourage tissue ingrowth, these materials are typically processed into foam or felt allowing cells to penetrate the surface and incorporate. Concerns include the occurrence of persistent infections and loss of integrity over time, where fragmentation may lead to mechanical failure or chronic inflammation. Studies evaluating restoration of massive rotator cuff defects with synthetic grafts have reported significant short-term improvements in pain and function. 7,99
ECM-based scaffolds aim to provide a temporary, collagen-based matrix to facilitate integration of local cells. ECM-based patches extracted from porcine intestinal mucosa (SIS), porcine dermis, human fascia, and human dermis are available. Despite performing well as substrates for tendon-derived cells in vitro, in vivo studies have produced conflicting results. 109,149 Concerns related to ECM-derived scaffolds include poor suture retention, limited mechanical properties, and toughness. 25,149 Traces of DNA and cellular content have also been found in some ECM-derived patches, raising a theoretical risk of disease transmission or an adverse inflammatory response. 48 Despite these issues, many ECM-derived patches are approved by the Food and Drug Administration and are commercially available. Porcine dermal collagen and SIS have both been used effectively to provide strength and support when connective tissues have been lost. 105,116,124 However, clinical data supporting their use for rotator cuff augmentation in humans are limited. 102 Using a porcine collagen dermal patch, Badhe et al 8 demonstrated significant functional improvements after augmented rotator cuff repair in a study of 10 patients, with 80% graft patency on MRI at a mean 4.5 years. Soler et al 128 investigated the ability of porcine dermal collagen to serve as an augmentation material in massive rotator cuff repair. Ultimately, all patients demonstrated signs and symptoms of a recurrent tear despite this treatment. Sclamberg et al 122 found that SIS was ineffective at reinforcing large and massive rotator cuff tears, with 10 of 11 patients having recurrent defects within 6 months. Furthermore, Iannotti et al 60 recommended against using porcine SIS for augmentation of large and massive rotator cuff tears after finding reduced postoperative functional scores in a randomized controlled study. Barber et al 9 performed a randomized controlled trial to compare arthroscopic single-row repair with or without acellular human dermal patch (commonly known as Graftjacket) augmentation. Augmented repairs had significantly greater American Shoulder and Elbow Surgeons (ASES) and Constant scores then nonaugmented repairs at 24 months. On MRI at 14.5 months, 85% of patients in the augmentation group had intact repairs compared with 40% in the control group. Unfortunately, studies funded by different companies have produced contradictory results and large, long-term, and impartial comparative trials are required. Overall, the efficacy and safety of using ECM-derived patches remains uncertain.
Degradable synthetic scaffolds provide a nonpermanent support for intrinsic or implanted progenitors while addressing issues relating to cost and potential disease transmission relating to ECM-based scaffolds.
54
The nonpermanent support aims to encourage cellular ingrowth and tendon self-healing. Degradable polyesters include poly-
A polyglycolic acid sheet cultured with autologous BMSCs has been used to repair resected infraspinatus tendons in rabbits. 145 An increased production of type I collagen and an increment of the mechanical strength was seen 4 months after repair as compared with both a nonaugmented control and a nonloaded scaffold group. Kim et al 71 harvested and expanded BMSCs from rabbits prior to seeding on a polylactic acid scaffold. This study showed that MSCs survived for 6 weeks within the scaffold, and type I collagen expression was increased in the scaffold with BMSCs. Shen et al 126 used a knitted silk-collagen scaffold loaded with allogeneic Achilles tendon stem cells to augment a rotator cuff repair in rabbits. Increased fibroblastic cell ingrowth and reduced infiltration of lymphocytes were observed at the repair site at 8 weeks, while structural and mechanical proprieties were improved at 12 weeks as compared with controls.
Several studies have focused on re-creating the complex tendon-bone interface.130 Spalazzi et al 130 showed that fibroblasts and osteoblasts initially confined to soft tissue and bone formation areas in a triphasic scaffold migrated into an intermediate layer leading to increased production of a type I collagen matrix. Moffat et al 91 investigated the use of a biphasic nanofiber scaffold containing mineralized and nonmineralized regions to augment rotator cuff repairs in rats. They demonstrated viable chondrocytes with collagen/proteoglycan matrix in both phases of the scaffold. Rats repaired with the scaffold showed an organized fibrocartilage-like transition zone. 91
Targeted Areas for Future Research
The technology of patches has greatly developed in recent years, but this has not yet translated into functional improvements for patients. 54 The concept of patches as a static, mechanical support that is biologically inactive is now waning. Newer generations of predominantly degradable synthetic patches are emerging that seek to combine targeted mechanical properties with stem cells or growth factors. 54 These patches are designed to completely absorb in a timely manner so as to minimize risk of infection. However, this strategy has yet to be proven in clinical trials, and future research must concentrate on comparative clinical trials of the commercially available patches. While industry involvement is integral to the development of patches, evaluative studies must take place in more neutral, academic settings.
A recurring theme throughout the literature is concern regarding potential adverse effects of synthetic degradation products. 87 Toxicities appear to vary between polymers, and there are currently no data relating to the shoulder. Future studies should evaluate the release of degradation products until the implant is completely degraded to definitively establish whether levels of acidic products released remain within safe levels. Key areas for future research and critical barriers to advancement of patches in the repair of rotator tears are summarized in Table 5.
Use of Patches for Rotator Cuff Healing

Biologic Strategies to Augment Healing in Rotator Cuff Muscle
Muscle atrophy and fatty degeneration are recognized factors implicated in rotator cuff repair failure, yet there is a paucity of work dedicated to addressing this process. Muscle atrophy and fatty degeneration decrease the compliance of the muscle-tendon unit and lead to rotator cuff retraction and increased tension at the repair site. 45
Anabolic Steroids
Anabolic-androgenic steroids are synthetic derivatives of testosterone that act on androgenic receptors to exert their anabolic effects. 37 At supraphysiologic doses, they increase muscle size and strength, promote collagen synthesis, and increase bone mineral density. 37 In a rabbit model of chronic rotator cuff tears, Gerber et al 44 demonstrated that administration of nandrolone decanoate reduced fatty infiltration within the supraspinatus muscle and prevented muscle impairment resulting from myotendinous retraction. While anabolic-androgenic steroids represent a promising strategy to temper muscle deterioration after tendon tear, they are recognized to have potentially serious adverse effects. 132 As immunosuppressants, steroids potentially increase susceptibility to infection, and the long-term health risk of their use in not known. Furthermore, anabolic-androgenic steroids are considered as performance-enhancing drugs, and their use is banned in athletic competitions. Further studies are required to establish whether anabolic-androgenic steroids have a role in rotator cuff healing.
Targeted Areas for Future Research
The inherent myogenicity of MSCs makes them an attractive substrate for skeletal muscle regeneration or augmentation, and MSCs have been demonstrated to differentiate into myotubes and regenerate skeletal muscle after cardiotoxin injury. 29 Furthermore, they release a host of trophic factors known to contribute to tissue regeneration. In addition to their proangiogenic effects, MSCs reduce hypoxia-driven angiogenesis. 64 It is conceivable that MSCs isolated from lipoaspirate at the time of rotator cuff surgery could be delivered during repair to improve postoperative muscle function and compliance. Conversely, this strategy could be used in a staged procedure to treat atrophic muscle prior to repair, improving myotendinous compliance and diminishing tension at the repair site. Key areas for future research and critical barriers to advancement of biologics in the regeneration of rotator cuff muscle are summarized in Table 6.
Use of Biologics in Rotator Cuff Muscle Regeneration a

a MSC, mesenchymal stem cell; TGFβ, transforming growth factor–β.
Conclusion
Rotator cuff tears often result in considerable morbidity and extended periods of sporting absence. Recent advances in understanding the pathophysiology of rotator cuff healing and the role of growth factors and progenitors have highlighted the potential role for biologics to augment surgical repair. Animal studies indicate that delivery or modulation of growth factors may provide a useful way of improving the biology of rotator cuff healing, although human studies to support this are currently lacking. The use of PRP in a number of recent randomized controlled trials of surgical rotator cuff repair has not met the expectations raised by in vitro studies. Clinical studies evaluating the use of MSCs in surgical rotator cuff repair show great promise, but randomized clinical trials are lacking and considerable work is required to establish the true mechanism by which these cells contribute to rotator cuff regeneration. With all the biologic approaches to rotator cuff repair discussed, considerable variation in experimental techniques and methods of production have so far limited meaningful interpretation of the literature. Currently, there exists a lack of consensus on the optimal preparation, source, delivery method, and dosing of biologics for orthopaedic applications. The problem is exacerbated by a lack of sufficient experimental detail in many publications, which impairs readers’ ability to evaluate the quality of results or compare results across studies. A collaborative approach to biologics research is required with standardization of techniques, guidelines for the minimum reporting of experimental details, and large multicenter randomized controlled studies.
Footnotes
One or more of the authors has declared the following potential conflict of interest or source of funding: The 2015 American Orthopaedic Society for Sports Medicine (AOSSM) Biologics II Think Tank was sponsored by RTI Surgical. The outcomes and opinions from this Think Tank are solely those of the authors and do not reflect any influence from the sponsor. R.F.L. is a consultant for and receives royalties from Arthrex, Ossur, and Smith & Nephew.
