Abstract
Objective
Adverse drug reactions (ADRs) induce iatrogenic harm in antiretroviral therapy (ART) care continuum. However, there is a dearth of concrete evidence in a resource-limited setting. Thus, this study was designed to consolidate existing knowledge, thereby informing policy and clinical care to improve patient safety.
Design
Systematic review and meta-analysis.
Data sources
PubMed, CINAHL, Web of Science, and EMBASE databases were searched.
Eligibility criteria
Employing the condition, context, and population framework, observational primary studies were included.
Data extraction and synthesis
Independent reviewers undertook data extraction and synthesis. This meta-analysis employed the random-effects restricted maximum likelihood (REML) method, with its protocol preregistered on the International Register of Systematic Reviews (CRD42024546390).
Results
The pooled prevalence of ADRs was 36.7% [95% CI: 26.6-46.9, I2 = 99.64%].
Conclusion
Altogether, this study revealed that ART-related ADRs in Ethiopia was 36.7%, underscoring rigorous monitoring. Giving special emphasis to patients with female gender, advanced disease, comorbidities, malnutrition, TB treatment, and poor adherence is a prudent decision.
Plain Language Summary
Medicines are crucial in the arena of the healthcare system. Despite the undeniable relevance of medicines, a substantial challenge is the potential for harmful drug reactions when used. Antiretroviral therapy (ART) is the application of antiretroviral medicines to combat human immunodeficiency virus (HIV). While it is pivotal in treating diseases, it is plugged with adverse drug reactions (ADRs). ADRs are harmful effects that can occur when people take medications. These undesirable drug effects are quite common and especially costly in the treatment of HIV. In countries with limited healthcare resources, there is limited information about how common these harmful effects are. This study aimed to bring together the available evidence about the rate of ADRs related to ART among people living with HIV in Ethiopia. In this study, we reviewed the results of 29 studies involving 19,003 people living with HIV. We found that about 4 in every 10 patients (36.7%) experienced ADRs from their treatment. Our analysis also showed that certain groups, including women, those with lower education levels, advanced HIV disease, other chronic health problems, weaker immune systems (low CD4 counts), opportunistic infections, malnutrition, and those taking medications containing zidovudine or nevirapine, were more likely to face these ADRs. Patients receiving tuberculosis treatment at the same time or struggling with medication adherence were also at higher risk. Based on these findings, special attention should be given to monitoring these vulnerable groups to catch and manage ADRs early.
Keywords
Background
Antiretroviral therapy (ART) involves the application of antiretroviral drugs (ARVs) to combat human immunodeficiency virus (HIV). 1 It is employed as a pivotal component of a multimodal approach in the management of HIV. Antiretrovirals are trustworthy in improving longevity and quality of life.
Yet, ART-related adverse drug reactions (ADRs) continue to be a healthcare concern. ADR was defined as “a response which is appreciably noxious and unintended, and which occurs from an intervention related to the use of a medicinal product at doses normally used in humans for the prophylaxis, diagnosis, or therapy of disease or for the modification of physiological function.” 2 It poses substantial healthcare and financial toxicity. 3 ADR-related hospitalization ranges 1.1-26.5% in developing countries. For instance a thesis by Moirana 4 in Nigeria revealed that ART-related hospitalization was 26.5% (95% CI: 18.4, 35.4). The number of years of life lost due to medication-related ADRs is twice as high for patients living in developing nations as for those in wealthy nations. 5 Furthermore, ADR-related mortality reaches 1.7% (0.7-4.8) and 1.8% (0.8-8.0) in developed and developing countries, respectively. 6 In agreement with this finding, a study by Moirana 4 reported an ART-related mortality of 6.1% (95% CI: 2.1-11.7).
People living with HIV (PLWHIV) were accompanied by significant safety concerns owing to life-long treatment, multimorbidity, polypharmacy, and medication regimen complexity.6,7 The global image of ART-related ADR was huge for instance 85.5% reported in Brazil8% and 94% from Iran. 9
In Africa, ARVs are the leading cause of ADRs, according to Ampadu et al. 10 ART-related ADRs in Africa was wide in range.11,12 Despite highly burdened with HIV/AIDS, there are few reports in the literature from sub-Saharan Africa (SSA) regarding antiretroviral-induced ADRs. For instance, a hospital-based cross-sectional study carried out among 312 PLWHIV on highly active antiretroviral therapy (HAART) attending HIV clinics at Mbarara Regional Hospital in Uganda revealed 76.60% ART-related ADRs, 11 62.8% in Eretria, 13 and 42.3% in Mali. 14 In Ethiopia, the prevalence of ART-induced ADRs spans from 7.72% to 85.78%.15,16 The most commonly occurred ART-related ADRs are metabolic effects, dermatologic effects, gastrointestinal toxicity, cardiovascular toxicity, neurological effects, and hematologic toxicities. 17
Risk factors for ADRs in PLWHIV were multitypes. The most common predictors includes female gender, rural residency, limited or no formal education, economic hardship, pregnancy, substance use, advanced World health organization (WHO) clinical stages (III and IV), opportunistic infections (OIs), chronic disease comorbidity, high level of treatment regimen complexity index, suboptimal adherence.18–20
Ethiopia implements precautionary measures based on pharmacovigilance evidence to protect the general population from avoidable assaults, 21 as 60% of ADRs are preventable. 22 Recently, the contemporary and recommended dolutegravir (DTG)-based first-line regimen has been implemented in Ethiopia. This policy change expands access to a new ARV, which may lead to significant ADRs. Despite prior research has touched upon ART-related ADRs, significant limitations persist. 23 Notably, existing studies have not primarily focused on elucidating the specific risk factors driving ADRs. Furthermore, issues such as limited article inclusion, potentially introducing selection bias and restricting the generalizability of findings, alongside incomplete statistical analyses, specifically the absence of meta-regression, constrain the robustness of current conclusions. Consequently, conclusions directed at policymakers regarding ART-related ADR mitigation strategies may lack the necessary foundation due to an incomplete understanding of the critical underlying risk factors.
Hence, the current study aimed at determining the epidemiology of ART-related ADRs and its predictors among PLWHIV in Ethiopia. By addressing the methodological limitations of prior research and focusing on high-quality articles, this work ultimately seeks to enhance the tolerability and effectiveness of ART in this setting. This focused approach would provide more robust and pragmatic insights for policymakers to develop targeted and effective strategies for preventing and managing ADRs in individuals undergoing ART. Furthermore, this finding may inform international research priorities and collaborative efforts aimed at improving ART tolerability and effectiveness worldwide.
Methods
Review Protocol
In order to ensure methodological rigor, a detailed review protocol was developed and Preregistered in the International Register of Systematic Reviews (PROSPERO) database with a reference number of CRD42024546390 before study commencement. The Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) checklists were used for conducting and reporting process 24 (PRISMA checklist 2020).
Study Design and Setting
This study was a systematic review and meta-analysis conducted in Ethiopia to estimate the prevalence of ART-related ADRs and its predictors among patients living with HIV/AIDS. Ethiopia is one of the developing countries in East part of Africa. It has 12 regions and 2 city administrations. The regions are Tigray, Afar, Amhara, Oromo, Somali region, Benshangul gumz, Central Ethiopia, Sidama, South West Ethiopia, South Ethiopia, Gambela, and Hareri. The two city administrations are Addis Ababa and Dire Dawa. 25
Eligibility Criteria
Despite the traditional approach to inclusion criteria for systematic reviews of effects is to follow the population, intervention, comparator, and outcome (PICO) structure, condition, context, and population (CoCoPop) framework was employed in this study since PICO approach clearly does not align with questions relating to prevalence. 26 The inclusion criteria were determined using the CoCoPop components: Condition: epidemiology (prevalence, characteristics) of ART-related ADRs. Context: Healthcare institutions (inpatient or outpatient departments) in Ethiopia. Population: Patients diagnosed with HIV/AIDS and receiving ART regardless of age group and gender distribution. Studies: Observational primary studies including cross-sectional, cohort and case-control studies. Publication status: Published or gray literatures (if rigorous). Time period: Studies conducted from inception of database to July 30 2024. Language: Published in any language. Associated Factors: Sociodemographic, clinical and treatment-related predictors. Due to the lack of widely accepted gold standard tools, it was necessary to include studies that assessed causality, severity, or preventability, regardless of the methodology they employed.6,27
Duplicate publications, incomplete studies, studies did not report the outcome of interest and studies conducted outside Ethiopia, not focusing on ART-related ADRs, concentrated on ADRs unrelated to the treatment of HIV, conference abstracts without full text, dissertations not publicly available, and were review articles were excluded.
Information Sources
Four electronic databases were queried using standard indexing procedures. Databases including the CINAHL, EMBASE, MEDLINE (via PubMed), and Web of Science were searched by authors. The citations of original publications and related review articles discovered in the database search were also obtained manually. Google and Google Scholar were used to obtain gray literature.
Search Strategy
PubMed (Medline), EMBASE, and CINAHL were searched with standardized technical medical subject heading search terms (MeSH terms), exact Emtree terms, CINAHL Subject Headings and free-text terms (key words) as shown in Supplemental material 1, which enables this systematic search to be transparent and reproducible. We have approached databases with the following search terms: “Epidemiology of ART-related problem,” “Prevalence of ART-related problem,” “Epidemiology of drug therapy problem,” “Drug-related side effects and adverse reactions,” “medication related problem,” “ARVs-related drug reaction”, “highly active antiretroviral therapy-related reaction”, “risk factor”, “associated factors”, and “predictors.” The search results were combined using Boolean operators (“OR” and “AND”). Each database's search results were recorded in its own electronic database, and then exported into the bibliographic software Endnote software version 20, Thomson Reuters, New York, NY. 28 The Endnote library was immediately updated with all the studies found through manual searches. The searches spanned from 09 April to till July 30 2024.
Study Selection
Study screening and selection was conducted meticulously by two investigators (W.S.Z. and S.B.D.) independently. All articles that were found were evaluated for eligibility according to the PRISMA standard.
At first, all the imported articles’ title suitability was assessed aimed at selecting only those studies that address our review question and match the inclusion screening criteria as prespecified in the protocol. Duplicate citations from other databases were eliminated. Afterwards, the abstracts were assessed using the same criteria by those authors. If the title and abstract evaluation do not yield a conclusive judgment, the inclusion and exclusion criteria were employed to evaluate the full texts of the article following retrieval. The inclusion decision for each article was recorded in paper format, indicating the reason for each article's inclusion or exclusion. Discrepancies were handled by discussion and consensus. The PRISMA 2020 flow diagram was employed to summarize the overall study selection processes.
Data Collection Process
In accordance with the PRISMA statement 29 a standardized paper data abstraction sheet was created by modifying and customizing the “Data collection form for intervention review-RCTs and non-RCTs” from the Cochrane Collaboration. 30 Subsequently, the included articles underwent a pilot test to ensure comprehensive capture of all pertinent data as well as to ascertain that the level of interobserver deviation between the two reviewers is satisfactorily minimal. After the items of the data abstraction tool have been evaluated, they were entered into the Microsoft Excel 2016 software spreadsheet. Data extraction was performed by the W.S.Z and T.A.M. Any divergence of viewpoint was duly acknowledged and addressed through consensus between the two reviewers, or alternatively, S.B.D was served as an arbitrator.
Data Items
All relevant data, including demographic data (eg, sex, age), first author, year of publication, region, study design, study population, study settings, sample size, the number of patients with ART-related ADRs, ADR definition employed, tools for causality, severity, and preventability assessment, data source, prevalence of ADRs and predictor variables were extracted.
The standard error for the prevalence values was calculated using a standard formula:
where P is the prevalence of the study n and n is the sample size of the study.
Risk factors influencing ADRs were systematically assessed by extracting and calculating the adjusted hazard ratio (AHR), lower and upper confidence limit (LCL, UCL), as well as their natural logarithmic transformations (lnAHR, lnLCL, lnUCL) and the standard error of lnAHR (SElnAHR) for each variable, before exporting the data into STATA for analysis.
LCL was calculated employing equation below:
The SElnAHR was computed employing the equation below
At a 95% confidence interval (CI), the critical value for the standard normal distribution (Zα/2) is 1.96.
Study Risk of Bias Assessment
In advance of collecting the data, the studies that met the inclusion criteria were criticized for their quality. Right now, there is a lack of consensus over the methodology for evaluating the validity of observational epidemiological studies. 31 The Joanna Briggs Institute (JBI) critical appraisal checklists for cross-sectional and cohort studies were utilized as quality indicators to examine the potential bias in the included studies.31,32 The instruments consist of 8 and 11 criteria, each with corresponding options of “Yes,” “No,” “Unclear,” and “Not applicable,” respectively. The two reviewers (W.S.Z and M.A.A) were separately appraising the research they had read for bias and recording their findings because some of the items covered may need some discretion. Any disagreement in the bias assessment was discussed and resolved by consensus between the two reviewers; if it persisted, a third reviewer (S.B.D) arbitrated and verified the appraisal. The average score of the two investigators determined the inclusion of each article, with studies scoring 50% or higher being included. Overall scores range from 0%-50%, 50%-70%, and 70%-100%, would be adjudicated as high, moderate, and low risk of bias, respectively. Aside from the initial qualifying criterion, the minimum standard that must be met is that the ADRs are defined in accordance with the definition provided by the WHO or a comparable definition.
Effect Measures
The primary effect measure for this systematic review was the prevalence proportion of ADRs among people living with HIV (PLWHIV). This measure would quantify the proportion of individuals within the study populations who experience one or more ADRs. As a secondary outcome, we sought to extract and synthesize hazard ratios (HRs) when reported. HRs would serve as a ratio measure to assess the relative risk of developing ART-related ADRs over time, allowing for an understanding of the instantaneous risk factors associated with different ART-related ADRs.
Data Synthesis
All studies included in the review were described in a table comprising their main characteristics and outcome measures. The extracted data in Excel were exported to the software STATA (StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC) for analysis. 33 Considering the clinical, methodological, and statistical heterogeneity present in the included primary studies, a random-effects inverse-variance model with the restricted maximum likelihood (REML) variance estimator was employed to pool the prevalence of ADRs. 34 This approach aligns with the Cochrane Handbook for Systematic Reviews of Interventions (version 6.5), which emphasizes that the choice between a fixed-effect and a random-effects meta-analysis should not be based solely on a statistical test for heterogeneity. 35 Cochran's Q test of heterogeneity and I2 statistics were considered in this study in revealing the presence of heterogeneity between studies. A Cochrane Q value indicates significant heterogeneity among studies if its P < .05. I2 statistics predict low, moderate, and high heterogeneity when it is below 25%, between 25% and 75%, and above 75%, respectively. 36 Furthermore, heterogeneity assessment, was conducted using the symmetry of the forest plot. Subgroup analysis was conducted based on age groups, study design, and, in order to explore potential sources of heterogeneity and identify moderators that might influence the overall pooled estimates of the effect size. The source of variation was further investigated via univariate metaregression analysis. A sensitivity analysis was conducted to check the stability of the summary estimate following omitted individual studies.
Reporting Bias Assessment
Herein, to assess reporting bias, both qualitative and quantitative methods were employed since relying on a single test is not advised. 37 A funnel plot was used for qualitative assessment, visually inspected for asymmetry. For quantitative evaluation, Begg's correlation test 38 and Egger's regression test 39 were utilized. In both Begg's and Egger's tests, a P-value > .05 indicated no statistically significant evidence of publication bias. In light of the possible drawbacks of conventional approaches, particularly the fact that funnel plots might not be accurate for proportions 40 and that Begg's and Egger's tests were underpowered, 41 DOI plots and LFK indices were also produced as supplemental measures to offer a more thorough examination of potential biases because of their improved qualitative and quantitative techniques in nature. 42 LFK index close to 0 (eg, between −1 and +1): indicates “absent asymmetry” or no evidence of publication bias, between >±1 and ≤±2: “minor asymmetry” or minimal publication bias, >±2: major asymmetry” or important/high publication bias. Publication bias would be handled by nonparametric trim and fill analysis using the REML model. 43
Certainty Assessment
In the era of evidence-based medicine, it is crucial to assess the degree of certainty in a given evidence. Herein, W.S.Z. and M.A.A. assessed certainty in the evidence independently based on guidance provided in the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) handbook using the five GRADE considerations (precision, consistency, publication bias, risk of bias, and directness). 44 The quality of evidence was downgraded if the CI spanned ≥10 percentage points, if the I2 upper CI was ≥75% (high statistical heterogeneity detected (I2 > 75%)), presence of high risk of publication bias, low level of JBI score or high risk of bias and serious indirectness. The evidence quality was divided into four categories (high, moderate, low, and very low). 45
Results
Study Selection
As per our preregistered protocol (PROSPERO ID: CRD42024546390), we have searched and identified 5197 primary studies from different databases. Our search could not find any relevant publications in other languages for the Ethiopian setting, although we are aware that excluding non-English literature can introduce publication bias. Four thousands and nineteen (n = 4019) duplicates were removed. Among 1178 studies eligible for title screening from PubMed (n = 609), CINAHL (n = 414),Web of Science (n = 54), EMBASE (n = 48), Google Scholar (n = 13), snow balling (n = 12), citation searching (n = 28), 1, 117 were excluded due to unrelated titles (n = 1003) and being a review articles (n = 114). Following abstract screening on 61 studies, 30 were excluded due to irrelevance to the research question. Ultimately, of the 31 studies assessed for eligibility, 29 satisfied the inclusion criteria and were included in the final qualitative and quantitative analysis where two were excluded due to full text inaccessibility 46 and due to absence of outcome of interest. 47 (Figure 1).

Preferred Reporting Items for Systematic Review and Meta-Analysis 2020 flow diagram of the study retrieval process.
Study Characteristics
Overall, the included studies were conducted among PLWHIV receiving ART at healthcare facilities across Ethiopia. As summarized in Table 1, all studies reported on the prevalence and patterns of ADRs. Collectively, the studies encompassed a total of 19 003 participants, of whom 11 056 (58.18%) were female. Study periods spanned from 2007 to 2024. Majority of the included studies were conducted in the Amhara region (n = 10).15,48–56 The rest were in southern Ethiopia (n = 6),12,57–61 Oromia (n = 5),62–66 Harari and Dire Dawa (n = 3),67–69 Addis Ababa (n = 2),70,71 and Tigray (n = 2)16,72 regions. The last study conducted by Gudina et al 73 was a multicenter study conducted in different regions of the country including Amhara, Addis Ababa, Oromia, Tigray and southern Ethiopia.
General Description of the Included Studies in this Systematic Review and Meta-analysis.

Note: Keys: AA, Addis Ababa; ACTG, AIDS Clinical Trial Group; Amh, Amhara; ALT, Alanine aminotransferase; DAIDS AE, Division of AIDS Adverse Event; Oro, Oromia; P, prevalence of ADR; SNNPR, Southern Nations, Nationalities and Peoples of Ethiopia; Tig, Tigray; PN, Peripheral neuropathy; SJS, Stevens-Johnson Syndrome; ART, antiretroviral therapy; HAART, highly active antiretroviral therapy; ARV, antiretroviral drug; HIV, human immunodeficiency virus; ADR, adverse drug reaction; PLWHIV, people living with HIV; WHO, World health organization; DTG, dolutegravir; NVD, nausea, vomiting, diarrhea; EFV, Efavirenz; ABC, Abacavir; NVP, nevirapine.
A total of 22 studies12,15,49–52,54,56–61,63,64,66,68–73 focused on adults. Two studies included participants from all age groups,16,62 while five studies specifically examined ADRs in pediatric populations.48,53,55,65,67 Seventeen studies employed a cohort design12,15,48,50,53,54,58–61,63,64,69,70,72,73 whereas the remaining studies used a cross-sectional approach (n = 12).16,49,51,52,56,57,62,65–68,71
Regarding the definition of ADRs, the majority of studies (n = 26) used the WHO definition, 2 while three studies50,51,69 applied the Edwards and Aronson's definition. 74 For ADR identification, reviewing medical records was the most common method (n = 22). Additionally, seven studies15,49,51,52,68,70,73 employed combined medical record reviews with patient interviews.
Causality, Severity, and Preventability of ADRs
Naranjo's algorithm, WHO-Uppsala Monitoring Committee causality assessment tools (WHO-UMC scales) and expert judgment techniques were employed in ADRs causality evaluation. ADRs were commonly classified under “probable” and “possible” categories. Severity grading criteria were varied between studies. The WHO severity grading scale, 75 AIDS Clinical Trial Group toxicity grading system, 76 Hartwig's and Siegel severity assessment scale, 77 and Division of AIDS Table for grading the severity of adult and pediatric adverse events (DAIDS AE Grading Table) 78 were employed. The majority of ADRs were graded as mild to moderately severe.15,48–54,57,59,62,63,67–73
Among the 29 studies that participated in this review, only Bezabhe et al 15 evaluated the preventability of ADRs based on Schumock and Thornton criteria. 79 That study revealed that a significant portion of severe ADRs were avoidable (47.5%). Concerning specific ADRs, serious ADRs such as hematologic toxicity (100%), hepatotoxicity (100%), nephrotoxicity (100%), and gastrointestinal toxicity (20.9%) were avoidable. 15
Common ADRs and Offensive Drugs
The commonly occurring ADRs in PLWHIV were classified in to metabolic and abdominal ADRs, hematologic ADRs, nervous system ADRs, musculoskeletal and integumentary system reactions, and cardio-renal, hepatic and systemic signs and symptom ADRs. The metabolic and abdominal organ ADRs encompass lipodystrophy, hyperglycemia, significant weight gain, gastrointestinal disturbances including nausea, vomiting, diarrhea (NVD), anorexia, constipation, gastritis and mucosal complications such as mucositis and oral ulcers. Further ADRs include diabetes mellitus, jaundice, lactic acidosis, mitochondrial toxicity, pancreatitis, and hepatotoxicity. These adverse effects have been predominantly associated with ARVs such as AZT, DTG, D4T, and the protease inhibitor combination lopinavir/ritonavir (LPV/r).16,53,55,56,61,64–66
In addition to these ADRs, cardio-renal and systemic toxicities have also been reported. Clinical presentations such as hypovolemia, persistent fatigue, fever, nephrotoxicity, interstitial nephritis, and progression to renal failure have been commonly documented. These complications are particularly associated with exposure to LPV/r, tenofovir disoproxil fumarate (TDF), didanosine (DDI), and abacavir (ABC).15,50–52,58,59,62,68,72,80–82
Musculoskeletal and integumentary system ADRs were frequently observed among patients on ART. Common musculoskeletal ADRs included myalgia, myopathy, muscle weakness, arthralgia, osteopenia, osteoporosis, and osteonecrosis. Integumentary reactions comprised alopecia, skin rash, allergic dermatitis, nail discoloration, pruritus, hyperpigmentation, and severe hypersensitivity reactions such as erythema multiforme and Stevens–Johnson syndrome. Efavirenz (EFV), AZT, Abacavir (ABC), and nevirapine (NVP) were predominantly implicated in these adverse effects.12,15,16,50–52,57,58,60,63,67,69,71,72
Hematological disorders including thrombosis/embolism, anemia, leukopenia, thrombocytopenia, neutropenia, and infections were frequently observed in this study. These ADRs were notably associated with the use of LPV/r, and nucleoside reverse transcriptase inhibitors such as ABC, AZT, D4T, as well as the non-nucleoside reverse transcriptase inhibitor NVP.12,50–52,60,67,69,71
Finally, nervous system disorders such as headache, agitation, ototoxicity, nightmares, confusion, somnolence, insomnia, vertigo, anxiety, paresthesia, dizziness, peripheral neuropathy, numbness, hearing loss, lethargy, psychosis, hallucinations, and depression were commonly reported. These adverse effects were primarily linked to the use of D4T, DDI, EFV, and DTG.51,54,55,59,66
Interventions Installed to Reverse ADRs
To overcome the ART-related ADRs, different interventions were undertaken. The main managements/measures taken by participant studies to resolve ADRs were symptomatic supportive treatment and inpatient care to minimize ADRs risk, offensive drug withdrawal, discontinuation of treatment, complete regimen change/switching therapy and enhancing patients’ ART adherence.15,16,51,52,68,71 It is also notable that insufficient interventions and a lack of proper laboratory monitoring may have exacerbated ADRs. 15 These insights underscore the need for comprehensive patient management and proactive monitoring to mitigate such risks.
Risk of Bias in Studies
A comprehensive assessment on risk of bias was performed on the 29 eligible studies using the standardized and tailored JBI critical appraisal tools, to the specific study designs included in the review. The evaluation process was detailed in (Supplemental Table S1
Meta-Analysis
Overall Prevalence of ADRs in PLWHIV
The effect sizes with 95% CIs, relative weights of each study, and the pooled prevalence of ART-related ADRs were summarized in the forest plot as shown in Figure 2. In this plot, it was found that the overall prevalence of ADRs in PLWHIV was 36.7% [95% CI: 26.6 − 46.9, I2 = 99.64%]. A total of 6695 ADRs were reported. The mean number of ADRs per patient was estimated to be (6695/19 003) 0.352.
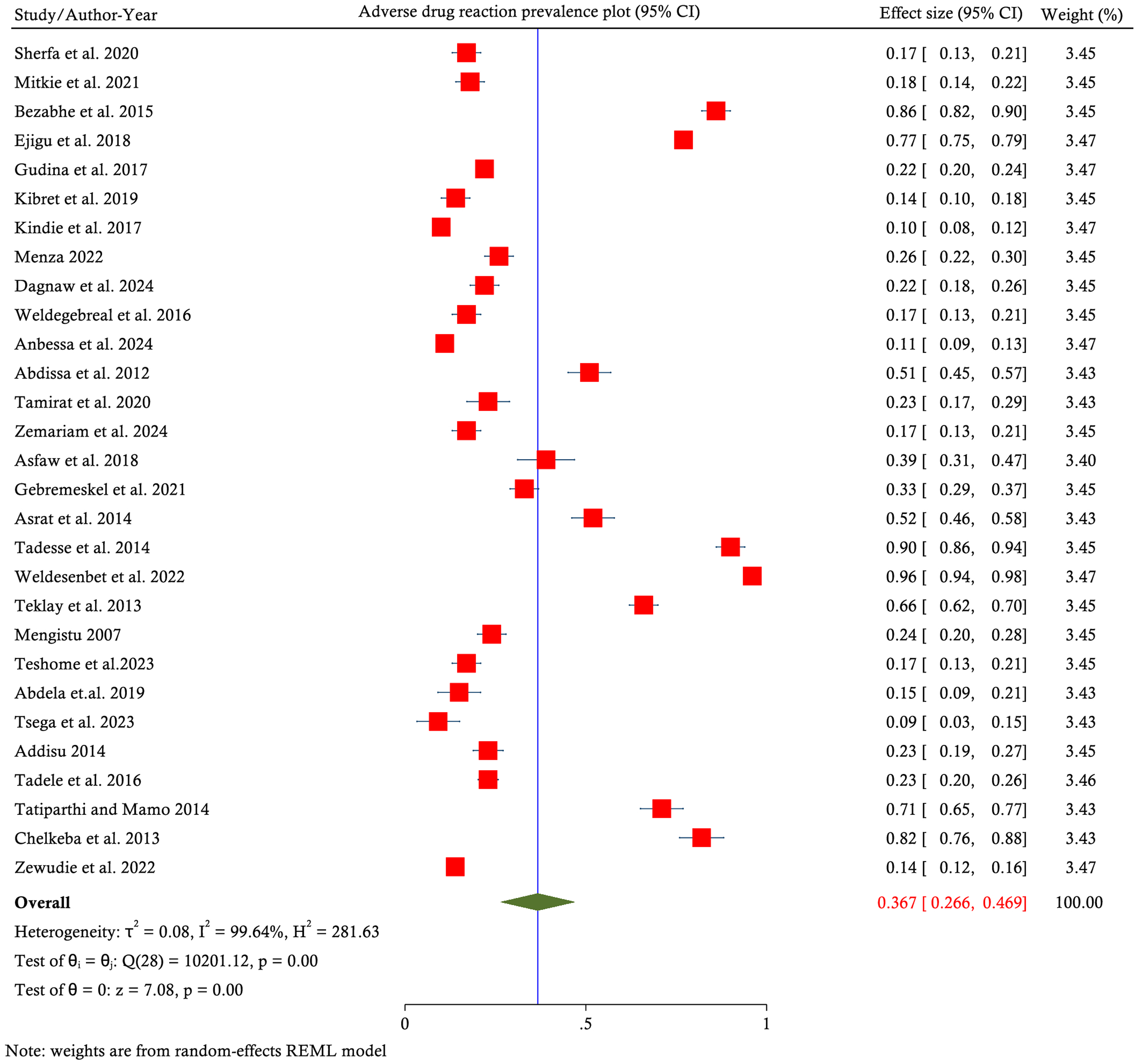
Forest plot for pooled prevalence of ADRs in patients living with HIV/AIDS in Ethiopia from 29 studies. ADRs, adverse drug reactions; HIV, human immunodeficiency virus.
Assesing Heterogeneity
Significant heterogeneity was confirmed from a random effects model pooled estimate as follows: Asymmetry of the forest plot, high Q value (9147.48) at P < .001 and I2 statistics 99.64% at P value <.001. Therefore, subgroup analysis, univariate meta-regression, and sensitivity analysis were conducted to locate the source of heterogenity between studies.
Subgroup Analysis
To explore the potential sources of heterogeneity, subgroup analyses were conducted based on age group, ADR definitions, study design, year of publication, and methodological quality. Despite extensive stratification, substantial heterogeneity persisted across most subgroups (I² > 78.56%). The summary of subgroup analyses is presented in (Table 2).
Subgroup Analysis of Prevalence of ART-Related-ADRs in Patients with HIV in Ethiopia, 2024.

Note: JBI: Joanna Briggs Institute quality score; CI, confidence interval; I², percentage of variation across studies due to heterogeneity rather than chance, high quality JBI score studies (JBI score: ≥ 73%), medium qualities (JBI score: 50%-72%); ART, antiretroviral therapy; HIV, human immunodeficiency virus; ADR, adverse drug reaction; WHO, World health organization.
The lowest ADR prevalence was observed among adult patients (16.1%, 95% CI: 11%-21.3%), while pediatric populations had higher prevalence rates (74.8%, 95% CI: 65.7%-83.9%). Cross-sectional studies tended to report higher ADR prevalence (46.4%) compared to cohort studies (29.9%). In addition, studies published before 2020 reported a higher pooled prevalence (44.8%) than those published after 2020 (25.3%). Notably, studies with medium JBI scores yielded a higher prevalence (50.2%) than those with high scores (30.6%). However, high to moderate level of heterogeneity persisted in all subgroups.
Meta-Regression Analysis
In light of subgroup analysis results, we executed a univariate meta-regression with year of publication, sample size, JBI quality score and mean age as the covariates. However, this analysis revealed that none of these variables were statistically significant predictors of heterogeneity across the participant studies. (Table 3).
Meta-Regression Analysis of Studies of ART-Related ADRs among Patients Living with HIV in Ethiopia, 2024.

Note: JBI quality score; Joanna Briggs Institute quality score; ART, antiretroviral therapy; HIV, human immunodeficiency virus; ADR, adverse drug reaction.
Sensitivity Analysis
In turn, the pooled estimate's robustness or stability was examined. No studies have a disproportionate impact on the total pooled estimate of ADRs, according to the random effects model in the leave-one-out sensitivity analysis. The pooled prevalence ranged between 35% and 38% upon sequential exclusion of studies. The highest estimate (38%) was observed when studies50,54,55,61,69 were excluded, while the lowest estimate (35%) occurred with the exclusion of studies15,16,51,63,65,66 (Figure 3).

Graphic representation of leave-one-out plot for pooled prevalence of ADRs in patients living with HIV/AIDS in Ethiopia. ADR, adverse drug reaction; HIV, human immunodeficiency virus.
Reporting Biases Assessment
Funnel plot indicated a symmetrical distribution of studies, suggesting a low likelihood of bias. This was supported by Egger's regression test, which yielded a nonsignificant intercept (β0 = 3.01, SE = 6.807, P = .6578), failing to detect small-study effects. Furthermore, Begg's test also did not provide an evidence of significant publication bias (Kendall's score = 108, SE = 50.098, z = 2.14, P = .0627). However, LFK-indices in DOI plot detected minor asymmetry (Figure 4 A and B).

(A) DOI and (B) funnel plot for assessment of the publication bias.
Risk Factors
Except three studies16,60,70 all (n = 26) were reported at least one influencing risk factor affecting ADRs. A total of 46 risk factors were predicted by those studies. These factors were grouped into three major categories namely sociodemographic, clinical, and treatment-related factors. Although categorizing diverse factors into a single group may introduce heterogeneity, we prudently opted to review them to a have better picture on the overall association between these potential risk factors and ADRs. We have aggregated risk factors which have been reported in more than one primary studies (Table 4). Publication bias was assessed using funnel plots, Begg's test, and Egger's regression test, all conducted under a random-effects model employing the REML method (Table 5). Additionally, the robustness of the pooled effect estimate was evaluated through a leave-one-out sensitivity analysis.
Meta-Analysis of Predictor Covariates for ART-Related ADRs among Patients Living with HIV/AIDS, 2024.

ART, antiretroviral therapy; HIV, human immunodeficiency virus; ADR, adverse drug reaction; OI, opportunistic infection; BMI, body mass index.
Publication Bias Analysis on Predictor Variables of ART-Related ADRs.

ART, antiretroviral therapy; ADR, adverse drug reaction; WHO, World health organization; OIs, opportunistic infections; BMI, body mass index.
Sociodemographic Risk Factors
Vulnerable individuals exhibited a cluster of sociodemographic risk factors: female gender, rural residency outside the catchment service area, marital status, limited or no formal education, economic hardship, lack of access to media, reliance on health centers, pregnancy, substance use, and unemployment.
Gender Related Risk Factors
The risk of ART-related ADRs was substantially higher in females living with HIV, almost two and half times that of males (AHR 2.51, 95% CI: 1.79-3.23; I2= 0.00) (Table 4). The robustness of this estimate was affirmed through leave-one-out sensitivity analysis (Supplemental Table S2). Furthermore, assessment for publication bias revealed no evidence of small-study effects (Table 5).
Educational status Related Risk Factors
Low educational status was emerged as one of the most pertinent risk factors of ART-related ADRs, with an AHR of 3.38 (95% CI: 1.429-5.328). A high level of heterogeneity was observed across the included studies (I² = 93.90%). The robustness of the overall estimate was confirmed through leave-one-out sensitivity analysis (Supplemental Table S3). Moreover, no evidence of small-study effects was detected (Table 5).
Clinical Risk Factors
Advanced WHO clinical stages (III and IV), OIs, comorbidity, baseline CD4 count <200 cell/μ, bedridden baseline functional status, underweight (poor nutritional status [body mass index—BMI<18]) were the reported clinical risk factors.
WHO Clinical Stage Related Risk Factors
Advanced HIV clinical stage (WHO stage III and IV) was consistently associated with ART-related ADRs in the 14 of primary studies.12,48–50,53–55,57–59,61,63,64,69 In this meta-analysis of those studies, individuals in advanced clinical stages of HIV were four times at risk to experience ART-related ADRs compared to those in earlier stages of the disease (AHR=3.571 95% CI: 2.351-4.791; I2 = 74.37%) (Figure 5).

Forest plot showing the association between advanced HIV clinical stage and ART-related ADRs across 14 studies in Ethiopia. ART, antiretroviral therapy; ADR, adverse drug reaction.
But it was not deemed significant in other studies by Bezabhe et al 15 Gudina et al, 71 and Mengistu et al. 73 The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S4). Although Begg's test indicated no evidence of publication bias, the asymmetry observed in the funnel plot, along with the statistically significant result from Egger's test (Table 5), suggested the potential presence of publication bias.
Consequently, a trim-and-fill analysis was conducted to adjust for this bias by imputing potentially missing studies (Figure 6). After adjusting for potential publication bias using the trim-and-fill method, the pooled point estimate for the association between advanced WHO clinical stage and ART-related ADRs was slightly attenuated to an AHR of 3.49 (95% CI: 2.24-4.60), indicating a persistent and statistically significant association.

Trim-and-fill analysis showing imputed studies to adjust for potential publication bias in the association between advanced WHO clinical stage and ART-related ADRs. ART, antiretroviral therapy; ADR, adverse drug reaction.
Chronic Diseases Commorbidity Related Risk Factors
Chronic disease multimorbidity increased the risk of ADRs in PLWHIV (AHR = 2.91, 95% CI: 1.31-4.99), with moderate heterogeneity (I² = 64.68%). The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S5). Additionally, no small study effect was observed in all Begg's, and Eggers test and funnel plot. (Table 5).
Baseline CD4 Count Related Risk Factors
Lower CD4 counts were associated with an increased risk of ADRs (AHR = 1.28, 95% CI: 1.09-1.659), with considerable heterogeneity observed (I² = 70.84%). The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S6). Additionally, no small study effect was observed in all Begg's, and Eggers test and funnel plots. (Table 5).
Opportunistic Infections
Individuals who experience OIs were two times at a higher risk of developing ADRs as compared to those who do not with no heterogeneity (I2 = 0.00). The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S7). Additionally, no small study effect was observed in all Begg's, and Eggers test and funnel plots. (Table 5).
Nutritional status
Individuals under a state of poor nutritional status (BMI<18) were more than three times at a higher risk of developing ART-related ADRs, AHR (3.312 2.116-4.509, I2= 64.64%) as compared to those with normal nutritional status. The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S8). Additionally, no small study effect was observed in all Begg's, and Eggers test and funnel plots. (Table 5).
Treatment Related-Risk Factors
Specific ART regimens, longer treatment durations, regimen changes, virological treatment failure, taking OI Prophylaxis such as CPT prophylaxis, IPT, and fluconazole, taking ART ≥2 times per day, concomitant tuberculosis treatment, and suboptimal adherence behavior were among the treatment-related factors identified by primary studies involved this review.
Types of ART Regimen Related Risk Factors
Patients living with HIV on AZT-based ART regimen were more than fourfold increased risk of developing ADRs as compared to those on the non-AZT-based regimens [AHR 4.24 (2.37 − 6.12)]. Moderate level of heterogeneity (I2 = 51.17) was noted, and the stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S9). Additionally, small study effect was not observed in all Begg's, and Eggers test and funnel plots.
Furthermore, PLWHIV receiving D4T-based ART regimen had approximately a threefold increased risk of experiencing ADRs [AHR 2.847 (1.162-4.532)]. Low level of heterogeneity was noted (I2 = 35.35%), and the stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S10). Additionally, no small study effect was found (Table 5). Patients who depend on NVP-as NNRTI in the HAART regimen carry 2 times higher risk as compared to those who use EFV (I2 = 43.11%).
Tuberculosis Treatment
Individuals undergoing concomitant tuberculosis treatment had approximately a twofold increased risk of experiencing ADRs. Low level of heterogeneity was noted, and the stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S11). Additionally, publication bias was diagnosed through Begg's, and Egger’s test and funnel plot. Therefore, trim and fill analysis was conducted (Figure 7).
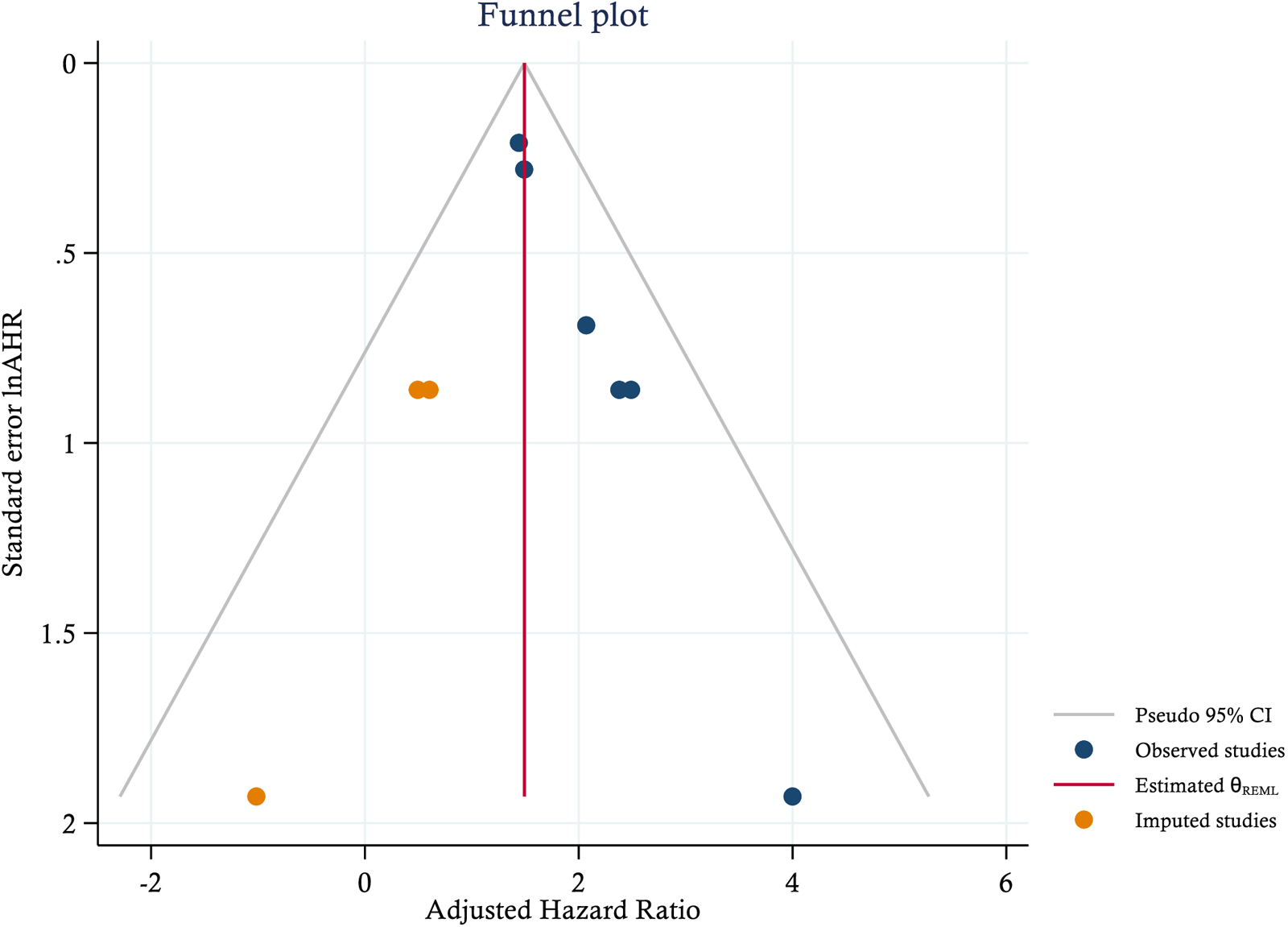
Funnel plot displaying imputed studies added Via trim-and-fill analysis to adjust for publication bias in the association between concomitant TB treatment and ART-related ADRs. ART, antiretroviral therapy; ADR, adverse drug reaction.
After adjusting for potential publication bias using the trim-and-fill method, the pooled point estimate for the association between concomitant TB treatment and ART-related ADRs was slightly attenuated to an AHR of 1.492 (95% CI: 1.195-1.790), indicating a persistent and statistically significant association.
Prophylactic Therapy to Opportunistic Infections
Includes cotrimoxazole prophylactic therapy, isoniazid prophylactic therapy and fluconazole for fungal infections such as Cryptococcal meningitis and Oropharyngeal and esophageal candidiasis. Individuals who took OI prophylaxis were two times at a higher risk in experiencing ADRs than who do not [AHR; 2.05 1.07-3.41]. High level of heterogeneity was revealed in this case. The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S12). Additionally, no publication bias was diagnosed through Begg's, and Eggers test and funnel plot.
Adherence status Related Risk Factor
Suboptimal adherence was a significant risk factor in the development of ADR in the ART continuum of care (AHR; 2.62 [1.355-3.869]; I2= 0.00). The stability of the overall estimate was confirmed by leave-one-out sensitivity analysis (Supplemental Table S13). Additionally, no publication bias was diagnosed through Begg's, and Eggers test and funnel plot (Table 5).
Certainty of Evidence
Risk of Bias: JBI-based risk of bias assessment revealed that 20 studies have high-quality JBI score studies (JBI score: ≥ 73%), 9 studies have medium qualities (JBI score: 50%-72%). Since most studies have low level of risk bias no downgrading was inevitable. Inconsistency (Heterogeneity): Both I2 statistic and visual inspection of the forest plot revealed high level of inconsistency inviting downgrading by 1 level from low grade to very low level of evidence certainty. Indirectness: Low risk of indirectedness due to appropriate population, healthcare condition and context in order to satisfy the review objectives. Furthermore, outcome measure “ADR” was defined consistently and in a way that aligns with our research question across all studies. Serious imprecision: Pooled prevalence proportion CI is too wide downgrade by 1 level. Regarding publication bias due to the absence of significant bias no change in certainty of an evidence level was done. Most analyses were downgraded due to imprecision, inconsistency, high risk of bias. However, publication bias and indirectedness did not significantly affect the certainty of the evidence. Altogether, using the GRADE approach, the evidence generated was adjudicated as very low.
Discussion
Medicines are crucial in the arena of the healthcare system. Despite the undeniable relevance of medicines, the potential harmful drug reactions remains a substantial concern. Realizing that no medication is without harm
The overall pooled estimate of ART-related ADRs in PLWHIV was found to be 36.7%. This finding was in agreement with previous studies conducted in Uganda (33.1%), 86 Eritrea (62.8%), 13 Sudan (55.5%), 87 South Africa (44.5%) 88 and Nigeria (28.33%). 89 However, it is more prevailed than studies in Tanzania (6.3%), 90 Cameroon (19.5%, 24.4%)91,92 UAS (6.7%) 93 and met-analysis studies by Hadia et al 94 (12.94%), (95% CI, 12.29-13.6, I2%-100%), and Insani et al 95 (8.3%, 95% CI: 7.82-8.83, I2-99.7%). On the contrary, this finding is less prevailed than reports from Kenya (65.2%), 96 Zimbabwe (70.4%), 97 Uganda (76%), 11 India (90%) 98 and Iran (96.50%). 99 This eye-opening figure carries significant clinical implications, which underscore a substantial public health concern, emphasizing the need for targeted interventions and improved ADR management strategies to decrease the risk of ADR-related morbidities and mortality. This discrepancy might be due to differences in definition, causality assessment, method of detection of ADRs, study settings, number of studies included, quality of studies, and regions where studies conducted. In addition, this difference might be due to poor ADR reporting experience, which is in congruence with a study conducted in Canada. 100
Heterogeneity between participant studies was high in this study. We deployed a standardized WHO definition for ADRs, comprehensive dataset avenues, and a critiqued high-quality articles to mitigate heterogeneity rates and prevent bias. Nevertheless, the studies that were included varied, maybe as a result of different study designs, locations, ADR definition, ADR reporting practice, ART-regimens and populations. 101 Yet, our overall estimates were largely consistent when sensitivity analysis techniques were applied. This finding is in line with a meta-analysis study conducted by Hakkarainen et al. 102 Finally, to avoid dissemination bias, we did not apply any restrictions to publication dates language, or irrespective of publication status
In the age-wise subgroups, pediatrics revealed a high rate of ADRs compared to adults’. This is in line with the scientific knowledge that pediatrics are prone to ADRs due in part to the high susceptibility of this group of patients, as they have smaller body sizes and larger surface areas than adults. A study conducted on hospitalized children shows that the overall pooled prevalence of ADRs was 9.53%, 103 which is less than the overall prevalence of ADRs in the pediatric age groups (74.8%) found in our study. Furthermore, the current review findings in the pediatric age group were higher than cohort studies conducted by Ray et al 104 (44.8%). This might be due to difference in the study setting and disease types.
Herein, the analysis of studies based on publication year suggests a trend towards lower ART-related ADR prevalence in more recent years, even if this difference didn't reach statistical significance. Specifically, studies published before 2020 reported an ADR prevalence of 0.448 (95% CI: 0.312-0.584), while those after 2020 showed a lower prevalence of 0.253 (95% CI: 0.121-0.385).This observed decline in ADR prevalence is plausibly attributed to the integration of DTG-based first-line ART, such as Tenofovir, Lamivudine, and DTG (TLD), into Ethiopia's national ART program. TLD is known for its improved side effect profile and higher tolerability compared to older ART regimens, which would naturally contribute to a reduction in reported ADRs. This aligns with the global shift towards DTG-based regimens due to their superior safety and efficacy.105,106
Regarding the drugs responsible for ADRs, the most common responsible ARVs were AZT, D4 T, EFV, DTG, LPV/r and NVP, which is in line with other studies.13,107,108 Another study in the USA found that protease inhibitors were the most common cause of ADRs. 109
In this systematic review, the most commonly affected systems were the metabolic, gastrointestinal, hematologic, nervous system, musculoskeletal and integumentary system, and cardio-renal, and hepatobiliary systems. Specifically, the types of ADRs most commonly introduced in patients with HIV were NVD, alopecia, anemia, hypovolemia, thrombosis/embolism, hepatotoxicity, peripheral neuropathy, skin rash, pruritus, dizziness, and neutropenia, in line with other studies.91,109
In this meta–analysis majority of ADRs were resides under the causality status of “probable” and “possible”. In agreement to this findings, a prospective cohort study in India reported that majority of ADRs were found to be possible (89.24%) under the causality of ADRs evaluation. 110 Furthermore a prospective observational study focused on the evaluation of ADRs in HIV positive patients in a tertiary care hospital in India revealed that more than two-third of ADRs were under probable status employing Naranjo's scale. 111 Regarding the degree of severity of the included studies, despite the fact that the majority were mild to moderate in severity grade, severe ADRs occurred significantly. This finding is in congruence with the clinical trial cohort in Thailand. 112
The absence of avoidability data was most noticeable in this systematic review, with only a single study providing avoidability data. Further studies are clearly required to determine which ADRs are potentially avoidable. These studies could provide the necessary data in order to enable clinicians to administer medications in the safest and most appropriate way. In line with the current study, a systematic review by Smyth et al, 113 including 102 articles, assessed preventability in only 19s.
ART-related ADRs were influenced by multi-risk factors, encompassing a complex interplay of sociodemographic, clinical and treatment-related risk factors. A salient finding of this review is the disproportionately elevated risk of ADRs among females living with HIV/AIDS, with an AHR of 2.65 (95% CI: 1.68-3.66), nearly threefold higher than that observed in males. This observation is consistent with findings from prior investigations conducted in diverse geographical contexts, including South Africa (AHR=1.67), 88 Brazil (AHR=1.93), 114 Ghana (AOR=1.66), 115 and Nigeria (AOR=1.8). 116 Furthermore, findings from ICONA cohort study revealed that females were at a higher risk of discontinuation for toxicity (AHR: 1.53%; 95% CI: 1.01 − 2.32) than men. 117 As evidenced by multiple studies this may be attributed to pharmacokinetic disparities where ARVs have higher systemic exposure in women compared to men. Several key physiological mechanisms are implicated in these pharmacokinetic differences, including variations in body weight and composition, renal clearance rates, and P-glycoprotein activity. For instance, women typically exhibit lower body weight and lean body mass, which can result in higher circulating drug concentrations. Furthermore, reduced activity of hepatic enzymes and diminished glomerular filtration rates may prolong drug elimination in women. Additionally, the concomitant use of other medications, such as contraceptives, hormonal supplements, and certain minerals and vitamins, particularly during women's reproductive years, may further predispose them to a higher incidence of ADRs due to potential drug-drug interactions.118–121
In this study, HIV positive patients with no formal education/low level of education attainments were about three and half times at risk of ADRs as compared to patients attended higher level of education. This finding is in line with a study done in Northern Nigeria, 116 Uganda 11 and India 122 where educational level of the patients was significantly associated with the development of ADRs. Higher educational status may protect against ADRs in patients on ART by enhancing health literacy, promoting better adherence, facilitating early recognition and reporting of side effects, improving communication with healthcare providers, and reducing reliance on potentially interacting traditional remedies.
Patients in the advanced HIV clinical stages were four times more likely to experience ART-related ADRs compared to those in earlier stages of the disease (AHR=3.92 95% CI: 2.83-5.01, I2 = 34.27%). This outcome corroborates earlier findings.98,123 The higher incidence of ADRs in patients with advanced HIV stages is primarily due to a combination of factors. These patients often face multiple comorbidities and OIs, leading to polypharmacy and an increased risk of drug–drug interactions, which heighten the likelihood of ADRs. Additionally, altered pharmacokinetics and pharmacodynamics in these individuals, along with compromised immune function, make them more susceptible to drug toxicities. Poor adherence, often caused by the pill burden, results in suboptimal drug efficacy, which may necessitate changes in treatment or even hospitalization, which further increase ADR risks. 58
Chronic disease multimorbidity increased the risk of ADRs in PLWHIV (AHR = 2.91, 95% CI: 1.31 − 4.99), with moderate heterogeneity (I² = 64.68%). As PLWHIV live longer due to ART, they are at an increased risk of developing chronic conditions like cancer, chronic kidney diseases, hypertension, diabetes, and cardiovascular disease. These comorbidities, along with the polymedications used to manage them, can increase the likelihood of ADRs, due to potential drug–drug interactions and overlap toxicities.11,124,125
Lower CD4 counts were associated with a 28% increased risk of ADRs, consistent with findings from a previous study conducted in Uganda, which reported that PLWHIV with CD4 counts below 200 cells/mm³ had a twofold higher risk of developing ADRs compared to those with higher counts. 11 This association may be explained by the persistent systemic inflammation observed in individuals with low baseline CD4 counts. Inflammatory cytokines such as interleukin-6, tumor necrosis factor-alpha, and interleukin-1 beta can suppress the activity of the hepatic cytochrome P450 (CYP) enzyme system, thereby altering the metabolism of many ARVs and increasing the risk of drug toxicity.126,127 Furthermore, a low CD4 count predisposes individuals to OIs, necessitating the use of additional medications, including antibiotics, antifungals, and antivirals. This results in polypharmacy, which further elevates the risk of drug–drug interactions and drug toxicity. In agreement with this idea in this study individuals who experience OIs were two times at a higher risk as compared to those who do not was noted.
This analysis indicates a significant association between poor nutritional status (BMI below 18 kg/m²), and an elevated risk of developing ART-related ADRs. Specifically, patients within this category exhibited a threefold increased risk compared to those with a normal BMI (AHR 3.312, 95% CI: 2.116-4.509). This finding aligns with existing literature suggesting that lower BMI is frequently correlated with a higher incidence of hospitalization, suboptimal ART response, and increased susceptibility to OIs. The subsequent polypharmacy to manage these complications may further contribute to the heightened risk of ADRs in this vulnerable population. It is crucial to consider that the physiological changes associated with malnutrition can alter drug pharmacokinetics and pharmacodynamics, potentially leading to increased drug toxicity. Therefore, these results underscore the importance of integrating nutritional assessment and support into the routine management of HIV patients initiating ART, particularly in resource-limited settings where malnutrition is endemic.128,129
In addition to the above predictors, specific ART regimens were among the pertinent risk factors of ADRs. PLWHIV who were on AZT-based treatment regimens had more than a fourfold increased risk of developing ADRs compared to those on non-AZT-based regimens. This finding is consistent with a prior cohort study conducted in India, which reported that individuals on AZT-based regimens were over eight times more likely to experience ART-related ADRs compared to those on TDF-based regimens. 130 Likewise, this result aligns with earlier studies indicating that TDF-containing regimens are better tolerated and associated with improved adherence when compared to AZT-based treatments.130,131 The heightened toxicity of AZT may be attributed to serious adverse reactions such as bone marrow suppression, nausea, myalgia, insomnia, and severe headaches. 132
Furthermore, PLWHIV receiving D4T-based ART regimens had approximately a twofold increased risk of experiencing ADRs. This finding corroborates previous study in Cameroon where D4T-based regimen carries a risk of ART-related ADRs by near to fourfold. 92 D4T-based treatment regimens are associated with significant long-term toxicities, predominantly lipoatrophy, neuropathy, lactic acidosis. 133 Therefore, D4 T was phased-out from ART-program mirroring its toxicity. 134
Patients on NVP-based regimens had a twofold higher risk of experiencing ART-related ADRs compared to those on EFV-based regimens, consistent with findings from studies in Uganda and South Africa.135,136 Likewise a meta-analysis by Shubber and his colleges revealed that adults on NVP were more than two times more likely to discontinue treatment due to any adverse event compared to patients on EFV (AHR 2.2, 95% CI: 1.9-2.6). 137
Individuals undergoing concomitant TB treatment with ART had approximately a twofold increased risk of experiencing ADRs. Concomitant ART and anti-TB medication, may introduce a potential pharmacokinetic interactions such as drug-drug interactions, drug toxicities overlaps, and TB immune reconstitution inflammatory syndrome.138–140 In addition to drug interactions between HIV and TB drug regimens, potential interactions with commonly prescribed drugs for OIs such as antifungal, antimalarial, antiviral, and other drugs used for treatment of OIs may introduce ADRs since such patients were liable for OIs. 139
PLWHIV on prophylactic therapy against OIs such as CPT, IPT, and fluconazole for fungal infections such as Cryptococcal meningitis and Oropharyngeal and esophageal candidiasis were two times at a higher risk of developing ADRs than who do not [AHR 2.05 95% CI: 1.07 − 3.41]. This may be due to the fact that these prophylactic therapy may increase patient-level medication regimen complexity index in those patients which is positively related to nonadherence and ADRs.141,142 However, on the contrary there are also studies which reported that prophylactic therapy reduces ADR for example a study in Nigeria revealed that CPT could protect the occurrence of ADRs. 91 This may be due to population, study design and treatment regimen differences.
Suboptimal adherence was a significant risk factor in the development of ADR in the ART continuum of care [AHR; 2.62 95%CI: 1.355-3.869), I2; 0.00]. Poor adherence stemming from complex ART regimens or treatment burden has the potential to trigger virologic failure, development of drug resistance and an increased risk for OIs. This requires a regimen switch to alternative possibly more complex ART regimens that may have different or more pronounced side effect profiles or hospital admission. This may put patients at a higher risk of drug toxicity due to drug–drug interactions when taking extra drugs to treat OI infection. 143
Strengths and Limitations
Given the paucity of data on this topic, to the best of our knowledge, this is the most comprehensive study addressing this issue by involving 19 003 participants from 29 studies. The robustness of our findings was underpinned by a comprehensive critical appraisal on participant studies, and sensitivity studies. This study was accomplished in accordance with the preregistered protocol make it replicable. As some of the studies had not evaluated ADRs as primary outcomes, the studies may have been underpowered for an effect on these variables. High level of heterogeneity across studies limits firm generalizations.
Conclusion
Altogether, ART-related ADRs in PLWHIV in Ethiopia were a crucial clinical issue with an overall pooled prevalence of 36.7%. Female gender, low education level, advanced disease stage, comorbidities, low CD4 counts, OIs and its treatment, low BMIs, AZT, D4T or NVP-containing regimens, concurrent TB treatment, and suboptimal adherence were the significant risk factors. Despite these results must be viewed with circumspection due to heterogeneity among studies, the findings from this comprehensive analysis may serve as a high-grade ore for policy makers in devising a pragmatic strategies.
Recommendations
Consequently, this finding implies that healthcare providers should pay worthy attention to the monitoring of ART-related ADRs in these vulnerable patient groups by adopting consistent and tailored ADR screening tools for early detection and treatment of ADRs, thereby improving ART safety and efficacy. Furthermore, policymakers should enhance education for vulnerable groups, ensure early HIV care, phase out high-risk ART drugs, integrate TB/HIV services, support adherence, address malnutrition, and prioritize high-risk patients through targeted monitoring and care.
Supplemental Material
sj-docx-1-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-3-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-3-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-4-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-4-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-5-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-5-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-6-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-6-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-7-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-7-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-8-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-8-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-9-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-9-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-10-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-10-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-11-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-11-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-12-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-12-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-13-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-13-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-14-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-14-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-15-jia-10.1177_23259582251358929 - Supplemental material for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-15-jia-10.1177_23259582251358929 for Epidemiology of Antiretroviral Therapy Related Adverse Drug Reactions and its Predictors Among Patients with Human Immunodeficiency Virus/AIDS in Ethiopia: A Systematic Review and Meta-analysis by Woretaw S Zewdu, Samuel B Dagnew and Mulugeta M Zeleke, Yared A Ferede, Achenef B Kassie, Tilay A Moges, Muluken A Alemu in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgements
We like to convey our appreciation to the authors of the studies featured in this systematic review and meta-analysis.
ORCID iDs
Author Contributions
All authors made a significant contribution to the work reported.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
We have provided all crude data that was utilized in this investigation as Supplemental materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.




