Abstract
There is no guideline concerning choice of antiretroviral therapy (ART) for HIV-infected patients after unplanned interruption. We conducted a retrospective cohort study of HIV-infected patients reintroduced to first-line ART after having unplanned interruption for at least 1 month. Viral load was evaluated at 6 to 18 months after the reintroduction. There were 100 patients included in our study, and 55 of them achieved virological success. History of single interruption (adjusted odds ratio [aOR] 5.51%, 95% confidence interval [CI] 1.82-16.68, P = .003) and CD4 count ≥200 cell/mm3 at the time of reintroduction (aOR 4.33, 95% CI 1.14-16.39, P = .031) increased likelihood to achieve virological success.
What Do We Already Know about This Topic?
There is no guideline concerning choice of antiretroviral therapy (ART) for HIV-infected patients after unplanned interruption, despite evidences that reintroduction of first-line ART may resulted in failure of control of viral load.
How Does Your Research Contribute to the Field?
This research show risk factors that might hinder successful reintroduction of first-line ART.
What Are Your Research’s Implications toward Theory, Practice, or Policy?
The research will be useful in determining factors that need to be reconsidered when forming guideline for first-line ART reintroduction.
Introduction
Antiretroviral therapy (ART) decreases morbidity and mortality arising from HIV infection. 1 Nevertheless, unplanned ART interruption may occur in nonmedical conditions such as patients being lost to follow-up due to poor adherence or unavailability of ART and in medical conditions such as severe drug intoxication, interfering intercurrent illnesses, or cessation of oral therapy due to surgery. 2 However, many of these patients return to health centers to resume ART and no recommendations to guide the choice of ART regimen for patients with history of unplanned interruptions. Reintroduction of first-line ART has a risk of failing because the virus could easily develop resistance to several drugs in first-line ART regimen such as non-nucleoside reverse transcriptase inhibitors (NNRTIs) and lamivudine (3TC). 3 Ideally, patients should have a drug resistance test before reintroduction as commonly practiced in the United States and Europe, 2,4,5 but resistance test is not yet an operating standard in Indonesia, for both first-line and second-line failure. On the other hand, a hasty decision to switch to second-line ART may lead to more serious concerns regarding the higher cost and the fact that if the virus had developed a resistance to second-line ART then it would had been resistant to every class of available antiretroviral drugs. 6 Consequently, there would be no more options available, since third-line ART is not yet available in Indonesia. 7
There were several studies on the probability of virological success of ART reintroduction and its associated factors. History of previous antiretroviral exposure reduces the probability of achieving virological success. 8 -10 Several studies used protease inhibitors (PIs) antiretroviral class, which is only used as second-line ART in Indonesia, 11,12 or the studies involved patients with history of planned interruptions. 8,11 -13 Other studies did not address tenofovir (TDF) as base nucleoside reverse transcriptase inhibitors (NRTIs), 14 while TDF is also the drug of choice for first-line ART in Indonesia. The outline of this study is to analyze factors that contribute to virological success in first-line ART reintroduction based on Indonesia ART regimen.
Methods
Study Patients
Retrospective cohort study was conducted on patients who received ART reintroduction in Dr Cipto Mangunkusumo Hospital (RSCM) in Jakarta. Inclusion criteria were 18 years of age or older and having an unplanned ART interruption for 1 month or longer before reintroduction. Patient was excluded from the analysis if he or she had history of confirmed virological failure in previous ART, had received anything other than standard first-line ART regimen, had an unknown previous ART regimen, had severe liver and/or kidney dysfunctions comorbidity, and did not have 6 to 18 months of viral load (VL) data after reintroduction. Study patients were drawn with total sampling by reviewing medical records of patients that fulfilled the inclusion criteria and did not fulfill the exclusion criteria.
First-line ART regimen in this study was a combination of 2 NRTIs and 1 NNRTI as defined by the recommendation of the Indonesian Ministry of Health. The choice of NRTI was a combination of zidovudine (ZDV) or stavudine (d4T) or TDF plus 3TC or emtricitabine. The choice of NNRTI was nevirapine (NVP) or efavirenz (EFV). 7,15,16 First-line ART reintroduction covered readministering of the same ART or a combination other than the ones regulated by switch strategy. Combination of ZDV or d4T-based NRTI was called a switch if replaced with TDF, and vice versa if TDF-based NRTI was replaced with ZDV or d4T. 17
Study Setting
This study was held on patients who had received ART reintroduction from 2005 until 2014. Data collected included (1) sample characteristics: age, sex, previous first-line ART duration, interruption interval, interruption frequency (single or multiple), body mass index (BMI) at the time of reintroduction, World Health Organization clinical stage at the time of reintroduction, tuberculosis (TB) coinfection at the time of reintroduction, and post-reintroduction adherence; (2) laboratory testing results: CD4 count at the time of reintroduction, hemoglobin (Hb) level at the time of reintroduction, and plasma VL 6 to 18 months after ART reintroduction. If patient had experienced repeated ART interruptions, only the ART duration before the last interruption and the last interruption interval were counted. Interruption interval was measured based on the interval between the estimated last day of ART consumption and the day patient received ART reintroduction. Interruption interval less than 1 month was excluded from the study because it might had not been properly recorded in the medical record. 2,18 Post reintroduction adherence was stated in percentage of dosage taken monthly according to patient’s statement as recorded in medical record. Patient was declared achieving virological success if VL count <400 copies/mL after receiving ART for a minimum of 6 months. If there were more than 1 VL tests in the period of 6 to 18 months after reintroduction, then the lowest VL count was used.
Statistical Methods
Analyses of factors affecting virological success were completed using bivariate and multivariate logistic regressions. On independent variables with less than 15% missing data, imputation was done using single imputation method. All of data processing was done electronically using SPSS 20.0 software.
Results
Recruitment and Characteristics of Study Patients
A search of medical records identified 100 patients restarted first-line ART and tested for VL after 6 to 18 months. Sixty-seven percent were male. Median age at the time of reintroduction was 31.4 (23.0-58.7) years. Most patients had interrupted once (79%), followed by twice (17%), 3 times (3%), and 4 times (1%). There were 61% of patients with CD4 count <200 cell/mm3. Median previous ART duration was 12.3 (0.7-94.0) months. Most patients had interruption interval ≥6 months (64%), BMI ≥ 18.5 kg/m3 (60%), and Hb ≥ 11.0 g/dL (49%). Only 24% of patients presented with TB coinfection, while stages III and IV seemed comparable to stages I and II (57% and 53%, respectively). Nine percent used TDF as NRTI basis. Only 69% patients reported ≥95% adherence.
Virological success was achieved in 55 patients (95% confidence interval [95% CI]: 45.25-64.75). Patients who had low CD4 count and multiple frequencies of interruptions were at higher risk of not achieving virological success (adjusted odds ratio = 4.33 and 5.51, 95% CI: 1.14-16.39 and 1.82-16.68, P = .031 and .003) (Table 1).
CD4 Count and Interruption Frequency Were Associated With Virological Success.
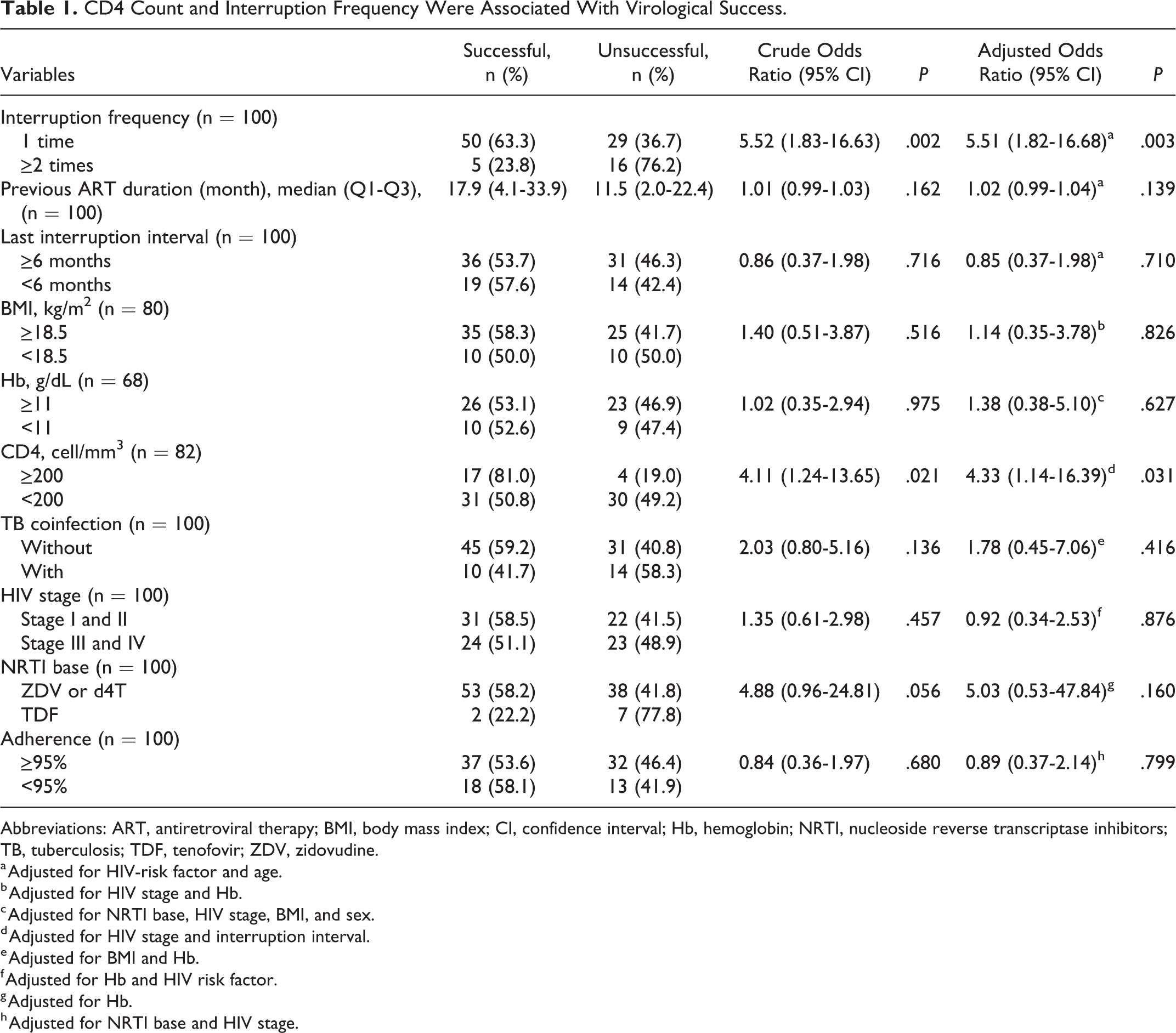
Abbreviations: ART, antiretroviral therapy; BMI, body mass index; CI, confidence interval; Hb, hemoglobin; NRTI, nucleoside reverse transcriptase inhibitors; TB, tuberculosis; TDF, tenofovir; ZDV, zidovudine.
a Adjusted for HIV-risk factor and age.
b Adjusted for HIV stage and Hb.
c Adjusted for NRTI base, HIV stage, BMI, and sex.
d Adjusted for HIV stage and interruption interval.
e Adjusted for BMI and Hb.
f Adjusted for Hb and HIV risk factor.
g Adjusted for Hb.
h Adjusted for NRTI base and HIV stage.
Discussion
Virological success was observed in 55% of patients. This finding is lower than similar study by Luebbert et al 14 in Malawi which observed virological success of 61%. Other studies with the same objective but different settings (by including PI usage and unplanned interruption) observed a probability of around 80%. 8,12 Virological success probability in our study is much lower compared to naive patients. A 2013 study in Bandung, Indonesia, observed virological success in 91% naive patients who had received ART for at least 6 months. 19 A history of previous unplanned interruptions causes the patients to possibly carry virus that has developed ART resistance mutations, especially against first-line ART. Mutant virus is still persist in the reservoir and could redominate after reintroduction of the same ART. 20 -23
Our study shows that patients with interruption frequency of 1 time have a higher odds to achieve virological success in ART reintroduction. Repeated ART interruptions can increase the number of mutant virus resistant to antiretroviral drugs. 3,24 Similar study by Luebbert et al 14 did not find any significant association between interruption frequency and post reintroduction virological failure. Based on interruption frequency distribution, of 133 study patients in Luebbert et al, 14 only 10% had 2 or more interruptions. This finding differs from our study where 21% of study patients had the interruption frequency of 2 or more. Other than that, the definition of interruption in Luebbert et al 14 study not only covered the complete interruption of ART but it also included patients with treatment gaps (still taking ART once in a while). Both differences may be the cause of the different results with our study.
A low CD4 count shows depleted immune system; thus, the capacity to recognize and suppress viral replication is also diminished. 25 In addition, the effect of a higher baseline CD4 count on virological success may be the effect of a low baseline VL on the effectivity of the treatment. 26 The association between CD4 count at the time of reintroduction and virological response has been proven by Greig et al. 8 Patients with higher CD4 count at the time of reintroduction have a lower odds to experience virological failure in ART reintroduction. Several other studies, notably by Vogler et al 12 and Touloumi et al, 13 which did not succeed in proving this association, had patients with different CD4 count characteristics from our study, where their average CD4 counts before treatment resumption was higher, ranging at 265 to 332 cell/mm3. 12,13 Patients in Vogler and Touloumi studies achieved virological response rate higher than our studies (81% and 86%). Vogler mentioned that baseline CD4 count was not associated with virological response but mentioned nonadherence as the main factor for failures.
The main limitation of this study is the retrospective study design that collected data from medical records. There were several variables with a large amount of missing data and possible information bias that we could not control. Viral load testing which was not done in series with equal interval on each patient might also cause this study to overlook post-ART reintroduction VL dynamics and time needed to achieve virological success. On both significant variables, there is a wide interval confidence. Hence, the application of our study on the population may not be precise.
Conclusion
Probability of virological success on first-line ART reintroduction after unplanned interruption was 55%. Patients who were interrupted once and patients with CD4 count ≥200 cell/mm3 at the time of reintroduction had a higher probability of success treatment.
Footnotes
Authors’ Note
This study does not directly involve human trial. Research methodology had been reviewed and approved by ethic committee of Faculty of Medicine Universitas Indonesia and ethical approval was obtained with ethical clearance number 196/UN2.F1/ETIK/2015. The ethic committee of Faculty of Medicine Universitas Indonesia waived the need for ethics approval and the need to obtain consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this noninterventional study. This study abide by Helsinki Declaration and all patient’s medical record were confidential.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
