Abstract
We compared the demographic and disease characteristics of HIV-positive (HIV+) and HIV-negative (HIV−) individuals with a diagnosis of cancer in South Carolina. HIV-positive patients with cancer were reflective of the HIV+ caseload in South Carolina, with HIV+ patients with cancer more likely to be male (odds ratio [OR]: 2.78: 95% confidence interval [CI]: 2.33-3.32), black (OR: 7.68; 95% CI: 6.52-9.06), and younger at cancer diagnosis (OR: 0.92; 95% CI: 0.91-0.92). Controlling for year of birth, HIV+ patients with cancer did not receive cancer diagnoses at a younger age than HIV− controls. HIV-positive individuals did not have more advanced tumor stages or grades at cancer diagnosis; however, after controlling for other factors, HIV+ individuals were still more likely to be deceased at follow-up (OR: 2.64; 95% CI: 2.20-3.17) when compared to HIV− controls. Future studies should use survival analysis methods to identify the characteristics that shorten survival among HIV+ patients with cancer.
Introduction
HIV infection has evolved from an acute, highly lethal disease to a chronic condition requiring continued treatment and management. With appropriate access and adherence to improved antiviral therapies, the average life expectancy of HIV-positive (HIV+) individuals approaches that of HIV-negative (HIV−) age-matched controls. 1,2 As patients live longer, leading causes of death are changing from AIDS-defining opportunistic infections to common comorbid conditions seen in the general population, including metabolic, vascular, and malignant diseases.
Prior to the development of highly active antiretroviral therapies (ART), several malignancies, or AIDS-defining malignancies (ADMs), associated with immune system suppression were common among HIV-infected individuals; these included non-Hodgkin lymphoma, Kaposi sarcoma, and invasive cervical cancer. 3 However, in the post-ART era, due to improved immune function and longer life expectancies while on ART, the incidence and mortality rates of Kaposi sarcoma and non-Hodgkin lymphoma have declined dramatically. 4,5
Since the introduction of ART, non-ADMs (NADMs) have notably increased in the HIV+ population, with the rates of several NADMs now higher among HIV+ individuals than in the general population. Although findings vary across studies, these includ Hodgkin lymphoma, melanoma, liver, lung, oropharyngeal, and colorectal cancers. 3,6,7
Currently, NADMs account for more than 50% of cancer diagnoses in patients with AIDS, and rates will likely continue to rise. 8,9 Theoretically, increased incidence rates of these malignancies in the HIV/AIDS population could be related to immune suppression, high rates of coinfections with other sexually transmitted infections (eg, hepatitis B and C and oncogenic human papillomavirus), toxicities associated with antiretroviral drugs, chronic inflammation due to persistent infections, higher rates of alcohol and cigarette consumption, or the aging process, which may be accelerated among individuals with HIV. 7,10 –13
The Southeastern United States has experienced the greatest increase in the incidence of AIDS since the 1990s. 14 Southern states accounted for the majority of incident cases of HIV in 2012 (51%), and this trend continues. 15 South Carolina specifically has some of the highest incidence and prevalence rates of HIV. 16 Racial disparities in HIV are common nationwide, 17 and in South Carolina, 70% of new diagnoses occur among African Americans. 18
In addition to high incidence/prevalence rates of HIV/AIDS, South Carolina consistently has high cancer incidence rates, with distinct racial disparities driven by the burden of cancer in African Americans. 19,20 In 2015, an estimated 25 550 cancer cases for all sites were diagnosed, with annual incidence rates from 2007 to 2011 at 544.6 cases/100 000 among males and 402.9 cases/100 000 among females. 21 South Carolina faces other notable health problems, including disparities in access to care, high poverty and unemployment, low life expectancy, high rural and minority populations, and high rates of morbidities and mortalities. 19,22
Despite the high rates of HIV/AIDS and cancer in the southeast, limited research has examined the interaction between to HIV, cancer, clinical characteristics, and demographic factors. Understanding this relationship is crucial for targeting resources and improving cancer screening, prevention, and treatment strategies. This research is especially important as the HIV/AIDS population continues to age and becomes at higher risk for developing various malignancies. Therefore, the purpose of this article is to identify the characteristics associated with HIV+ patients with cancer. Specifically, we hypothesize that HIV+ patients with cancer will be more likely to occur in younger and minority individuals. Clinically, we hypothesize that HIV+ patients with cancer will have a higher tumor grade and later stage at cancer diagnosis and poorer survival when compared to HIV− patients with cancer.
Methods
We linked all HIV+ cases reported to the South Carolina enhanced HIV/AIDS Reporting System (eHARS) to the South Carolina Central Cancer Registry (SCCCR) to identify all HIV+ patients with cancer in South Carolina. HIV− cancer controls were selected from SCCCR via computer probabilistic matching by SCCCR at a ratio of 1:5 matched on cancer site and year of cancer diagnosis. Linkages were performed by the data custodians based on Social Security number and other personally identifying variables, of which none were available to our investigative team. Inclusion criteria were age >15 years at cancer diagnosis; diagnosed with their first primary tumor between January 1, 1996, and December 31, 2010; and having a malignant tumor.
Characteristics associated with an individual’s first cancer diagnosis in both HIV− and HIV+ individuals (HIV or AIDS diagnosis at or before date of cancer diagnosis) were of interest. Rather than approximating the HIV+ seroconversion date using an individual’s CD4+ T-cell count at HIV diagnosis, this study used a more conservative classification based on the HIV or AIDS diagnosis date. Individuals who received an HIV or AIDS diagnosis prior to the date of cancer diagnosis or up to 1 year after their cancer diagnosis were classified as HIV+. HIV+ individuals who received an HIV or AIDS diagnosis more than 1 year after their cancer diagnosis (0.88% of sample) and individuals listed as being HIV+ but with no HIV diagnosis dates available (0.34% of sample) were excluded from the analysis.
Age at cancer diagnosis, tumor characteristics (ie, grade and stage), gender, race, date of cancer diagnosis, vital status, and vital status at follow-up were obtained from the SCCCR. The SCCCR is gold certified by the North American Association of Central Cancer Registries and holds national recognition for data timeliness, completeness, and accuracy. 23 The dates of HIV/AIDS diagnoses for HIV+ cases were obtained from eHARS. The eHARS is maintained by the South Carolina Department of Health and Environmental Control HIV Surveillance Division. The eHARS quality exceeds the Centers for Disease Control and Prevention minimum standards on timeliness and completeness of reporting, with 95% of cases being reported within 6 months of diagnosis and 98% of all cases reported to eHARS. 24
Statistical Analysis
Using morphology and histology codes, cancers were classified by tumor site. Cancer sites with less than 5 cases (3.48% of sample) and those classified as “miscellaneous” were excluded from the analysis (3.31% of sample). Cancers were further collapsed into ADMs and non-ADMs.
Differences in gender, race, vital status, age at cancer diagnosis, tumor grade, and tumor stage between HIV+ patients with cancer and HIV− cancer controls were compared through χ2 tests. Prior to analysis, age at cancer diagnosis was categorized according to the National Cancer Institute’s standards (age 16-34, 35-44, 45-54, 55-64, 65-74, 75-85, and 85+ years). Race was collapsed into 3 groups due to low numbers in certain minority groups: white, black, or “other.” Tumor stage, a measure of disease extent that describes how far the cancer has progressed from its site of origin, was collapsed into 4 groups based on the Surveillance, Epidemiology, and End Results Program (SEER) summary stage at diagnosis: localized, regional, distant, and not applicable/unstaged. Tumor grade, a measure of how closely tumor cells resemble the parent tissue, was collapsed into 3 categories due to the small number of graded tumors: grade 1/grade 2 (well- or moderately differentiated), grade 3/4 (poorly differentiated or undifferentiated), and grade not applicable.
An unconditional logistic regression analysis, rather than conditional, was performed because HIV+ and HIV− patients with cancer were statistically different across every variable assessed, including cancer type, one of the matching variables. In the logistic regression model, all variables were categorized or collapsed as described for the χ2 tests; however, age at cancer diagnosis was included as continuous variable. Individuals of “other” race were excluded from the logistic regression model due to small numbers (n = 82). Univariable logistic regression analyses were performed to identify the demographic and clinical characteristics that were associated with the outcome variable based on a P value of .05. Significant covariates in the univariable models were included in the final multivariable logistic regression model.
Age at cancer diagnosis, race, vital status, sex, tumor grade, and tumor stage were found to be significant in univariable regression analyses with HIV status as the outcome variable and were all included in the final multivariable logistic regression analysis. Adjusted odds ratios (ORs) and 95% confidence intervals (CI) were obtained through the multivariable logistic regression model.
To determine whether HIV-infected individuals received cancer diagnoses at a younger age as a function of their disease status (rather than HIV population being intrinsically younger), a linear regression analysis was conducted with HIV status, year of birth, and an interaction term with year of cancer diagnosis as the outcome variable.
SAS v9.3 (SAS Institute) was used for all analyses. The institutional review board of the University of Georgia and the Data Oversight Committees of eHARS and SCCCR approved this study.
Results
From 1996 to 2010, there were 1292 individuals living with both HIV and cancer in South Carolina (based on linked data) and 6448 matched HIV− cancer controls. After applying the exclusion criteria explained in the Methods section, 1133 HIV+ patients with cancer and 5,988 HIV− cancer controls remained in the sample for final analyses. From 1996 to 2010, the number of HIV+ patients with cancer has gradually increased (Figure 1).

HIV-positive patients with cancer counts by year in South Carolina.
The distribution of cancer sites and vital status are displayed in Table 1. The most common primary cancer sites in the HIV+ cases were non-Hodgkin lymphoma (27.5%); lung and bronchus (17.7%); Kaposi sarcoma (8.7%); prostate (6.0%); anus, anal canal, and anorectum (5.5%); Hodgkin lymphoma (4.5%); female breast (4.2%); and colon and rectum (4.1%). White and African American HIV+ individuals with ADMs and NADMs were both significantly more likely to be died at follow-up when compared to HIV− controls.
Cases of AIDS-Defining and Non-AIDS-Defining Malignancies among HIV-Positive Cases and HIV-Negative Controls in South Carolina from 1996 to 2010, Stratified by Race.a

aN = 7121.
The distribution of patient demographics and clinical characteristics are displayed in Table 2. Comparing HIV+ and HIV− patients with cancer using χ2 tests, HIV+ patients with cancer were significantly more likely to be younger, male, and black. Clinically, HIV+ patients with cancer were significantly more likely to be deceased at follow-up and have more advanced stage tumors at cancer diagnosis when compared to HIV− controls. Tumor grade was significantly different between HIV+ cases and HIV− controls; however, a large proportion of individuals had tumor grades classified as “not recorded” or “unavailable.”
Demographic and Tumor Characteristics of HIV+ Cases and HIV− Controls in South Carolina from 1996 to 2010.a
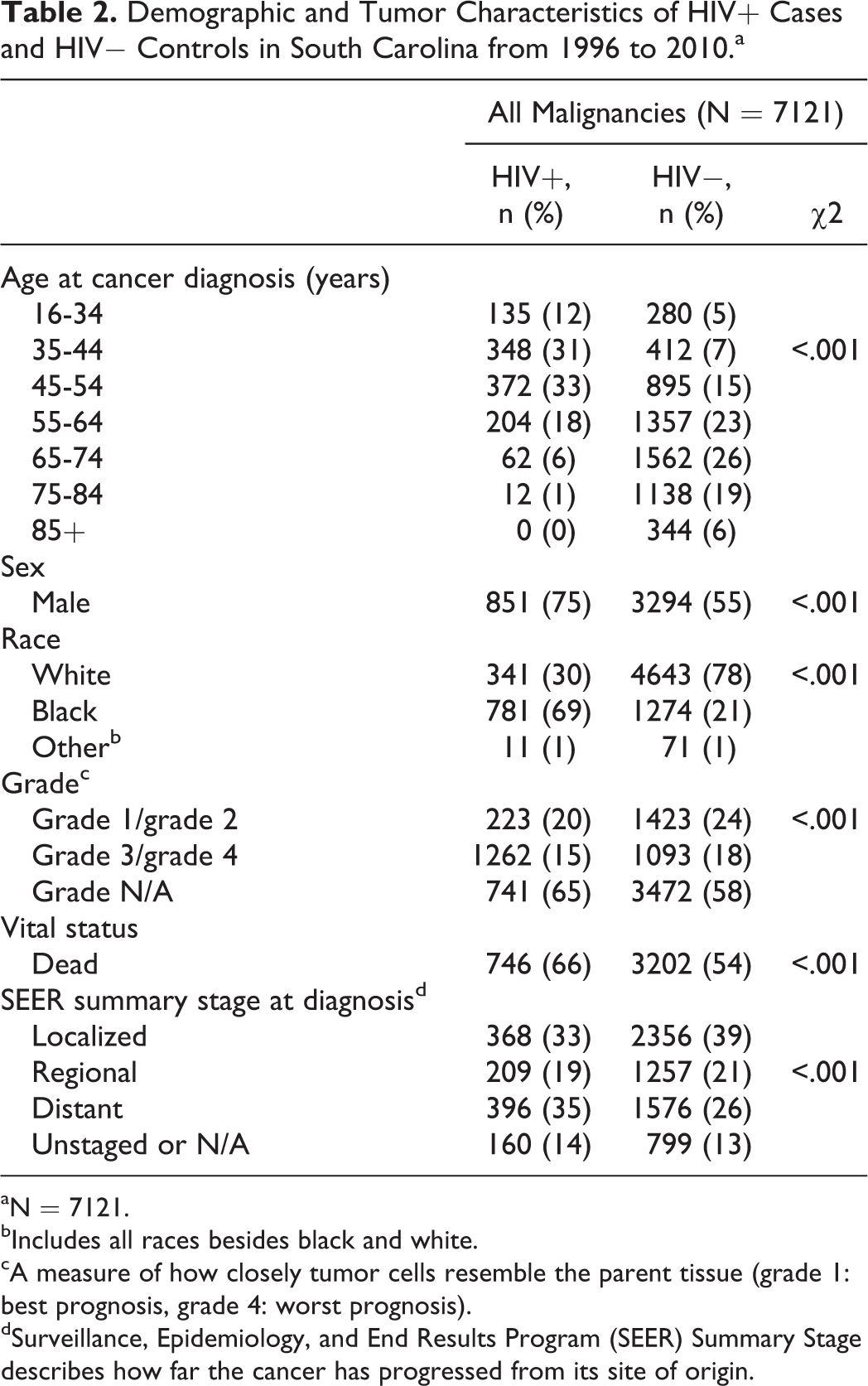
aN = 7121.
bIncludes all races besides black and white.
cA measure of how closely tumor cells resemble the parent tissue (grade 1: best prognosis, grade 4: worst prognosis).
dSurveillance, Epidemiology, and End Results Program (SEER) Summary Stage describes how far the cancer has progressed from its site of origin.
Adjusted ORs using HIV status as the outcome variable were generated with a multivariable logistic regression model (Table 3) and confirmed the patterns observed in Table 2. Demographically, HIV+ patients with cancer were significantly more likely to be black (OR: 7.68; 95% CI: 6.52-9.06), male (OR: 2.78 95% CI: 2.33-3.32), and have a younger age at cancer diagnosis (OR: 0.92; 95% CI: 0.91-0.92) when compared to HIV− cancer controls. Clinically, HIV+ cases were not significantly more likely to have a higher cancer grade or stage at cancer diagnosis; however, after controlling for other factors, HIV+ individuals were still more likely to be deceased at follow-up (OR: 2.64; 95% CI: 2.20-3.17) when compared to HIV− controls with cancer.
Multivariable Logistic Regression Results Using Patient Demographic and Tumor Characteristics to Predict HIV+ Status, Using Black and White HIV+ patients with cancer and HIV− Cancer Controls.a

aexcluding race “other”: n = 7039.
bA measure of how closely tumor cells resemble the parent tissue (grade 1: best prognosis, grade 4: worst prognosis).
cDescribes how far the cancer has progressed from its site of origin.
In the final linear regression analyses, using year of cancer diagnosis as the outcome variable, HIV status and year of birth were both significant predictors (P < .0001) and were both negatively correlated (HIV+ individuals and those born more recently were more likely to be younger at cancer diagnosis). When the interaction term was added to the model, however, year of birth was the only variable significantly related to the outcome variable (age at cancer diagnosis).
Discussion
This study aimed to identify the patient demographic factors and clinical characteristics that are associated with HIV+ patients with cancer. Understanding this relationship can help guide future research and help improve cancer screening, prevention, and treatment strategies among the HIV/AIDS population as they continue to age and become at greater risk of developing NADMs.
In our study population, 62% of the HIV+ patients with cancer were diagnosed with an NADM, which supports current observed trends in the literature documenting that NADMs account for more than 50% of cancers among patients with HIV/AIDS. 1,2,8,9 Due to increased life expectancy and better immunologic functioning from improved antiviral therapies, HIV+ individuals are less likely to be diagnosed with classic AIDS-associated opportunistic infections and are more likely to acquire NADMs and other common comorbid conditions seen in the general population.
Focusing first on demographic characteristics, as expected, HIV+ patients with cancer were more likely to occur in younger, black, and male individuals. Although little research has been conducted on HIV+ patients with cancer in South Carolina, these results were expected as they align with the HIV epidemic in South Carolina, in which 71% of all HIV+ cases are black, 70% are male, and 80% are below the age of 49. 25 Independent of HIV, these results are also consistent with trends in cancer rates; due to biological and lifestyle factors, men have higher incidence and mortality rates across all cancer sites, excluding gender-specific cancers (ie, cervical, breast, vaginal). 26 Among men, black individuals have higher incidence and mortality rates for all major types of cancer when compared to non-Hispanic white men. 27
While previous research suggests that HIV+ individuals receive cancer diagnoses at younger ages than the general population due to the aging process, which may be accelerated in this population, 10,13 we believe that this observation may be due to a function of the HIV epidemic (as HIV+ individuals are younger than the general population). To test this hypothesis, and to control for the possible cohort effect, a linear regression analysis was conducted with year of cancer diagnosis as the outcome variable. As the interaction term in this model (HIV status by year of birth) was not significant, this suggests that HIV+ individuals receive cancer diagnoses at a younger age because they come from a younger cohort. Controlling for year of birth, future studies should explore this hypothesis.
Clinically, HIV+ patients with cancer were not more likely to have a higher grade or stage tumors at cancer diagnosis when compared to HIV− cancer controls. However, after controlling for these variables and other demographic characteristics, HIV+ patients with cancer were still over 2 times more likely to be deceased at follow-up when compared to HIV− cancer controls. As SEER regularly updates its vital status at follow-up with data from the National Center for Health Statistics, which records all death certificates at the national level, it is unlikely that this result is due to differential misclassification. Additionally, as all cancers were accurately matched on year of cancer diagnosis, it is likely that this result is representative of a true effect. To explain this difference in mortality, future studies should use survival analysis methods with additional clinical and tumor characteristics to identify the factors that are associated with survival of HIV+ patients with cancer.
Strengths and Limitations
South Carolina is one of a few states to successfully implement and integrate multiple statewide surveillance systems, which allowed for the creation of the HIV/cancer data set used in this study. The SCCCR and eHARS data sets utilized in this study have over 15 years of experience in surveillance and data collection, and these rich data sources have never been examined in unison to evaluate the burden of certain malignancies among South Carolina residents with and without HIV. This article is significant as it utilizes this new resource to comprehensively examine the epidemiology of HIV and malignancy in South Carolina. While HIV− cancer controls were matched for year of cancer diagnosis, as expected, some malignancy types did not occur in large enough numbers in the general population to allow for perfect matching, creating some imbalances in malignancy groups which could have affected our results. Although these registries exceed national standards, they were not created for research purposes, and it is possible that some cancer or HIV diagnoses went unreported or that the records could be incomplete for some individuals. Given the nature of the cancer registry and HIV surveillance system, the problem of nonreporting of data could not be addressed. However, most variables used in this analysis required data by the respective databases (or by South Carolina state law), and data quality controls are employed by both agencies. Unfortunately, for this analysis, we did not have access to HIV treatment information for the HIV+ patients with cancer included in the study. Future studies should assess the impact ART treatment could have on the clinical characteristics of HIV+ patients with cancer at diagnosis.
Conclusion
This study compared patient demographic factors and clinical characteristics of HIV+ and HIV− patients with cancer in South Carolina in order to better characterize HIV+ cancer with cancer for future screening, prevention, and research efforts. Reflective of the current distribution of demographic factors among HIV+ individuals, we found that HIV+ patients with cancer were more likely to occur in younger, male, and black individuals when compared to HIV− cancer controls. Despite no significant differences in tumor stage or grade at diagnosis, HIV+ patients with cancer were significantly more likely to be died at follow-up. With follow-up conducted with national surveillance data, and with cases matched by diagnosis year, it is likely that this result represents a true effect. Future studies should use survival analysis methods with additional clinical and tumor characteristics to better identify the characteristics that put HIV+ patients with cancer at increased risk.
Footnotes
Acknowledgments
The authors would like to thank South Carolina Department of Health and Environmental Control and South Carolina Revenue and Fiscal Affairs Office for conducting the data linkage and Dr J. Michael Kilby (Medical University of South Carolina) for his guidance in designing the initial study protocol and analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
