Abstract
Background:
Patients with drug-resistant HIV often require complex antiretroviral regimens. However, combining fixed-dose combination tablets such as tenofovir–disoproxil–fumarate, emtricitabine, and cobicistat-boosted elvitegravir (TDF/FTC/EVG/cobi) with darunavir (DRV) can provide a simple, once-daily (QD), 2-tablet regimen for patients with drug-resistant HIV. Primary objective was to determine the percentage of patients with HIV-1 RNA <40 copies/mL at 48 weeks.
Methods:
We performed a retrospective chart review of patients initiated on TDF/FTC/EVG/cobi plus DRV.
Results:
Among the 21 included patients, prior resistance showed a median of 2 nucleoside reverse transcriptase inhibitor mutations, 1 nonnucleoside reverse transcriptase mutation, and 1 protease inhibitor mutation. At week 48, 14 (67%) patients achieved HIV-1 RNA <40 copies/mL, 1 patient experienced viral rebound, and 6 (29%) had missing data or discontinued therapy. No patient discontinued for adverse events.
Conclusion:
According to this observational study, QD TDF/FTC/EVG/cobi plus DRV is considered safe, well tolerated, and generally effective in suppressing HIV drug-resistant virus.
Introduction
Patients with drug-resistant HIV often require complex regimens involving numerous tablets taken more than once daily (QD) in order to successfully suppress the virus. 1–2 Treatment attributes that contribute to regimen complexity such as pill burden, multiple administration times, and side effects are major factors that adversely affect adherence to antiretroviral therapy (ART). 3–4 Poor adherence may lead to the development of drug resistance and treatment failure. 5 The QD dosing of ART and the introduction of fixed-dose combination tablets have been associated with improved adherence to HIV treatment 6 –9 ; thus, treatment simplification to reduce dosing and the number of pills may be a strategy to maximize adherence, improve quality of life, and achieve therapeutic success. 10
In recent years, the fixed-dose antiretroviral (ARV) combination tablet tenofovir disoproxil fumarate/emtricitabine/elvitegravir/cobicistat 300/200/150/150 mg (TDF/FTC/EVG/cobi), has been approved to be a complete regimen for the treatment of ARV-naive HIV-infected adults. 11 By pairing this single-tablet regimen with 800 mg darunavir (DRV), a 3-class, 2-tablet QD regimen can be constructed to treat HIV-positive patients with select drug-resistant mutations. The pharmacoenhancer cobi coformulated in the single-tablet regimen that increases EVG drug levels would also function to augment the level of DRV, enabling QD dosing of a potent protease inhibitor. There exists, however, some conflicting data that demonstrate that this combination may result in lower levels of EVG and DRV in serum while not resulting in a consequential loss of efficacy. We therefore sought to assess the real-world effectiveness and safety of TDF/FTC/EVG/cobi plus DRV in patients with multidrug-resistant HIV-1 infection.
Methods
Study Design and Population
We conducted a retrospective chart review of all HIV-positive adult patients initiated on TDF/FTC/EVG/cobi plus DRV at the St Michael’s Hospital Positive Care Clinic, Toronto, Ontario, Canada, between September 1, 2013, and March 19, 2015. Eligible patients were identified through manual review of clinic charts. Ethical approval for this study was obtained from the St Michael’s Hospital Research Ethics Board. Demographic data, baseline clinical characteristics, prior ART history, documented genotypic resistance mutations, and concomitant medications prior to the ARV regimen change were collected. Reasons for initiating the new regimen were also recorded.
Objectives
The primary objective was to assess the real-world effectiveness of the novel ARV combination TDF/FTC/EVG/cobi plus DRV 800 mg by determining the proportion of patients with HIV-1 RNA <40 copies/mL at 48 weeks of therapy. The secondary objectives were to (1) quantify the change in CD4 count 48 weeks after initiating TDF/FTC/EVG/cobi plus DRV, (2) characterize treatment-emergent drug-resistance mutations among those with virologic failure, and (3) describe adverse events.
Statistical Analysis
Analyses were primarily descriptive. Demographic and baseline clinical characteristics were summarized using measures of central tendency and dispersion for continuous variables and using frequencies and percentages for categorical variables. For primary analysis, the proportion of patients with HIV viral load <40 copies/mL at 48 ± 6 weeks was calculated using the US Food and Drug Administration (FDA) Snapshot Algorithm. 12 Primary efficacy analyses were conducted on an intent-to-treat basis, including all patients meeting the eligibility criteria, regardless of whether they discontinued therapy or were lost to follow-up during the observation period.
Median change in absolute CD4 count and CD4 percentage were calculated at 48 ± 6 weeks. Drug resistance-associated mutations were interpreted by using both the International Antiviral Society-USA (IAS-USA) 2014 guidelines and the Stanford HIV Drug Resistance Database. 13–14 Adverse events and their relationship with the study regimen were ascertained using information available in the patient chart and based on the treating physician’s opinion. Adverse events including laboratory values were considered as defined by the Division of AIDS Table for Grading the Severity of Adult and Pediatric Adverse Events. 15
Results
Overall, 21 patients were initiated on TDF/FTC/EVG/cobi plus DRV from September 1, 2013, through March 19, 2015. Patient baseline characteristics are shown in Table 1. The median (interquartile range, IQR) age was 43 (33-48) years, duration of HIV diagnosis was 15 (3-22) years, and baseline CD4 count and percentage were 428 (60-579) cells/mm3 and 22 (8-27), respectively. Median (IQR) plasma HIV-RNA level among those with viremia at baseline was 36267 (2206-204 478) copies/mL; 6 (29%) individuals had a viral load <40 copies/mL before the antiretroviral switch. Sixteen were treatment-experienced patients, having been on ART for a median (IQR) of 14 (11-17) years and having tried a median of 3 (2-5.5) prior ARV regimens.
Baseline characteristics of 21 patients initiating TDF/FTC/EVG/cobi plus DRV.
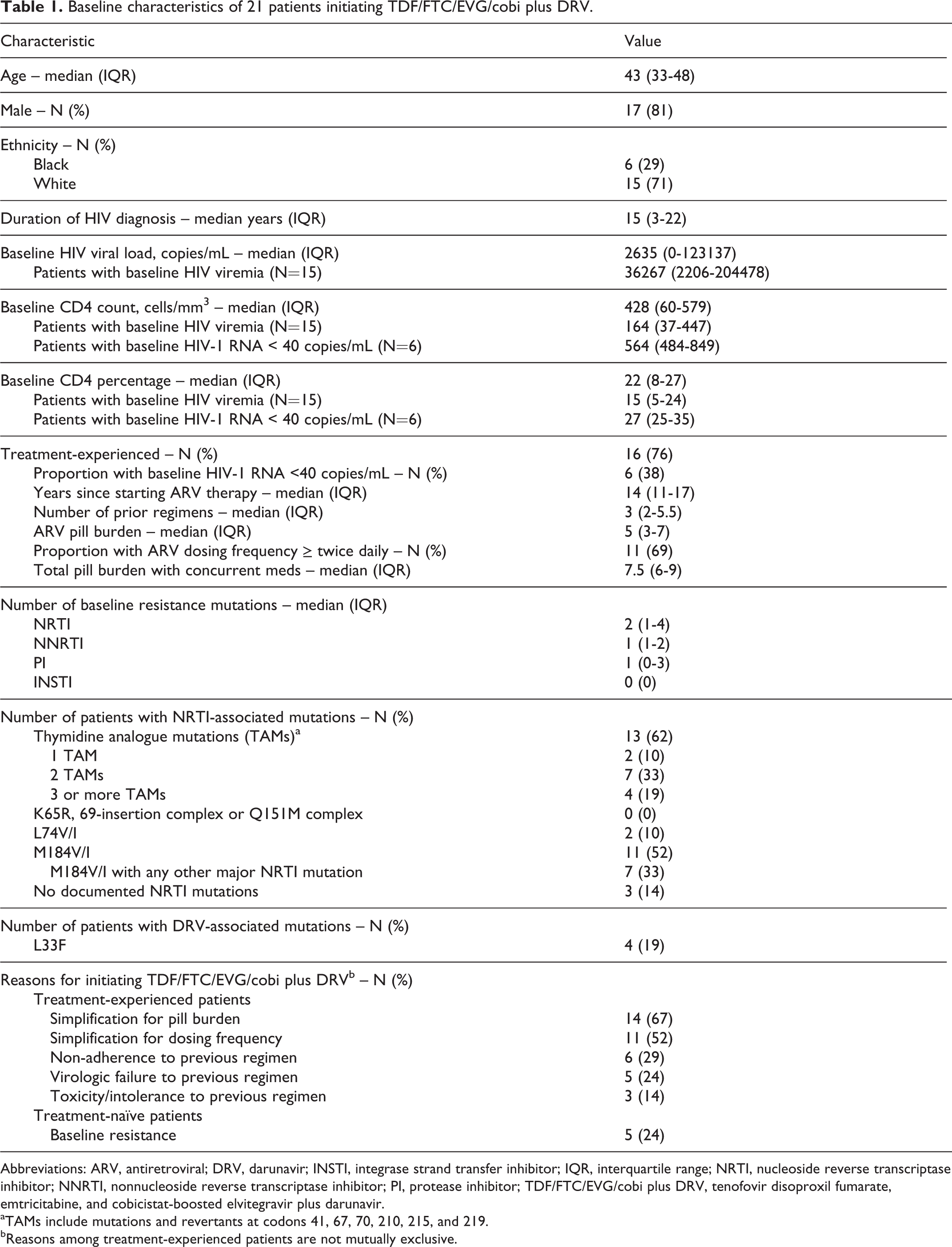
Abbreviations: ARV, antiretroviral; DRV, darunavir; INSTI, integrase strand transfer inhibitor; IQR, interquartile range; NRTI, nucleoside reverse transcriptase inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; PI, protease inhibitor; TDF/FTC/EVG/cobi plus DRV, tenofovir disoproxil fumarate, emtricitabine, and cobicistat-boosted elvitegravir plus darunavir.
aTAMs include mutations and revertants at codons 41, 67, 70, 210, 215, and 219.
bReasons among treatment-experienced patients are not mutually exclusive.
The most common reason for selecting the study regimen was simplification of an existing ARV regimen for pill burden, representing 67% of participants (Table 1). In the 5 treatment-naive patients, TDF/FTC/EVG/cobi plus DRV was initiated due to the presence of acquired triple-class resistance mutations. Baseline genotypic resistance data showed that the median number of nucleoside reverse transcriptase inhibitor (NRTI), nonnucleoside reverse transcriptase inhibitor, and protease inhibitor (PI)-associated mutations was 2, 1, and 1 respectively; although no integrase strand transfer inhibitor (INSTI) mutations were documented. Eleven patients had M184V/I mutations conferring resistance to FTC, 4 patients had 3 or more thymidine analogue mutations, and 4 patients had an L33F mutation, a protease mutation included in the Tibotec DRV genotypic susceptibility score. Using the Stanford algorithm, all patients started on TDF/FTC/EVG/cobi plus DRV were considered to have at least 2 fully active antiretrovirals provided by this combination.
Of the 21 patients included in the study, 18 (86%) were still in follow-up at week 48. Fourteen (67%) had plasma HIV-1 RNA <40 copies/mL at week 48, 2 did not have a viral load result at week 48, and 2 patients had viremia. Both patients who had missing data at week 48 remained virologically suppressed on repeat testing at weeks 68 and 76, respectively. Of the 2 patients who were not suppressed at week 48, 1 patient elected to discontinue ART altogether due to a psychiatric crisis deemed unrelated to ART, despite achieving virologic suppression after 12 weeks of therapy, and the other patient experienced breakthrough viremia after switching to the study regimen. Serum drug concentration monitoring in this latter patient on 2 separate occasions (after 39 and 50 weeks of therapy) did not detect any measureable amounts of FTC, DRV, EVG, or cobi. This patient did not have any DRV mutations, gastrointestinal complaints, and was not taking any medications that could explain the undetectable drug levels. He refilled medications regularly, reported rarely missing doses; however, complete adherence could not be ascertained. Follow-up genotypic resistance tests also revealed no new mutations.
Three patients did not complete 48 weeks of treatment: 2 were lost to follow-up and one had an undetectable viral load at week 24 then subsequently died of malignancy. Table 2 shows that the proportion of patients who had plasma HIV-1 RNA levels <40 copies/mL at week 48 appeared to be similar in those with and without baseline viremia. The median CD4 count and CD4 percentage increased to 561 (IQR, 305-698.5) cells/mm3 and 26 (IQR, 18.5-38.5) at 48 weeks, respectively (Table 3).
Virologic results after 48 weeks of TDF/FTC/EVG/cobi plus DRV.

Abbreviations: ITT, intention-to-treat; TDF/FTC/EVG/cobi plus DRV, tenofovir disoproxil fumarate, emtricitabine, and cobicistat-boosted elvitegravir plus darunavir; SAE, serious adverse event.
Immunologic results after 48 weeks of TDF/FTC/EVG/cobi plus DRV.

Abbreviation: TDF/FTC/EVG/cobi plus DRV, tenofovir disoproxil fumarate, emtricitabine, and cobicistat-boosted elvitegravir plus darunavir; IQR, interquartile range.
No new AIDS-defining events occurred during the study period. One patient died of small cell lung cancer. One patient developed a grade 1 rise in serum creatinine after 24 weeks of therapy, but testing for evidence of TDF-associated proximal tubular damage including glucosuria, a high fractional excretion of phosphate or urine protein to creatinine ratio, was negative. There were no reports of other drug-related toxicities in clinic documentation.
Discussion
TDF/FTC/EVG/cobi with DRV is a simple and compact, 2-tablet QD regimen that incorporates high potency of DRV while exploiting the pharmacokinetic boosting effect of cobi on both EVG and DRV.
In our study, every patient on TDF/FTC/EVG/cobi plus DRV was receiving 2 to 3 fully active drugs from at least 2 different ARV classes without any documented or presumed INSTI resistance. This 2-tablet combination decreased the median daily pill burden by 3 tablets and simplified the daily dosing frequency for the 69% of the treatment-experienced group that had previously used a twice-daily regimen. At week 48, 67% of patients had an undetectable HIV-1 RNA as defined by the FDA Snapshot algorithm, in which patients with missing data are considered failures. However, among patients with available data at 48 weeks, 88% (14 of 16) were undetectable. Of the 2 with viremia at 48 weeks, 1 had suppressed his viral load by week 12 but subsequently discontinued all medications for psychiatric reasons, while in the other, therapeutic drug monitoring on 2 separate occasions suggested nonadherence. Taken together, our data suggest that this regimen is a feasible, well-tolerated, and generally effective treatment regimen for patients with limited treatment options.
These results are notable because this combination of TDF/FTC/EVG/cobi and DRV has the potential of numerous drug–drug interactions. In addition to both EVG and DRV being substrates of CYP450 isoenzyme 3A4, and the potential of EVG to be chelated in the gut by polyvalent cations, drug interactions within this ARV combination have also been identified. A pharmacokinetic study on the combination of EVG 150 mg daily, cobi 150 mg daily, and DRV 800 mg daily demonstrated an approximate 52% and 21% decrease in EVG and DRV
The clinical significance of these changes in serum concentrations is unclear. Although serum concentrations were decreased in some of these studies, they are considered to remain sufficiently above the EVG IC95 and the IC50 for PI wild-type virus.
20–21
In fact, 3 additional studies examined this novel ARV combination or the analogous combination of tenofovir alafenamide (TAF)/FTC/EVG/cobi plus DRV and found favorable results similar to our study.
17–18–22
Ricard et al performed therapeutic drug monitoring on 8 treatment-experienced patients with multidrug-resistant HIV switched to TDF/FTC/EVG/cobi plus DRV and reported that at the end of the observation period 88% of patients had an undetectable HIV viral load.
17
In the cohort studied by Diaz et al, 20 treatment-experienced patients were switched to TDF/FTC/EVG/cobi plus DRV and reported that 88% of the patients with data at 24 weeks had an undetectable viral load.
18
Also, in a recent open-label randomized control study of 135 virologically suppressed patients with multidrug-resistant HIV, 97% of the 89 patients switched to a similar combination tablet of TAF/FTC/EVG/cobi plus DRV 800 mg daily remained virologically suppressed at 24 weeks.
22
In that study, both EVG and DRV mean
The ARV regimen used in this study was generally well tolerated. Only one grade 1 adverse event involving an increased serum creatinine was observed, and it did not lead to treatment discontinuation during the study period. In contrast, in the retrospective study by Diaz et al, 25% of participants had worsening creatinine clearance while on the combination treatment. 18 Potential causes of such changes include cobi-induced inhibition of the tubular secretion of creatinine, which usually causes a 10% rise in serum creatinine within the first few weeks of therapy, 23 and true renal dysfunction from TDF, which has been associated with serious renal dysfunction including Fanconi syndrome in up to 1.5% of patients. 23 Alternatively, clinicians may wish to consider TAF/FTC/EVG/cobi plus DRV, as studied by Huhn et al, since none of their 89 patients developed a serious renal adverse event; overall, they showed improvements in markers of proximal tubular proteinuria compared to patients who remained on their baseline regimen. 22
Limitations of our study include the open-label, nonrandomized design, the lack of a comparator treatment arm, and the modest sample size. Another limitation of our study is the nature of a retrospective chart review, which precludes a standardized approach to assessing treatment intolerance, adverse events, and satisfaction in every patient.
In this small observational sample, QD TDF/FTC/EVG/cobi plus DRV appears safe, well tolerated and effective in suppressing HIV drug-resistant virus in selected patients. This simple 2-tablet ART option and the closely related regimen TAF/FTC/EVG/cobi plus DRV, warrant further study including pharmacokinetic investigations and correlation with longer-term outcomes.
Footnotes
Acknowledgements
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
