Abstract
We report the first identified case of suspected iatrogenic adrenal insufficiency after an interlaminar injection of triamcinolone acetonide while on concomitant Stribild (elvitegravir 150 mg/cobicistat 150 mg/tenofovir disoproxil fumarate 300 mg/emtricitabine 200 mg [EVG/c/TDF/FTC]). A 49-year-old female with HIV on EVG/c/TDF/FTC therapy presented to our endocrinology clinic to be evaluated for suspected Cushing syndrome. Prior to presentation, the patient had been given 2 interlaminar spinal injections of triamcinolone. Thereafter, she developed a swollen face, had unexplained weight gain, and fatigue. Cosyntropin stimulation test was positive for adrenal insufficiency. By applying the Naranjo Nomogram for Causality and the Drug Interaction Probability Scale to this drug–drug interaction, we calculated a score of 6 (probable) and 5 (probable), respectively. Symptoms resolved without further intervention. The EVG/c/TDF/FTC contains cobicistat, a strong cytochrome P450 3A4 (CYP3A4) inhibitor, which can potentiate drug interactions involving metabolizing of medications via this pathway. Clinicians are reminded to be vigilant while assessing the potential pharmacokinetic drug interactions not mentioned by the manufacturer.
Introduction
Stribild is a single-tablet daily highly active anti-retroviral therapy (HAART) regimen approved by the US Food and Drug Administration in 2012 for HIV antiretroviral-naive patients. This regimen comprised elvitegravir 150 mg, cobicistat 150 mg, tenofovir disoproxil fumarate 300 mg, emtricitabine 200 mg (EVG/c/TDF/FTC). 1 The EVG functions as an HIV integrase strand inhibitor, cobicistat as a cytochrome P450 3A4 (CYP3A4) inhibitor to boost EVG activity, and TDF and FTC as nucleotide/nucleoside analog reverse transcriptase inhibitors. A concern with the administration of EVG/c/TDF/FTC is its potential for pharmacokinetic drug interactions with the concurrent use of other medications utilizing similar metabolic pathways. These interactions may be clinically significant even with medication administration via atypical routes. In this study, we present a case of suspected iatrogenic adrenal insufficiency secondary to interlaminar injection of triamcinolone acetonide.
A MEDLINE search (1950-August 2016) was conducted with a combination of the following terms: “Stribild,” “Cushing’s syndrome,” “Adrenal suppression,” “Corticosteroids,” “Cobicistat,” “Elvitegravir,” “Emtricitabine,” “Tenofovir,” “Steroid,” “Interaction,” and “HAART therapy.” The development of drug-induced Cushing syndrome has been well-documented, and there are several case reports in the literature of drug-induced Cushing syndrome due to the CYP3A4 inhibition of ritonavir (RTV) when used with triamcinolone. 2 –11 No published case reports of adrenal insufficiency with EVG/c/TDF/FTC and triamcinolone were found. There was one case report of intranasal fluticasone with concomitant EVG/c/TDF/FTC causing iatrogenic adrenal insufficiency. 12 We report the first case of probable iatrogenic adrenal suppression secondary to the use of EVG/c/TDF/FTC with the administration of interlaminar injection of triamcinolone.
Case Report
We present the case of a 49-year-old female who received outpatient care at our institution’s clinic network. Her past medical history was significant for HIV/AIDS (history of AIDS-defining illness with pneumocystis pneumonia) on HAART, hyperlipidemia, pelvic inflammatory disease, and a history of lower back pain after falling from a flight of stairs. She reported no known drug allergies.
The patient’s HAART regimen was switched from atazanavir (ATV) 300 mg orally daily, RTV 100 mg orally daily, abacavir (ABC) 600 mg orally daily, and lamivudine (3TC) 300 mg orally daily to EVG/c/TDF/FTC one tablet orally daily. At a subsequent clinic visit 6 months later, the patient reported worsening lower back pain with a scale of 7/10 on the pain scale. She said the pain exacerbated while sitting for a long time, without radiation of pain, and that naproxen was not providing her with any significant relief. The patient was then referred to the pain management service for possible epidural spinal injection, and she was prescribed meloxicam 15 mg orally daily.
At a follow-up pain management visit, her medication regimen was switched to meloxicam 15 mg orally daily with gabapentin 400 mg orally at bedtime. One month later, she showed no improvement. At this time, gabapentin was increased to 400 mg orally 3 times a day, and an interlaminar epidural spinal injection of triamcinolone acetonide 80 mg was administered. Months later, the patient’s back pain symptoms flared up again despite maintaining oral therapy, and she received a second interlaminar epidural spinal injection of triamcinolone 80 mg.
One month later, the patient was diagnosed with a deep venous thrombosis of the right popliteal vein and was started on enoxaparin therapy. At her follow-up with her primary care physician several days later, the physician noted the patient’s cheeks to be visibly enlarged and puffy, suggestive of a cushingoid appearance. She confirmed that a month prior she had noticed her face becoming more “puffy,” and painfully swollen across her cheeks and jawline. The swelling was severe enough to cause constant pain and she stated that her friends and family had noticed a rapid change in her appearance. Patient stated the swelling had no discharge or redness. It was evenly distributed and cosmetically disfiguring. She also noted mild fatigue; however, she denied other symptoms of Cushing syndrome such as fatty deposition on her upper back and neck, striae of her abdomen, thinning of skin, impaired wound healing, and muscle weakness. Physical examination concurred with no apparent dorsocervical fat pad or striae noted. The patient’s weight had increased from 128 to 137 pounds the year prior, and she denied any change in eating habits or activity. She had not experienced such a weight fluctuation before. At the time, her CD4 count was 350 cells/mm3; and her viral load was not checked at the time.
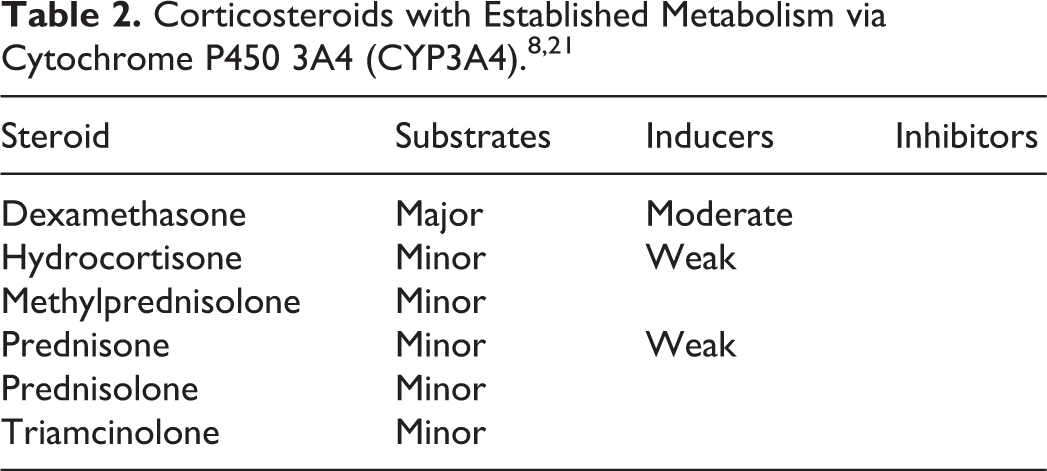
Current medications included enoxaparin 60 mg subcutaneously daily, EVG/c/TDF/FTC one tablet orally daily, rosuvastatin 20 mg orally daily, meloxicam 15 mg orally daily, gabapentin 300 mg orally at bedtime, and fluoxetine 10 mg orally daily. Her medications included zolpidem 10 mg as needed for sleep, naproxen 500 mg orally every 12 hours based on pain, sennosides 8.6 mg every 12 hours as needed for constipation, and butalbital/acetaminophen/caffeine one tablet every 4 hours but not to exceed 6 tablets daily. The patient’s adrenocorticotropic hormone (ACTH) level was maintained low at <5 pg/mL (reference range 6-50 pg/mL; Figure 1). Cortisol was not measured during the visit.
Adrenocorticotropic hormone (ACTH) measurements.
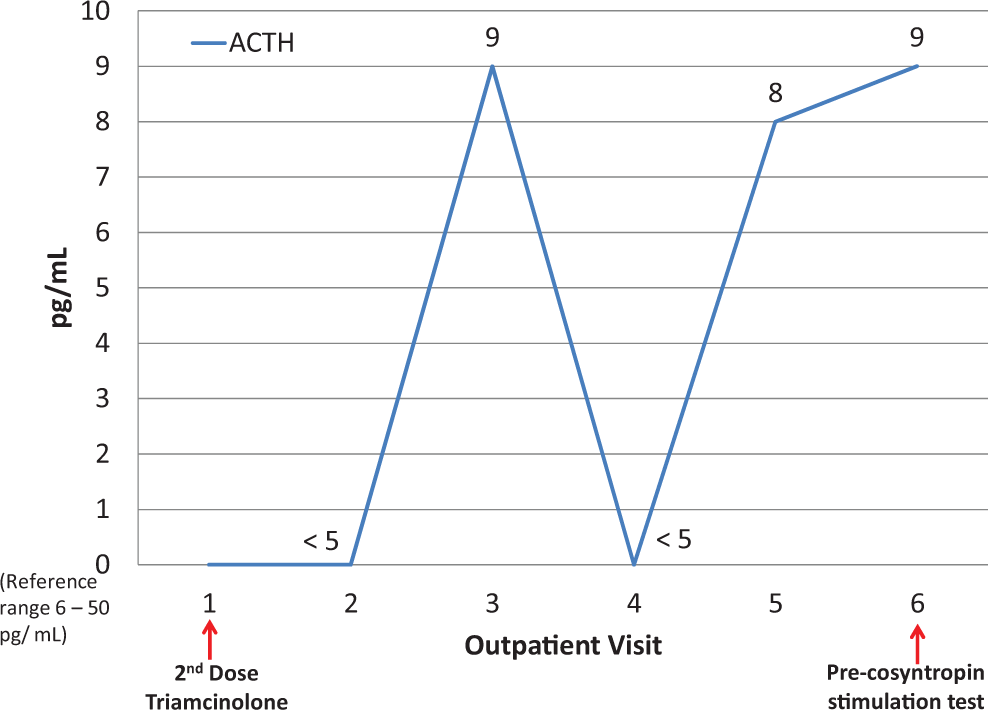
The patient was referred to outpatient endocrinology for suspected Cushing syndrome. At this initial endocrinology visit, physical examination concurred with cushingoid appearance and the ACTH level was rechecked and found to be <5 pg/mL (Figure 1). Further blood work showed her cortisol level was at 1.0 μg/dL (reference range 6.7-22.6 μg/dL; Figure 2). Patient was counseled on symptoms of steroid deficiency and was closely monitored. A cosyntropin stimulation test was scheduled to administer a supraphysiologic dose of synthetic ACTH, which showed the expected result of elevated cortisol ≥18 μg/dL in the setting of proper adrenal function. The patient’s cortisol level was 1.4 μg/dL pretest and 6.1 μg/dL posttest, indicating adrenal insufficiency 13 (Figure 2). The ACTH level was 9 pg/mL pretest (Figure 1).

Cortisol measurements.
At this time, patient’s facial swelling was no longer painful but was still present and she felt less fatigue. Her ACTH and cortisol levels were not measured. Follow-up clinic visits describe resolution of cushingoid face and she could resume her normal activities without further intervention. Patient was continued on her EVG/c/TDF/FTC therapy and did not receive further corticosteroid injections.
Discussion
The management of HIV in recent years has been ameliorated by the development of various HAART regimens, allowing patients to live longer and preventing the development of opportunistic infections and progression to AIDS. 14 The National Institute of Health recommends initiating HAART for all HIV-positive patients, regardless of CD4 count, to maximally inhibit the replication of virus and to decrease the risk of morbidity and mortality associated with the infection. 15 Regimens that are recommended as first-line treatment are either integrase strand transfer inhibitor–based combinations or protease inhibitor-based combinations. The HAART regimens generally consist of 3 to 4 medications, a complicated predicament for all patients who require lifelong therapy. In recent years, combination, once-daily treatments have been developed, which have shown increased patient compliance because of the more cumbersome nature of older regimens. 16 One such combination therapy, containing a newer integrase strand transfer inhibitor, EVG, is EVG/c/TDF/FTC. 1,17
Of the 4 drug entities within the product, FTC and TDF are not significantly metabolized and do not affect the metabolic enzymes and transporters. 17 The components that are cause potentially clinically significant drug interactions are cobicistat and EVG. The EVG is mostly metabolized by CYP3A4 and to a lesser extent via glucuronidation. Cobicistat is metabolized primarily by CYP3A4 and undergoes some minor metabolism by CYP2D6. Cobicistat is a potent CYP3A4 inhibitor and will likely increase the concentration of medications metabolized via this enzyme. 17 Other enzymes within the CYP450 family are weakly affected by cobicistat. Both EVG and cobicistat are CYP3A4 substrates. Given concomitantly with strong inducers, concentrations can decrease significantly and put the patient at risk of treatment failure. Because cobicistat is a CYP3A4 substrate and inhibitor and because EVG is a CYP3A4 substrate, according to the manufacturer, the coadministration of Stribild is contraindicated with major CYP3A4 substrates and strong CYP3A4 inducers. 1 Table 1 summarizes further details the metabolism of the 4 components.
Metabolism/Transport Effects of the Components of EVG/c/TDF/FTC. 1
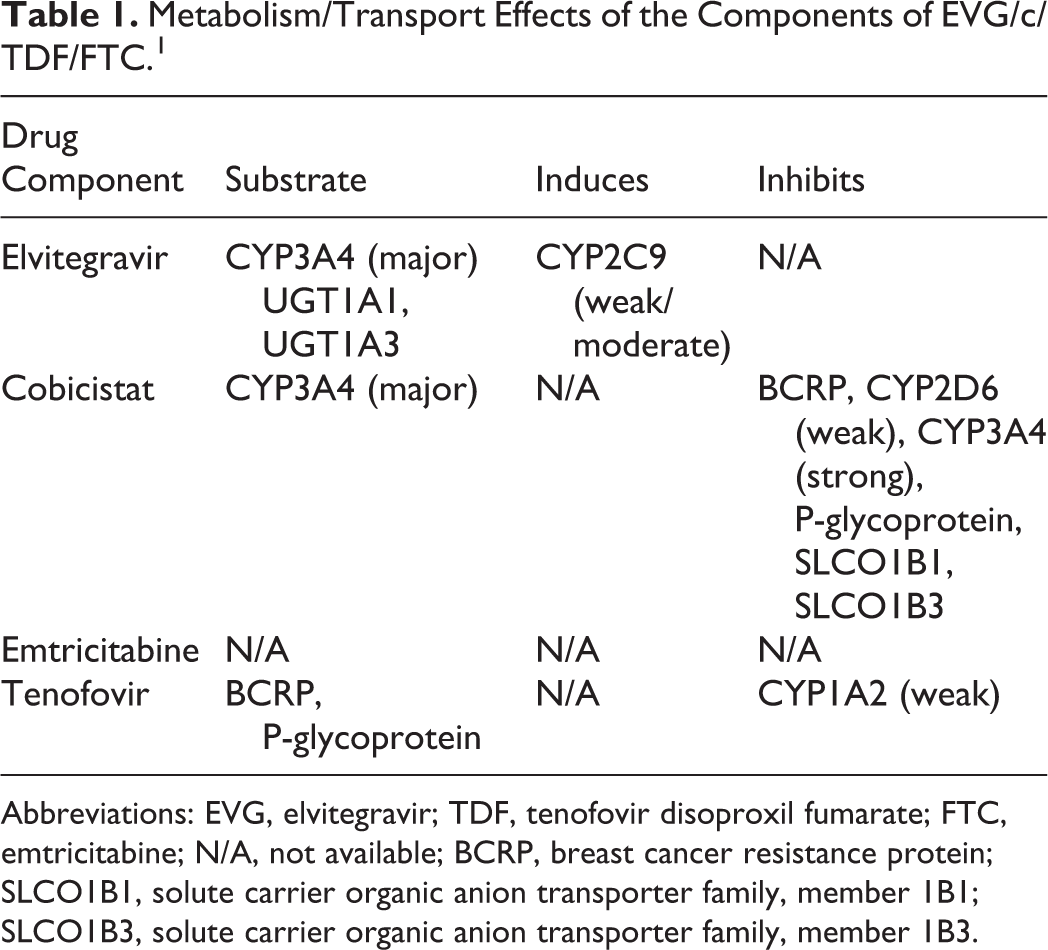
Abbreviations: EVG, elvitegravir; TDF, tenofovir disoproxil fumarate; FTC, emtricitabine; N/A, not available; BCRP, breast cancer resistance protein; SLCO1B1, solute carrier organic anion transporter family, member 1B1; SLCO1B3, solute carrier organic anion transporter family, member 1B3.
Corticosteroids are commonplace in clinical practice for their various immunosuppressant and anti-inflammatory effects, and they are utilized in acute and chronic conditions. 18 The use of corticosteroids at higher dosages and for longer durations comes with increased risk of various adverse effects. Potential long-term adverse effects of corticosteroids include muscle wasting, osteoporosis, skin manifestations including impaired wound healing, striae, skin thinning, easy bruising, and hypersensitivity reactions, edema and hypertension, ocular disorders including glaucoma, cataracts, and exophthalmos, hyperglycemia, weight gain, central nervous system effects, hypocalcemia, hypokalemia as well as adrenal insufficiency and opportunistic infections. 18
Triamcinolone is a long-acting corticosteroid with minimal sodium-retaining potential. 19 It undergoes hepatic metabolism forming 3 identified metabolites significantly less active than the parent compound and is also a minor substrate of CYP3A4. Protein binding is approximately 68%, with an elimination half-life of 88 minutes and a biologic half-life of 12 to 36 hours. Elimination occurs via the urine and feces, 40% and 60%, respectively. Epidural steroid injections are a common pain management procedure, which are administration into the epidural space via 3 major routes: transforaminal, interlaminar, and caudal. 20 Steroids commonly utilized for these administration routes include dexamethasone, methylprednisolone, and triamcinolone. As with any other systemic corticosteroid, systemic adverse effects can occur with epidural administration of steroids. There are no clear guidelines regarding the frequency of epidural corticosteroid injections, and duration of effect varies on a patient-by-patient basis, but a repeat injection should not be given less than 2 weeks apart to prevent adverse effects and allow for proper assessment of the full response.
Hepatocellular molecular studies as well as human reports have demonstrated that corticosteroids are affected by the CYP450 system 8,21 (Table 2). In certain scenarios, these effects may manifest as clinically relevant drug interactions with systemic adverse events. When administering a corticosteroid with a strong CYP inhibitor, one clinically significant interaction is the potential for adrenal insufficiency. 22
When given in high doses and/or for long durations, glucocorticoids may cause secondary adrenal insufficiency by suppressing the release of corticotropin from the pituitary gland and thereby causing decreased secretion of endogenous corticosteroids. 18 Acute adrenal insufficiency may occur if patients are withdrawn from corticosteroids too rapidly after a prolonged duration of use, and therefore, tapering is generally recommended. When given in combination with an inhibitor of the CYP3A4 system, this risk of adrenal insufficiency may be increased because of the pharmacokinetic drug–drug interaction. 22
The manufacturer of EVG/c/TDF/FTC cites 2 corticosteroids as having the potential for drug–drug interactions, systemic dexamethasone and inhaled/nasal fluticasone. 1 The mechanism for the dexamethasone interaction is attributed to the potential for decreasing EVG and cobicistat levels via CYP3A4 induction. Fluticasone is not recommended to be used concomitantly with EVG/c/TDF/FTC because of the potential for increasing the serum concentration of fluticasone and iatrogenic adrenal suppression, despite nasal administration. 1 Data regarding the use of other corticosteroids in combination with EVG/c/TDF/FTC are limited. Although the implicated drug interaction here is likely attributed to the CYP3A4 inhibition of the cobicistat component, some integrase inhibitors metabolized as CYP3A4 substrates can potentially interact with corticosteroids via different mechanisms. In one pharmacokinetic study of 12 healthy participants, coadministration of dolutegravir and high-dose prednisone resulted in non-clinically significant elevations in dolutegravir concentration due to prednisone CYP3A4 induction. 23
The challenges to treating the HIV-positive patient are multifaceted. The HAART regimens often require manipulation of the CYP450 enzymatic system to boost the regimen components, making drug–drug interactions a potential complication in effectively and safely treating patients. 24 Ritonavir-boosted regimens have been well established as offenders in causing drug–drug interactions, as is demonstrated with case reports attributing adrenal insufficiency to interactions with RTV and triamcinolone. 2 –11 We could not find published case reports describing the pharmacokinetic interaction between EVG/c/TDF/FTC and triamcinolone injections causing adrenal insufficiency. The EVG/c/TDF/FTC has been found to interact with fluticasone, and there are reports of adrenal insufficiency. When applying the Naranjo Nomogram for Causality and the Drug Interaction Probability Scale to this drug–drug interaction, we calculated a score of 6 (probable) and 5 (probable), respectively. 25,26 After cessation of the triamcinolone injections, our patient did not report additional signs of adrenal insufficiency.
Conclusion
HIV-infected patients maintained on multiple medications often metabolized via the CYP450 system are predisposed to potentially significant drug–drug interactions. It is important for the clinician to remember that corticosteroids given via various routes of administration and intermittently can cause significant side effects in patients, and a medication history should always be comprehensive enough to include intermittent medication administration, over-the-counter and herbal products, and diet. Clinicians administering EVG/c/TDF/FTC to their patients should be cognizant of this potential interaction with triamcinolone as well as other pharmacokinetic interactions involving CYP3A4 inhibition not recognized yet in the literature.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.