Abstract
Purpose:
The purpose of this study is to assess postmarketing safety and tolerability of Stribild (elvitegravir [EVG]/cobicistat [COBI]/tenofovir disoproxil fumarate [TDF]/emtricitabine [FTC]).
Methods:
A retrospective, pharmacoepidemiologic study in 2 outpatient HIV clinics in the Southeast United States was conducted among adults receiving EVG/COBI/TDF/FTC. We evaluated incidence and treatment-related adverse events, including change in serum creatinine (SCr).
Results:
Patients were primarily treatment experienced (n = 173, 60%), African American (n = 210, 73%), and males (n = 187, 65%). One hundred ninety-five (68%) patients had any increase in SCr, and 65 (23%) had an increase of ≥0.3 mg/dL. Mean SCr change from baseline to peak was 0.2 mg/dL. Being treatment experienced (odds ratio [OR] = 2.21, 95% confidence interval [CI]: 1.12-4.38) was associated with SCr ≥0.3 mg/dL, while body mass index ≥30 kg/m2 (OR = 0.41, 95% CI: 0.18-0.93) was protective. Twenty (7%) patients discontinued therapy, 3 due to acute kidney injury.
Conclusion:
Our results demonstrate limited adverse events and low discontinuation rates associated with EVG/COBI/TDF/FTC.
Introduction
Over 1.1 million people are living with HIV in the United States, disproportionally impacting the Deep South (Alabama, Georgia, Louisiana, Mississippi, North Carolina, and South Carolina). 1,2 In 2012, South Carolina and Alabama ranked 10th and 18th, respectively, in annual HIV diagnosis rate. 1 Rural cases of HIV/AIDS in the Deep South continue to drive high disease burden and are critical for population evaluation. 1 Demographics of HIV-infected individuals in the Southeast United States demonstrate a comparatively lower incidence among men who have sex with men and higher incidence among African Americans. 1,3
Potent combination antiretroviral therapy continues to be the standard for treatment, with a strong emphasis on single-tablet, once-daily regimens. Stribild (elvitegravir [EVG], cobicistat [COBI], tenofovir disoproxil fumarate [TDF], and emtricitabine [FTC]) was US Food and Drug Administration approved in August 2012 as a fixed-dose, once-daily integrase inhibitor–based combination tablet for treatment-naive patients and is a preferred regimen in the 2016 US Department of Health and Human Services Guidelines for treatment-naive patients. 1,2,4 These new guidelines now include EVG, COBI, tenofovir alafenamide (TAF), and FTC for initial therapy in patients with estimated creatinine clearance (CrCl) ≥30 mL/min. 4 Tenofovir alafenamide is a novel prodrug that results in 90% lower serum concentrations compared to TDF. Large comparator studies demonstrate consistent efficacy when compared to TDF with lower systemic adverse effects (AEs) including nephrotoxicity. 4 Given the recent availability of such combinations, TAF-based regimens will not be discussed further in this article.
Cobicistat, a component of EVG/COBI/TDF/FTC, is a novel pharmacokinetic boosting agent with no direct activity against the retrovirus. 4,5 In addition to inhibiting the cytochrome P450 3A4 enzyme, resulting in increased EVG serum concentrations, COBI is a potent inhibitor of the multidrug and toxin extrusion-type transporter 1 (MATE-1) mediating the tubular secretion of creatinine. The resulting increases in serum creatinine (SCr), however, do not reflect reduction in glomerular function based on iohexol studies. 6 These effects were seen in phase 1 and 2 clinical trials as early as week 1 study visits. 6 –8 The package labeling states patients should not be initiated on EVG/COBI/TDF/FTC with baseline CrCl <70 mL/min and should be discontinued if CrCl falls below 50 mL/min. Close monitoring is suggested for patients with an increase in SCr >0.4 mg/dL. 5
In treatment-naive patients in phase 3 clinical trials, EVG/COBI/TDF/FTC has been shown to be well tolerated. In a noninferiority study comparing efavirenz (EFV)/TDF/FTC and EVG/COBI/TDF/FTC, Sax, Zolopa, and colleagues demonstrated lower rates of AEs including central nervous system effects, lipid changes, and rash when compared to EFV/TDF/FTC. 9,10 There was a modest increase in nausea in the EVG/COBI/TDF/FTC arm. Also, discontinuation rates at week 96 due to AEs were approximately 5% in the EVG/COBI/TDF/FTC arm. 10 With respect to renal outcomes, Sax et al demonstrated an increase in SCr of approximately 0.15 mg/dL in the EVG/COBI/TDF/FTC arm by week 2, which increased until week 8 where it plateaued through the end of week 48 and continued through week 96. 9,10 One patient discontinued EVG/COBI/TDF/FTC due to a renal adverse event, which was reversed upon drug discontinuation. 11 DeJesus et al confirmed tolerability among treatment-naive patients, demonstrating lower rates of AEs among EVG/COBI/TDF/FTC–treated patients when compared to those receiving ritonavir-boosted atazanavir (ATV/r) plus coformulated TDF/FTC. 11
Treatment-experienced patients have demonstrated similar tolerability of EVG/COBI/TDF/FTC. A switch study (treatment-experienced patients continuing a nonnucleoside reverse transcriptase inhibitor [NNRTI] plus TDF/FTC versus changing therapy to EVG/COBI/TDF/FTC) demonstrated overall AEs occurred at similar rates between groups (75% compared to 81% in the EVG/COBI/TDF/FTC arm). 12 Reported increases in SCr from baseline were consistent with treatment-naive data. Those on EVG/COBI/TDF/FTC experienced mild elevations in SCr (median 0.12 mg/dL) compared to those receiving EFV/TDF/FTC (decrease by 0.01 mg/dL), and 1 patient discontinued EVG/COBI/TDF/FTC due to acquired Fanconi syndrome. 12 Arribas et al demonstrated no discontinuations due to renal toxicities in their treatment simplification study. 13 Collectively, in both treatment-naive and treatment-experienced patients treated with EVG/COBI/TDF/FTC, renal changes that resulted in treatment discontinuation were renal failure, increased SCr, and Fanconi syndrome. In most instances, signs of proximal tubulopathy were observed in patients with renal impairment at baseline including evidence of proteinuria, hypophosphatemia, and glucosuria, in patients with glomerular filtration rate (GFR) <70 mL/min, or in patients taking a TDF-containing regimen at screening. 9 –13
Despite controlled, randomized clinical trial data, there are limited postmarketing data demonstrating renal safety outcomes and patient tolerability of EVG/COBI/TDF/FTC therapy, especially among African Americans in the Southeast United States. The purpose of this study was to assess safety and tolerability of EVG/COBI/TDF/FTC therapy in treatment-naive and treatment-experienced patients receiving care in the Southeast United States. The impact of host factors on rates of AEs, specifically related to changes in SCr, was evaluated.
Methods
This multicenter, retrospective, observational, pharmacoepidemiologic cohort study was conducted at 2 sites in the Southeast United States: the Immunology Clinic at the University of South Carolina School of Medicine in Columbia, South Carolina, and the Copeland Care Clinic in Montgomery, Alabama. Institutional review board expedited approval was granted from both study sites prior to study initiation. A waiver of consent was granted given the retrospective nature of the study. Electronic medical records were screened retrospectively for EVG/COBI/TDF/FTC initiation, dating back to September 1, 2012, and followed through August 1, 2013. Patients ≥18 years of age with a documented SCr pre- and post-EVG/COBI/TDF/FTC initiation were included. Patients were excluded if they were receiving any type of renal replacement therapy or laboratory data were unavailable. Data were collected using clinic visit documentation and/or electronic medical records during the study period.
Data collected included demographic data (height, weight, age, sex, and race/ethnicity), reason(s) for initiation of EVG/COBI/TDF/FTC, hospitalization during therapy (yes/no), treatment naive or treatment experienced, concomitant disease states (eg, congestive heart failure, chronic kidney disease with stage, liver disease, other cardiovascular diseases, such as hypertension and/or hyperlipidemia, and cerebrovascular disease), EVG/COBI/TDF/FTC refill records, and treatment-related AEs. Data collected at baseline and at each subsequent study visit were SCr, CD4 count, HIV viral load, and concurrent nephrotoxic agents including sulfamethoxazole/trimethoprim (SMX-TMP), nonsteroidal anti-inflammatory agents, diuretics, acyclovir, or valacyclovir. Reasons for discontinuing EVG/COBI/TDF/FTC were documented for each patient as applicable. Creatinine clearance was calculated using Cockcroft-Gault formula. 14
Analysis
Predefined subgroups were set a priori for analysis. Patients with an increase in SCr of ≥0.3 mg/dL and those patients who had an increase in SCr of ≥0.5 mg/dL were analyzed. Subgroups were defined based on the Acute Kidney Injury Network (AKIN) criteria and the risk stratification in the package labeling, which indicates close monitoring is required for SCr elevations >0.4 mg/dL equating to 0.5 and above in clinical practice. 5 It was therefore decided to utilize 2 cohorts of patients for analysis, those with increases of 0.3 as defined by the AKIN criteria and those >0.4 as described in the package labeling. Data were analyzed using descriptive statistics. Frequencies and percentages were reported for categorical variables, and mean and median were reported for continuous variables. Multivariate logistic regression model was implemented to evaluate the factors associated with an increase in SCr of ≥0.3 mg/dL. Covariates included age, race, gender, body mass index (BMI), CD4 count, HIV viral load, and finally, concurrent nephrotoxic agents and comorbid conditions. Kaplan-Meier analysis to demonstrate the differences in treatment approaches in terms of SCr change was performed. All statistical tests were conducted at a significance level of .05. Analysis was performed using SAS software, version 9.3 (SAS Institute).
Results
During the study period, 306 patients initiated on EVG/COBI/TDF/FTC at the 2 study sites were screened for study inclusion. Twenty patients were excluded from evaluation, primarily due to lack of baseline or follow-up SCr values. Overall, 286 patients were included in the analysis with an average follow-up of 226 days. Patients were primarily African American (n = 210, 73%) and males (n = 187, 65%) with a mean age of 39 years (range: 18-75 years; Table 1). Psychiatric disorders were the most predominant comorbid condition (n = 117, 41%; Table 1). Ninety-seven (34%) patients were classified as obese (BMI ≥30 kg/m2). The reason for EVG/COBI/TDF/FTC initiation was initial regimen (ie, treatment naive) in 41%, simplification of therapy in 30%, and those who had experienced AEs on previous regimens (17%; Figure 1).
Baseline Characteristics.a
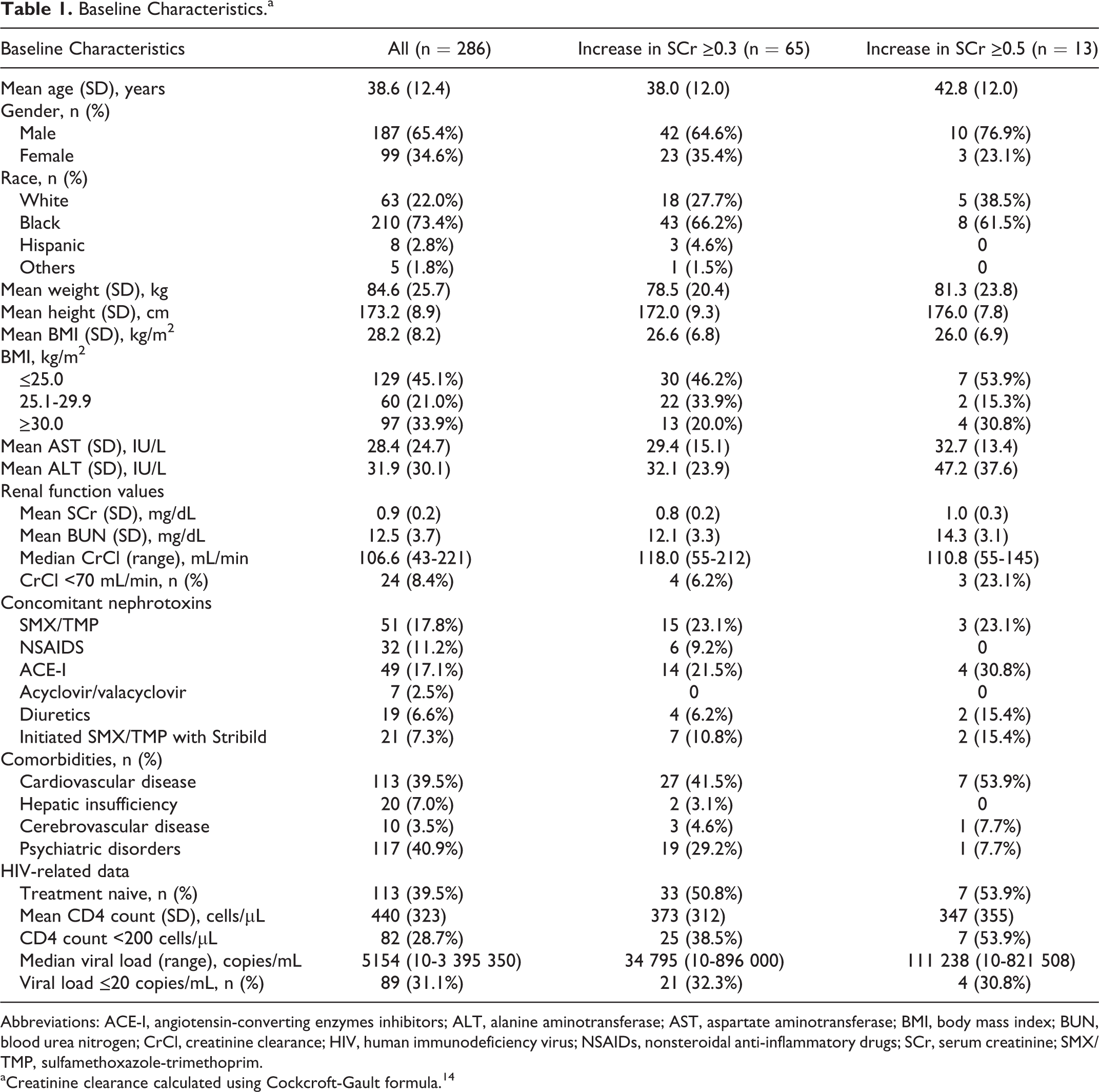
Abbreviations: ACE-I, angiotensin-converting enzymes inhibitors; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; BUN, blood urea nitrogen; CrCl, creatinine clearance; HIV, human immunodeficiency virus; NSAIDs, nonsteroidal anti-inflammatory drugs; SCr, serum creatinine; SMX/TMP, sulfamethoxazole-trimethoprim.
aCreatinine clearance calculated using Cockcroft-Gault formula. 14

Reasons for starting EVG/COBI/TDF/FTC. Reinitiation of therapy occurred in 3% of patients (included in the new start patients). Other includes less strict meal requirements, pregnancy, prior noncompliance, work-related changes, and not documented. ADE, adverse drug event; COBI, cobicistat; EVG, elvitegravir; FTC, emtricitabine; TDF, tenofovir disoproxil fumarate.
Patients were primarily treatment experienced (n = 173, 60%) and had received EFV/TDF/FTC (n = 39), ATV/r with TDF/FTC (n = 28), or raltegravir with TDF/FTC (n = 28) prior to EVG/COBI/TDF/FTC initiation. One hundred forty-two (50%) patients were on TDF prior to EVG/COBI/TDF/FTC initiation. One-third of the population had an undetectable viral load (<20 copies/mL), and the mean baseline CD4 count was 440 cells/µL. The average SCr at baseline was 0.9 mg/dL, correlating with a mean CrCl of 109 mL/min.
One hundred ninety-five (68%) patients had any increase in SCr from baseline, among those, 65 (23%) patients had an increase of ≥0.3 mg/dL and 13 (5%) patients had an increase of ≥0.5 mg/dL (Table 2). Twenty-four (8.4%) patients were initiated on EVG/COBI/TDF/FTC with a baseline CrCl <70 mL/min. At initial follow-up visit, an average of 65 days from baseline, mean increase in SCr was 0.1 mg/dL correlating with a calculated CrCl decrease of 5.1 mL/min.
Renal Outcomes on EVG/COBI/TDF/FTC Therapy.a,b,c

Abbreviations: COBI; cobicistat; CrCL, creatinine clearance; EVG, elvitegravir; FTC, emtricitabine; SCr, serum creatinine; SD, standard deviation; TDF, tenofovir disoproxil fumarate.
aAverage time to the first follow-up was 65 days. bAverage number of days between baseline and peak was 111. cAverage number of days between EVG/COBI/TDF/FTC start and last follow-up was 226.
Comparing baseline and peak SCr values, the average SCr change was 0.2 mg/dL and calculated CrCl decrease of 16.1 mL/min; average time from baseline to peak SCr was 111 days. Patients with SCr of ≥0.3 mg/dL (n = 65) and ≥0.5 mg/dL (n = 13) had an average decline in calculated CrCl of 41 and 42 mL/min, respectively. Increases in SCr were similar at both study sites with average peak increases of approximately 0.16 and 0.17 mg/dL at the South Carolina Immunology Clinic and Copeland Care Clinic, respectively.
In the logistic regression, being treatment experienced (odds ratio [OR] = 2.21, 95% confidence interval [CI]: 1.12-4.38) was associated with an increase in SCr of at least 0.3 mg/dL. Conversely, having a BMI >30 kg/m2 was protective (OR = 0.41, 95% CI: 0.18-0.93, P = .002) of elevations in SCr. Baseline undetectable viral load and CD4 count >200 cells/mm3 were associated with nonsignificant reductions in SCr elevations. No other variables were found to be associated (Table 3).
Associated Factors in Patients with a SCr Increase of ≥0.3 mg/dL.

Abbreviations: ACE-I, angiotensin-converting enzymes inhibitors; ARV, antiretroviral; BMI, Body Mass Index; CI, confidence interval; CrCl, creatinine clearance; OR, odds ratio; SMX/TMP, sulfamethoxazole-trimethoprim.
Forty-one (14.3%) of the 286 patients experienced a nonrenal, treatment-related AEs, primarily gastrointestinal (n = 26) followed by fatigue (n = 3), headache (n = 3), and skin manifestations (n = 3). Other AEs included dizziness, body aches, weight gain, hot flashes, altered mental status, edema, and increased liver function tests (Figure 2). The majority was considered mild in nature.

Nonrenal adverse events on EVG/COBI/TDF/FTC therapy. Other reasons include gallbladder pain, weight gain, body aches, altered mental status, edema, and hot flashes. COBI, cobicistat; EVG, elvitegravir; FTC, emtricitabine; TDF, tenofovir disoproxil fumarate.
Overall, 20 patients discontinued therapy: 5 from AEs including fatigue, gastrointestinal complications, and dizziness, 5 due to virologic failure with documented integrase resistance, 3 due to acute kidney injury (AKI), 2 due to economic burden, and 5 for undefined reasons (Figure 3). Two of the 3 patients who discontinued due to AKI had elevations in SCr ≥0.5 mg/dL by their first follow-up appointment, an average 49 days prior to discontinuation. Kaplan-Meier analysis showed similar changes in SCr over time when comparing treatment-naive and treatment-experienced patients (Figure 4).

Reasons for EVG/COBI/TDF/FTC discontinuation. AKI, acute kidney injury; COBI, cobicistat; EVG, elvitegravir; FTC, emtricitabine; TDF, tenofovir disoproxil fumarate.

Probability of increased SCr: treatment naive versus treatment experienced. SCr, serum creatinine. Time to Peak SCr represented in months.
Significant declines in viral load were observed among all patients including those with an increase in SCr. Compared to the baseline, the mean decrease in viral loads was 4.9 log10 upon the first follow-up and 5.1 log10 at the last follow-up. Patients with an undetectable viral load increased from 31% at baseline to 55% at the first follow-up and to 78% at the last follow-up (Table 4).
EVG/COBI/TDF/FTC Clinical and Laboratory Outcomes.a,b

Abbreviation: COBI; cobicistat; EVG, elvitegravir; FTC, emtricitabine; SD, standard deviation; TDF, tenofovir disoproxil fumarate.
aAverage time to first follow-up = 65 days or 2.1 months.
bAverage time to last follow-up = 223 days or 7.3 months.
Discussion
In our study in the Southeast United States in a predominant African American, treatment-experienced population, SCr changes and renal AEs associated with EVG/COBI/TDF/FTC were comparable to those observed in clinical trials (mean increase of 0.15 mg/dL). 9 Being a guideline-recommended preferred regimen in treatment-naive patients, EVG/COBI/TDF/FTC is also a standard option in treatment-experienced patients seeking simplification or a potentially less toxic regimen. Treatment-experienced patients had twice the odds of an increase in SCr >0.3 mg/dL when compared to treatment-naive patients in our study. 11 –13 Additionally, those with more advanced disease as evidenced by lower baseline CD4 count and higher viral loads were more likely to have significant increases in SCr. This may be directly related to underlying HIV-associated nephropathy or other non-HIV–related effects. One study in virologically stable treatment-experienced patients indicated low rates of discontinuation due to renal AEs when switching to EVG/COBI/TDF/FTC. 13 In a 48-week switch trial from an NNRTI-based regimen to EVG/COBI/TDF/FTC, the median SCr increased 0.124 mg/dL during the first 4 weeks with no further progression. 12 Comparatively, the median SCr change was −0.01 mg/dL in those patients who were maintained on the NNRTI-based regimen. In a simplification study, the median change in SCr was 0.07 mg/dL for patients changing from a ritonavir-boosted protease inhibitor to EVG/COBI/TDF/FTC and it was −0.01 mg/dL for those continuing the PI-based regimen. 13 The smaller increase in SCr observed in this study was thought to be attributed to the inhibitory effects of ritonavir on the MATE-1 in the previous regimen. 13 The peak changes in SCr following initiation of EVG/COBI/TDF/FTC therapy in our study were 0.2 mg/dL with a mean decrease in CrCl of 16.1 mL/min. This is similar to the observed decreases in GFR in clinical trials of treatment-naive patients (14 mL/min). 11,12
Patients in the present study who had an increase in SCr had an average decline in estimated CrCl of 26 mL/min, which increased to 45 and 48 mL/min in those with increases of 0.3 and 0.5 mg/L, respectively. The increase in SCr typically occurs early, within the first several weeks of therapy, however, is not thought to impact actual GFR. 11 -13 Arribas et al details treatment-experienced patients who displayed increases in SCr starting at week 4, which stabilized by week 48. 13 Calculated CrCl, using Cockcroft-Gault formula or other renal estimation equations, is often utilized for dosing recommendations of antiretroviral (ARV) drugs and other medications. Although we did not document any changes in medication dosing due to SCr elevations thought to be COBI induced, using a calculated CrCl may provide a false indication of true GFR. Because most medications are not dose adjusted until estimated CrCl falls below 50 mL/min, small modest changes in SCr secondary to COBI should not routinely impact medication dosing. However, over 20% of our patients in the present study experienced an increase in SCr of at least 0.3 mg/dL. Additionally, 24 (8.4%) patients were initiated on EVG/COBI/TDF/FTC with a baseline CrCl <70 mL/min, below the threshold recommended in the package labeling. 5 Because these patients are at risk for AKI due to other drug (eg, TDF, SMX/TMP) and nondrug-related reasons, careful monitoring of renal function should be evaluated with urine output, drug concentrations when available, and SCr trends.
Concomitant use of other potential nephrotoxic medications (18% of patients) was not statistically associated with increased risk of significant elevations in SCr in our study. In 7% (n = 21) of patients, SMX/TMP was coinitiated with EVG/COBI/TDF/FTC and was listed as a concomitant medication at any point during therapy in 18% (n = 51) of patients. Among the 21 patients started concomitantly with SMX/TMP, 1 developed AKI (detailed above). Of the 65 patients who had an increase in SCr of at least 0.3 mg/dL, 15 were on SMX/TMP concomitantly. This was the most common concomitant nephrotoxic agent followed closely by angiotensin-converting enzyme inhibitors (ACE-I; 17% of patients). Although coadministration of SMX-TMP and ACE-I was more common in those with changes in SCr ≥0.3 mg/dL, logistic regression did not indicate additional risk for renal changes. Because these medications (TMP and ACE-I) are known to cause increases in SCr, often without impacting GFR, it is important to recognize this potential when initiated simultaneously with EVG/COBI/TDF/FTC. The SCr changes observed with COBI do not represent a renal toxicity but small SCr and estimated glomerular filtration rate (eGFR) changes from baseline (<0.2 mg/dL and <15 mL/min, respectively) related to inhibition of secretion of SCr following initiation of therapy. Comparatively, tenofovir renal tubular damage characteristically includes greater changes in SCr and eGFR from baseline (>0.2 mg/dL and > 15 mL/min, respectively) that are more progressive and include hypophosphatemia, hypouricemia, normoglycemic glycosuria, and proteinuria. 7,9 -13 Careful monitoring and interpretation of SCr changes are essential (Table 3).
Patients that were overweight having a BMI between 25.1 and 29.9 kg/m2 had nearly twice the odds of developing an elevated SCr (>0.3 mg/dL) compared to those at a normal weight. Patients at this BMI range represented approximately 20% of the study population. Interestingly, patients with obesity having a BMI >30 kg/m2 were less likely to develop significant increases in SCr. The impact of BMI on drug-related renal outcomes in HIV-infected individuals has not been extensively studied. Obesity is increasingly common in the HIV-infected population, as evidenced by the 33% of patients in our study population. Individuals with obesity are known to have higher GFRs and thus increased clearance of renally eliminated medications. Estimated CrCl in this population is challenging, as weight is a factor in most conventional renal function estimation equations. Some have proposed other methods for estimating CrCl in this population, but they have not been validated in an obese, HIV-infected population. No pharmacokinetic data are available for EVG/COBI/TDF/FTC or COBI alone in patients with obesity. Future study should focus on medication-induced renal changes in specified weight categories.
The discontinuation rate of EVG/COBI/TDF/FTC in our study was low at 7%. Five patients had a resistant mutation documented or virologic failure, 5 patients discontinued due to tolerability (eg, dizziness, gastrointestinal effects, and fatigue), and 3 patients developed AKI. All 3 patients who experienced AKI while on EVG/COBI/TDF/FTC were treatment naive. One patient with baseline SCr of 1.6 mg/dL that increased to 2.7 mg/dL at the first follow-up visit was on an ACE-I during EVG/COBI/TDF/FTC therapy. The second patient had a baseline SCr of 0.8 mg/dL and reached 3.3 mg/dL at the first follow-up and was initiated on SMX/TMP concurrently with EVG/COBI/TDF/FTC. The third patient had a baseline SCr of 1.1 mg/dL and increased to 1.7 mg/dL at a subsequent follow-up visit (not first follow-up) and was on an ACE-I during EVG/COBI/TDF/FTC therapy. The discontinuation rate was comparable or lower than previously published studies with an average follow-up of 32 weeks.
The retrospective nature of our study limits assessment of timely changes in renal function and other potential factors associated with SCr changes. Our population is predominantly male and African American, so it may not reflect the entire HIV-infected population in the United States. Consistent results across our multicenter study and inclusion of both treatment-naive and treatment-experienced patients, however, do support application to similar populations. Patients were followed for approximately 32 weeks, however, we cannot comment on further potential long-term effects. All previous ARV exposures and tolerability were not documented in treatment-experienced patients and may contribute to current tolerability.
Conclusion
Our results support previous randomized, controlled trial data of modest increases of SCr in a majority of patients initiated on EVG/COBI/TDF/FTC therapy. Discontinuation rates were low, and overall AEs were limited and mild in this predominantly African American, male population in the Southeast United States. Significant elevations in SCr and reports of AKI were infrequent but increased in treatment-experienced patients. Concomitant use of other potential nephrotoxic medications, notably SMX/TMP, was not statistically associated with increased risk of significant elevations in SCr. Obese patients having a BMI >30 kg/m2 were less likely to develop significant increases in SCr. Monitoring of SCr trends remains important in patients on EVG/COBI/TDF/FTC. Careful interpretation of impact of elevated SCr on actual GFR and potential impact of concomitant nephrotoxins is important for clinicians managing HIV-infected patients. Future studies should investigate similar outcomes in EVG/COBI-based regimens containing TAF.
Footnotes
Acknowledgments
The authors acknowledge Kara White, PharmD Candidate, for her contributions to data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A portion of this research was conducted using an undergraduate research grant from the University of South Carolina.
