Abstract
Introduction:
Anemia is a common problem in HIV in sub-Saharan Africa. We describe the contribution of antiretroviral therapy (ART) regimen to the incidence of anemia and changes in hemoglobin (Hb) in HIV-infected patients in Uganda.
Methods:
This study was nested in a prevention of cryptococcal disease trial (CRYPTOPRO; ISCRTN7648152). Patients received 3 different backbones of nucleoside reverse transcriptase inhibitor in a nonrandomized manner.
Results:
Of the 852 patients (161 on zidovudine [ZDV], 628 on stavudine [d4T], and 63 on tenofovir [TDF]; all received lamuvidine), the risk of developing grade 4 anemia was higher (adjusted hazard ratio 2.7) for those receiving ZDV than those receiving d4T. Those receivingd4T had a higher average increase in Hb than those receiving ZDV (P = .024) or TDF (P = .014).
Conclusion:
In this observational study, ZDV was associated with severe anemia compared to d4T or TDF; those receiving ZDV and TDF had smaller increases in Hb after ART initiation. We encourage publication of data on cohorts using TDF from Africa.
Introduction
Anemia is a common clinical problem in HIV-infected individuals, especially in those with advanced immunosuppression and is an independent risk factor for morbidity and mortality. 1,2 It is more common in sub-Saharan Africa, which is known for its high incidence of anemia due to non-HIV-related factors such as poor nutritional status, tuberculosis, and malaria. 3 In those with advanced immunosuppression, opportunistic infections (OIs) such as pneumonia, candidiasis, and HIV, wasting syndrome may compound the problem 4 –6 and drugs that are commonly used for the treatment of OIs (eg, ganciclovir, flucytosine, amphotericin, and sulfonamides) can also cause anemia. 7
Overall, the treatment of HIV infection with combination antiretroviral therapy (ART) reduces the incidence of anemia and increases hemoglobin (Hb) levels over time. 8,9 However, zidovudine (ZDV) has also been clearly demonstrated to cause anemia, 10,11 and in cohort analysis in the United States, ZDV-containing regimens showed a higher rate of new or worsening anemia. 12,13 Consequently, World Health Organization (WHO) and other guidelines recommend avoiding ZDV in patients with anemia. 14 From the Gilead 934 study, 15 it was found that the risk of anemia is known to be greater in those starting ZDV compared to those starting on tenofovir (TDF), but the 903 study of d4T compared with TDF did not report a difference in Hb level or anemia. 16 Initially, many ART programs in sub-Saharan Africa used stavudine (d4T) as the first-line treatment, but in most countries, this has now been replaced with ZDV or TDF, which have fewer side effects overall, and the most recent WHO guidelines highlight TDF (with lamivudine [3TC]) as the best first-line nucleoside reverse transcriptase inhibitor (NRTI) option. 17 Unfortunately, there are still challenges in sub-Saharan Africa as particular drugs or combinations may not be often available despite guidelines being in place. Additionally, unlike in Western settings, where ART combinations are closely tailored to individual patients, this luxury is often not available in sub-Saharan Africa where patients are treated with a public health approach and often receive whatever regimen is available despite individual relative contraindications to a particular regimen.
Findings suggest that in sub-Saharan Africa anemia is more common in patients receiving ART than those in industrialized countries. 18 Rates of severe anemia in the first 6 to 12 months of ART vary between 3.9 and 9.6 per 100 patient years at risk (PYAR). 18 –20 In the Therapeutics Research, Education, and AIDS Training in Asia (TREAT Asia) HIV observational database, anemia was reported in 13% of patients treated with ZDV. 21 One study has shown a higher incidence of anemia was reported in up to 15% of patients after switching from d4T to ZDV 22 and therefore switched from ZDV. 23 However, other studies have shown that those receiving therapy containing ZDV had less anemia than ART naive patients 23,24 and that starting ZDV in those with severe baseline anemia can be acceptable. 25 Direct comparisons of incidences of anemia on different ART regimens in sub-Saharan Africa are rare, but findings recently published from the IeDEA database (from sub-Saharan Africa, Asia Pacific, and South America) have shown a 0.5 g/dL decrease in Hb level in the first 3 months of ZDV compared with alternatives (mainly d4T, but this was not fully explored). 20
Between 2004 and 2008, we performed a randomized placebo-controlled trial of primary prophylaxis for cryptococcal disease with fluconazole (cryptococcal disease trial [CRYPTOPRO]). 26 Participants received 3 different ART combinations, thereby providing an opportunity to compare the incidence of anemia and changes in baseline Hb between 3 different regimens of non-ZDV and ZDV.
Methods
This study was nested in a randomized double-blind placebo-controlled trial of primary prophylaxis of cryptococcal disease in HIV-infected individuals (CRYPTOPRO; ISRCTN 7648152), which ran between 2004 and 2008, and full trial details have been published previously. The trial was performed among Ugandan adults in the Masaka region, Southwest Uganda and compared fluconazole 200 mg with identical placebo thrice weekly in patients with a CD4 count <200 cells/mm3. Participants in the CRYPTOPRO who commenced the ART had the potential to be observed for 48 weeks before the end of the trial and were eligible for inclusion in the anemia study analysis. Participants were excluded from the anemia study analysis if they did not start ART, did not have a pre-ART Hb reading, or had grade 4 anemia at the time of initiating ART. A total of 7 patients started on ART with grade 4 anemia, and they were analyzed separately.
Antiretroviral therapy was provided free of charge by 3 local service providers, namely, The AIDS Support Organization (TASO), Kitovu Mobile Home Care and Orphans Programme (Kitovu MAHCOP and Masaka Ministry of Health [MOH]). These providers obtained their ART supplies through the Uganda MOH ART roll-out program and/or from donations. Participants received a backbone of 2 nucleotide/NRTIs, d4T/3TC, ZDV/3TC, or tenofovir/emtricitabine (TDF/FTC), depending on the provider and availability of supply, in combination with either nevirapine (NVP) or efavirenz (EFV; non-NRTIs [NNRTIs]). At the time, the TDF/FTC was only provided at 1 clinic, and it was the first-line NRTI backbone of choice at that site. All patients received cotrimoxazole per Ugandan MOH guidelines.
Participants were seen every 8 weeks, and complete blood count plus CD4 count was taken every 16 weeks until the CD4 was >200 cells/mm3. Hb was checked if the participant was unwell or admitted to Masaka MOH Regional Referral Hospital. The timing of Hb measurements was determined during the entry into the trial and not when patients started on ART. Patients who required a transfusion under strict Hb criteria were not always able to receive a transfusion due to resource and logistic constraints within the hospital, at the time of the study.
Anemia was graded according to the AIDS clinical trials group criteria, namely, grade 1: Hb 8.0 to <9.5 g/dL, grade 2: 7.0 to <8.0 g/dL, grade 3: 6.5 to <7.0 g/dL, and grade 4: <6.5 g/dL. Mean corpuscular volume (MCV) was classified as microcytic if <68 fL, normocytic if 80 to 98 fL, and macrocytic if >98 fL, using reference ranges for healthy adults in Eastern and Southern Africa. 23 If anemia was diagnosed at baseline or during the trial, efforts investigations were made to treat the cause of anemia. All participants were offered co-trimoxazole prophylaxis per Ugandan national guidelines.
Statistical Analysis
The statistical analysis aimed to investigate the associations within the first 48 weeks of ART between the type of ART regimen and (1) incident grade 4 anemia episodes and (2) change in Hb level after the initiation of ART (with the initial ART regimen classified as d4T based, ZDV based, or TDF based). A 4-week window for tests was allowed, so all Hb readings within 52 weeks of initiating ART were considered.
For the analysis of grade 4 anemia, we used time-to-event analysis, with the event being grade 4 anemia. The incidence rates of grade 4 anemia were summarized by initial ART regimen, and anemia incidences illustrated using a Kaplan-Meier curve. Cox proportional hazards regression models were fitted to investigate the effect of ZDV-based and TDF-based regimens relative to d4T-based regimens. Potential confounders considered in this analysis were Hb at initiation of ART, baseline CD4 count, treatment arm in the CRYPTOPRO trial (fluconazole or placebo), and NNRTI used. Patients with grade 4 anemia initiating ART and patients with no pre-ART Hb reading were excluded from this analysis. Hemoglobin levels at ART initiation and baseline CD4 count were treated as continuous variables and fractional polynomials were used to allow for nonlinearity in their effect. 24 Confounders were included in the final model if they were significant at the 15% level, using a backward elimination approach. A liberal inclusion value was used to ensure adequate adjustment for the confounders.
In order to examine the effect of initial ART regimen on change in Hb level, for each participant a change in Hb was calculated as the difference between the post-ART Hb closest to week 48 and the pre-ART Hb. The change was summarized by initial ART regimen, and further analysis was carried out by fitting general linear models, adopting an analogous strategy for analyzing incident anemia, with the addition of time between pre-ART Hb and post-ART Hb as a potential continuous confounder. All data required for the analysis were extracted from the CRYPTOPRO trial database, and all analyses carried out using Stata release 10.1.
Results
A total of 855 patients were eligible for inclusion in the anemia study; of which 852 were available for analysis (3 patients did not have initial Hb estimations). In all, 161 patients were receiving ZDV/3TC, 628 were receiving d4T/3TC, and 63 were receiving TDF/FTC. The baseline characteristics of the study population are shown by initial ART regimen in Table 1. A total of 845 patients contributed to the analysis of the anemia incidence, and 7 patients were excluded because of grade 4 anemia at ART initiation. In all, 842 patients contributed to change in Hb, and 10 (1.2%) were excluded as they did not have an Hb result at least 28 days after initiating ART (8 on d4T, 1 on ZDV, and 1 on TDF). Of the 10 patients, 4 died within 1 month of initiating ART and 6 withdrew from the trial for other reasons. Figure 1 shows the summary of the patients in the separate analyses.
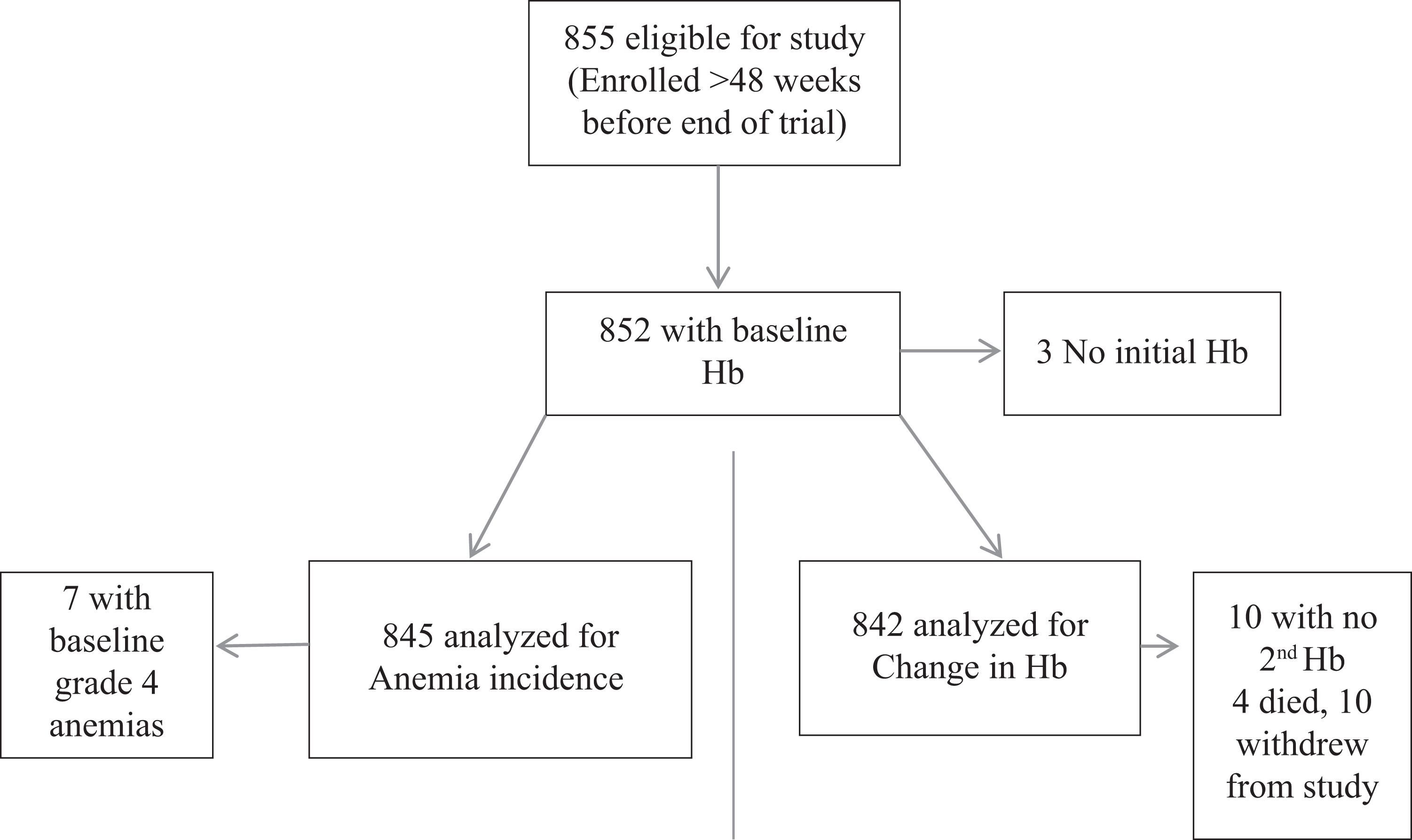
Summary of patients analyzed.
Baseline Characteristics of Study Participants.

Abbreviations: ZDV, zidovudine; d4T, stavudine; Hb, hemoglobin; IQR, interquartile range; MCV, mean corpuscular volume; NNRTI, nonnucleoside reverse transcriptase inhibitor; SD, standard deviation; WHO, World Health Organization.
The age, gender, and baseline WHO stage distributions were similar in the 3 groups, and there were roughly equal numbers in the 2 CRYPTOPRO treatment arms. However, those who received a TDF-based regimen had lower CD4 counts and lower MCV. Patients initiated on ZDV were more likely than those in the other 2 groups to be given EFV as their NNRTI. The initial mean Hb was similar in the 3 groups, although patients with either grade 3 (n = 3) or grade 4 (n = 7) anemia at ART initiation were started on a d4T-based regimen.
Grade 4 Anemia Post-ART
Figure 2 shows a Kaplan-Meier plot, giving the time to the first grade 4 anemia event post-ART among the 845 patients who did not have grade 4 anemia at the initiation of ART. Overall, 51 of the 845 patients at risk developed grade 4 anemia, with an incidence rate of 8.7 per 100 PYAR (95% confidence limits 6.6-11.5 per 100 PYAR). Table 2 shows the incidence rates and the adjusted hazard ratios (aHRs) of grade 4 anemia in patients receiving ART regimen. The rates were highest for those on an ZDV-based regimen (17.1 per 100 PYAR) followed by those on a TDF-based regimen (10.6 per 100 PYAR) and were lowest for those on a d4T-based regimen (6.5 per 100 PYAR). Confounders included in the regression model were Hb at ART initiation (as a fractional polynomial with power −2, ie, inverse quadratic), baseline CD4 count (as a linear effect), and gender (with females being at a lower risk after adjusting for other factors).

Time to first post antiretroviral therapy (post-ART) grade 4 anemia.
Comparison of Incidence of Grade 4 Anemia between Initial ART Regimens.a

Abbreviations: ART, antiretroviral therapy; CI, confidence interval; CRYPTOPRO, cryptococcal disease trial; d4T, stavudine; Hb, hemoglobin; NNRTI, nonnucleoside reverse transcriptase inhibitor; ZDV, zidovudine.
aAdjusted for baseline CD4 count, baseline Hb, gender, CRYPTOPRO treatment arm, and NNRTI.
Patients starting on ZDV were at a 2.7 times greater risk (95% confidence interval [CI] 1.5-5.0; P < .001) of grade 4 anemia than those starting on d4T. The point estimate of the TDF effect showed an increased risk of grade 4 anemia compared to participants on d4T, but due to the small number of patients on TDF, this did not reach statistical significance (aHR 1.6, 95% CI 0.6-4.1; P = .35).
Developing a new grade 4 anemia was a risk factor for death, with 19 (37.2%) of the 51 patients, who developed grade 4 anemia on ART, dying compared to 43 (5.4%) of the 794 patients who did not (P < .001). In 8 patients (5 on d4T, 2 on ZDV, and 1 on TDF), anemia was listed as a cause of death; there was no evidence that the risk of death differed between the 3 regimens although the numbers are very small (Fisher exact P = .74). Overall, 30 (58.8%) of the 51 patients with grade 4 anemia were hospitalized within a 2-week window either side of the anemia event. There was no difference in the probability of being hospitalized between the 3 ART regimens (18 were on d4T, 9 on ZDV, and 3 on TDF; Fisher exact P = .65). Only 18 (35.3%) of the patients who developed grade 4 anemia received a blood transfusion (11 on d4T and 7 on ZDV; Fisher exact P = .29).
HB Level Rises After ART Initiation
The change in Hb level after 48 weeks on ART differed significantly between the 3 groups (unadjusted changes were d4T +1.40 g/dL, ZDV +1.0 g/dL, and TDF + 0.84; P = .012 from a 1-way analysis of variance; Table 3). We used the Hb reading nearest to week 48 post-ART (interquartile range [IQR] = 206-327 days); therefore, an adjusted analysis was performed for the day on which the reading was made with an adjustment for day of Hb measured, using a second-order fractional polynomial with terms in day and day × log(day), 27 as well as baseline Hb and gender. The predicted changes in Hb at week 48 obtained from this model are shown in Table 3, confirming that while all 3 regimens result in small increases in Hb level, the increase observed in the d4T group was significantly greater than that in the other 2 groups.
Predicted Change in Hb over 48 Weeks of Treatment.a

Abbreviations: ZDV, zidovudine; CI, confidence interval; d4T, stavudine; Hb, hemoglobin; TDF, tenofovir.
aAdjusted for days to Hb reading, baseline Hb, and gender.
bDifference between d4T and ZDV statistically significant (P = .024).
cDifference between d4T and TDF statistically significant (P = .014).
Observation on Those Starting ART with Baseline Anemia
Of the 7 patients with grade 4 anemia who started on ART, 2 (28.5%) patients died; of the 3 patients with grade 3 anemia, 1 (33.3%) subsequently died; of the 23 with grade 2 anemia, 6 (26.1%) subsequently died; of the 107 with grade 1 anemia, 16 (15.0%) subsequently died, and of the 740 without anemia, 54 (7.3%) subsequently died. This indicates that baseline anemia incidence was associated with increased mortality (χ2 = 15.57 on 4 df; P = .004).
Of those with grade 1 and 2 anemia at baseline, 10% starting on d4T developed subsequent severe anemia, 13% starting on ZDV, and none starting on TDF. Table 4 provides details on Hb changes. All 10 patients with baseline grade 3 or 4 anemia were initiated on d4T. The median post-ART Hb level in patients who had grade 4 anemia at baseline was 11.0 g/dL (IQR = 9.2-12.8) compared to a median post-ART of 12.9 g/dL (IQR = 11.8-13.8) in patients who did not have grade 4 anemia when initiating ART.
Summary of Patients with Grades 1 and 2 Anemia at Baseline.a

Abbreviations: ART, antiretroviral therapy; ZDV, zidovudine; d4T, stavudine; IQR, interquartile range; Hb, hemoglobin; TDF, tenofovir.
aDue to the small numbers involved, none of the differences between the 3 treatment regimens approached statistical significance.
Discussion
In the sub-Saharan African context, it is difficult to make comparisons between ART regimens as the regimens are usually decided at a country level, with limited individual patient variation. It is clear from the previous findings that many people eligible for ART will have pre-existing anemia, and in keeping with ART provision in a public health setting, the DART trial suggested targeted laboratory monitoring in a resource-poor setting. 28 The CRYPTOPRO cohort offers a unique insight into patients starting ART in Uganda, as the participants were recruited from 3 different HIV service providers who independently selected the ART regimens for the participants, and so we have been able to present some comparative findings on the effect of different regimens on Hb level. At the time of this study in Uganda, the supplies for ART were limited; only 1 site was able to offer TDF/FTC (this was the first-line NRTI backbone at this site), and d4T was frequently the only NRTI backbone available at the other sites. The major limitation of this study is the non-randomization of NRTI backbones; however, the method of allocation meant that the risk of channeling bias in the study is somewhat reduced, as clinicians started patients on whatever drug was available to them at each site. As the ART regimens were not randomly allocated, this led to baseline inequalities between treatment groups. Of these, the most relevant is the lower median CD4 cells in the TDF arm, as there is an association between nadir CD4 and the risk of anemia 3 ; therefore, we adjusted for this in the logistic regression models. Another limitation is the relatively small number of patients on TDF giving limited statistical power to detect differences between TDF and other regimens. All participants had CD4 counts <200 cells/µL, so the cohort is more immunosuppressed than those currently starting ART with the new WHO recommendations of starting at a CD4 count of 500 cells/µL. Nevertheless, as many ART-eligible patients in Africa still begin ART at low CD4 counts, our study provided valuable insights. There may have been some bias related to ART selection as patients initiated on ZDV were more likely to be given EFV as their NNRTI than the other 2 groups; however, we believe this was simply due to NNRTI availability, but it is difficult to interpret. Despite these limitations, we feel that this study offers some important insights about the development of anemia between patients within the same cohort receiving regimens with ZDV, d4T, and TDF backbones.
This study showed that clinically important grade 4 anemia occurred with all ART regimens at an overall rate of 8.7 per 100 person years of exposure (PYO) for the first 48 weeks of starting ART. As seen in the large US cohorts 4,5,6 and as well as in 1 study in Uganda, 18 this was associated with increased mortality. The incidence rate of grade 4 anemia in our study was comparable with previously recorded rates of 4.0 to 9.6 per 100 PYO in West Africa. 19,29 In those taking ZDV, there was an increased risk (aHR 2.7) of incident grade 4 anemia compared to d4T. However, in our study this did not lead to excess mortality.
Overall, ART increased the mean Hb level in all 3 arms, which is in keeping with the other studies of post-ART Hb changes. 25,29 However, in the adjusted analysis, the increase was greater for those taking d4T than for those taking both ZDV and TDF, which is different from the previous reports in sub-Saharan Africa. Our results differ somewhat from those of a similar study in urban Uganda, which found no significant difference in Hb increase in ZDV compared with d4T-containing regimens in urban Uganda, and the reason for the differences is not entirely clear. 25 The baseline data of the 2 cohorts were similar, although the CRYPTOPRO cohort was slightly more immunosuppressed, as all participants had a CD4 count <200, compared to 85% of those in the urban cohort. This may explain a higher incidence of anemia (6% over 48 weeks) in our cohort than in the urban cohort (4% at 6 months), which may have made a difference between regimens which was easier to detect.
We have not been able to find other reports of sub-Saharan Africa on the effect of TDF on Hb level compared with other ART regimens. Tenofovir is one of the first-line regimens recommended in the 2010 WHO ART guidelines, which along with ZDV is now being used in preference to d4T in sub-Saharan Africa. These findings suggest that Hb level might also need to be monitored in those receiving TDF. While there is no statistical difference in the number of grade 4 anemia events in this group, the Hb rise was the lowest of all 3 regimens. It will be important to study Hb changes and watch for anemia in larger cohorts of patients receiving TDF in sub-Saharan Africa over the next few years.
Conclusion
In this study population, we observed higher levels of anemia and a higher incidence of severe anemia among patients receiving ZDV than other NRTIs. As suggested by the DART trial, 28 we would encourage close clinical surveillance in all patients starting ART in order to avoid the potentially fatal consequences of undiagnosed iatrogenic (and other) anemia. We would encourage those with larger cohorts of patients now receiving TDF as the first line in sub-Saharan Africa to describe their experiences, as more information on development of anemia in this population is important.
Footnotes
Acknowledgments
We wish to acknowledge all the CRYPTOPRO trial participants. We also acknowledge all of the CRYPTOPRO trial staff.
Authors’ Note
RPR contributed to study design, data collection, data analysis, and manuscript preparation. DK contributed to data collection, data analysis, and manuscript preparation. JL contributed to study design, data analysis, and manuscript preparation. KW contributed to data analysis and manuscript preparation. HG contributed to study design and manuscript preparation. AK contributed to study design and manuscript preparation. DL contributed to study design, data analysis, and manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CRYPTOPRO trial was funded by the Medical Research Council, United Kingdom.
