Abstract
Background:
Once-daily (QD), combination antiretroviral therapy (ART) can impact the willingness and ability of patients to take medications as directed. The impact of antiretroviral (ARV) drug adherence influenced by single-tablet (STR) versus multi-tablet regimens (MTR) among patients enrolled in the AIDS Drug Assistance Program (ADAP) in a rural environment has not yet been assessed.
Material and Methods:
A retrospective chart review evaluated adherence and outcomes in adult HIV-infected patients enrolled in the ADAP at 2 ambulatory clinics in the Southeast, taking either a QD STR (efavirenz [EFV]/emtricitabine/tenofovir [TDF]) or a QD protease inhibitor (PI)-based, MTR (atazanavir [ATV], ritonavir [RTV], and emtricitabine/TDF) by evaluating pharmacy refill records, patient self-reported adherence, and virologic response.
Results:
A total of 389 patient records were analyzed (STR, n = 165 versus MTR, n = 224). There were more males, a higher percentage of treatment-naive patients, and more patients with a baseline CD4 count of >200 cells/mm3 in the MTR group. Based on refill records, more patients on MTR were >90% adherent (61.6% versus 51.5%, P = .047). In a multivariable analysis, being treatment experienced was a negative predictor (odds ratio [OR] = 0.48, 0.29-0.78) for adherence. Regimen choice was not associated with adherence. More patients taking MTR were virologically suppressed at the end of the observation period. Regardless of the regimen, being >90% adherent was a significant predictor of virologic suppression (OR = 3.51, 1.98-6.23).
Conclusion:
Treatment-experienced patients enrolled in ADAP are less likely to be adherent. A QD PI-based MTR may result in comparable adherence to an STR in a rural HIV-infected population.
Introduction
Potent combination antiretroviral therapy (ART) is the cornerstone of current HIV treatment. Antiretroviral therapy (ART) adherence is one of the strongest predictors of progression to AIDS and death among people living with HIV/AIDS. Nonadherence to ART compromises medication effectiveness and decreases the likelihood of achieving viral suppression. Quality of life of HIV-infected patients is significantly improved with high rates of ART adherence. Erlen et al 1 demonstrated that patients believed their lives were better after taking ARV drugs and that ART offered them a second chance in life. Paterson and colleagues 2 showed that an adherence rate of 95% to a protease inhibitor (PI)-based ART regimen is necessary to achieve viral suppression. Others have demonstrated a nonnucleoside reverse transcriptase inhibitor (NNRTI)-based ART regimen may require a slightly lower adherence rate of 70% to 90%.2,3 Similarly, positive clinical outcomes correlated well with a 90% or higher adherence rate to the integrase strand transfer inhibitors. 4
Single-tablet regimens (STRs) have proven effective in suppressing viral replication and are the guideline-preferred regimens for treatment-naive patients. 5 Single-tablet regimens may further increase adherence rates compared with once-daily (QD) multi-tablet regimens (MTRs). In a multicenter, randomized trial investigating the therapeutic switch from a PI- or NNRTI-based MTR to a STR (efavirenz [EFV]/emtricitabine [FTC]/tenofovir [TDF]; Atripla®) in virologically suppressed HIV-infected patients, self-reported adherence was 96% or higher in both groups, with no statistically significant differences between the 2 regimens. 6 A recent study in a Veteran’s Affairs (VA) population evaluated the impact of ART as STR and MTR on clinical outcomes. 7 Patients taking STRs had fewer hospitalizations, a 20% greater odds of achieving an undetectable viral load, and were 2 times more likely to be adherent compared to those taking MTRs.
The AIDS Drug Assistance Program (ADAP), a federally funded program that provides ART to low-income, uninsured, or underinsured Americans living with HIV/AIDS, has dispensed more than 450,000 prescriptions (60% ARVs) to more than 135,000 enrollees. Program operations, including dispensing mechanisms (eg, centralized pharmacy versus mail order) and formulary decisions, are handled at the individual state level. Proven barriers to adherence that threaten medication access are common in the ADAP population. 8 Godwin et al 9 has shown a medication possession ratio (MPR) of 84% among ADAP users in a single clinic in Alabama with younger age, nonwhite race, and a lower baseline CD4 count all associated with lower adherence rates.
In the Deep South, HIV-related health outcomes are disproportionate compared to national statistics. 10 This may be due to the higher prevalence of patients in rural areas and challenges with access to care. Additional common characteristics among these states include high rates of poverty, lower educational levels, and high rates of comorbid disease states such as heart disease and diabetes.11,12 In light of the unique adherence barriers in the Deep South, specifically among ADAP enrollees, and mounting data supporting improved adherence rates and clinical outcomes associated with STRs compared with MTRs, the impact of these regimens on adherence in HIV-infected patients warrants evaluation. This study evaluated comparative adherence and virologic outcomes in adult HIV-infected patients enrolled in ADAP at two clinics in labama or South Carolina taking either a STR or MTR, QD ART regimens.
Methods
We performed a multicenter, retrospective, observational cohort study at 2 independent, HIV/AIDS clinics in the Southeast United States. The Immunology Clinic (Study Site A) is a state-funded, Ryan White clinic associated with the University of South Carolina, Department of Internal Medicine in Columbia, South Carolina. It serves 2,000 HIV/AIDS-infected patients, the majority of whom have an income below the federal poverty line and no medical insurance. There are approximately 961 patients receiving ART through ADAP at the Columbia clinic. The Medical AIDS Outreach of Alabama Clinic (Study Site B) is a state and federally funded, Ryan White clinic in Montgomery, Alabama. The clinic medically serves approximately 1,300 patients who are almost evenly distributed between those with and without medical insurance. There are approximately 450 patients receiving ART on ADAP at the Montgomery clinic. We consecutively enrolled patients aged ≥18 years with a documented visit to 1 of the 2 clinics between January 2007 and December 2010, who were prescribed 1 of the 2 QD ARV regimens, that is, a PI-based MTR (FTC) consisting of atazanavir (ATV), boosted with ritonavir (r), and FTC/TDF (Reyataz®/Norvir®/Truvada®) or an NNRTI-based STR, EFV/FTC/TDF. These 2 regimens were selected because EFV/FTC/TDF was the only STR available during the study period, and AZV/r /TDF/FTC consists of the same NRTI components and had the lowest pill burden of the QD boosted-PI regimens at that time. Both regimens were appropriate first-line ART for HIV-1-infected adults according to 2007 guidelines prepared by the Department of Health and Human Services. 13 Patients were included if they filled their ART through the South Carolina or Alabama ADAP at least twice during the study period. Selection of an ADAP population allowed for directly measured adherence by drug dispensing records in the respective clinics. Patients were excluded from the study if they were prescribed ART less frequently than once a day.
The following data were collected on each study participant for up to 24 consecutive months: patient adherence determined by computerized pharmacy refill records and patient self-reporting of missed doses at each medical visit as documented by the nursing intake; patients’ age, gender, and race; number of concomitant medications and ARV regimen; and treatment status. Treatment status was divided into treatment experienced or naive. Treatment experienced was defined as previous history of taking ART including one of the study ARV regimens any time before the day of study inclusion. Treatment naive was defined as no prior history of ART exposure and initiating ART with one of the study regimens during the study period. Viral load (HIV RNA) and CD4 count were recorded for clinic visits throughout the study inclusion period.
Adherence according to pharmacy refill records was recorded as the average number of days between monthly refills. Adherence according to self-reporting was estimated by assuming that each day of ART missed was an additional day between refills of a 30-day supply. The adherence rate for each study participant was calculated using MPR by calculating the percentage of days missed out of 30 days and subtracting from 100%.
An adherence rate greater than 90% was used to categorize a patient as sufficiently adherent to ART in accordance with the previous studies.2 –4 An HIV RNA of ≦75 or ≦50 copies/mL, depending on the assay available at the Immunology Clinic and Medical AIDS Outreach clinic, respectively, during the study period, was defined as undetectable. The number of patients in each treatment group with undetectable HIV RNA at the end of study was calculated. In addition, the percentage of time of undetectable HIV RNA for each participant was calculated and compared between treatment groups. The mean change in CD4 count was compared between adherent and nonadherent patients and between treatment groups.
Patients’ demographics and basic clinical outcomes were compared between the 2 treatment groups. Categorical variables were compared with chi-square or Fisher exact tests, as appropriate. Continuous variables were not normally distributed and compared with Mann-Whitney U tests. A multivariable logistic regression model was used to evaluate factors associated with adherence. A multivariable regression was also performed to evaluate the impact of adherence on patients’ virologic suppression defined as undetectable HIV RNA, as well as to predict the probability of achieving virologic suppression. The statistical significance level was defined as a P value of <.05. All analyses were performed using SAS Software version 9.2 (Statistical Analysis Systems, Cary, North Carolina).
Results
Baseline Characteristics
A total of 389 patients met the inclusion criteria for this analysis, that is, 224 (57.6%) in the MTR group and 165 (42.4%) in the STR group (Table 1). Patients had a median age of 42 years, were predominantly African American, and were >60% were male. Patients were taking an average of 2.5 concomitant medications, and 46% were taking up to 4 concomitant medications. At baseline, ≥75% of patients had an undetectable viral load. The median CD4 count was 360 and 296 cells/mm3, in the MTR and STR groups, respectively (P < .0087), and the percentage of patients with a baseline CD4 count < 200 cells/mm3was 21% and 31%, respectively. The median duration of follow-up was 22 and 14 months in the MTR and STR groups, respectively (P < .001). Patients were predominately treatment experienced (69.2%) prior to the study. More of these patients were prescribed the STR (78% and 63%, P = .0009), whereas more treatment-naive patients were prescribed the MTR (38% and 22%, respectively, P = .0009).
Baseline Characteristics of the Study Participants by Regimen Status.a

Abbreviations: IQR, interquartile range; VL, viral load.
aData are percentage of participants, unless indicated otherwise.
bUndetectable defined as <75 or <50 copies/mL for the respective clinics.
Adherence Comparisons and Clinical Comparisons between Regimens
Median adherence rates based on clinic records were similar between groups, that is, 91% and 93% in the STR and MTR groups, respectively (P < .14; Table 2). Median self-reported adherence rates were higher than recorded by refill records for both groups (100% and 99%, respectively), and a much higher proportion in the MTR compared to the STR group (93% and 85%, P = .02). The median change in CD4 count from baseline was higher for the MTR group (80 and 53, respectively) but not statistically significant. Numerically, more treatment-experienced patients taking the STR were virologically undetectable at the end of the study (75% and 62%, respectively). Virologic suppression was observed in more treatment-naive patients taking the MTR (39% and 25%, respectively).
Adherence and Other Clinical Differences by Regimen Status.

Abbreviations: IQR: interquartile range; VL, viral load.
a<90% nonadherence.
b≥90% adherence.
Based on clinic refill records, more treatment-naive patients were >90% adherent with the MTR (44% and 24%, P = .003; Table 3). Alternatively, more treatment-experienced patients were adherent with the STR (77% and 57%, respectively). Patients taking the MTR with an adherence rate <90% had a greater percentage of time undetectable and more patients were virologically suppressed. In patients with >90% adherence, the proportion of patients achieving a CD4 count of >200 cells/mm3 was comparable between regimens. However, in those who were <90% adherent, more patients taking the MTR had a CD4 count of >200 cell/mm3 (78% and 54%, respectively). The median number of concomitant medications was similar between the STR and MTR groups, regardless of the adherence status.
Clinical Characteristics by Adherence Status.
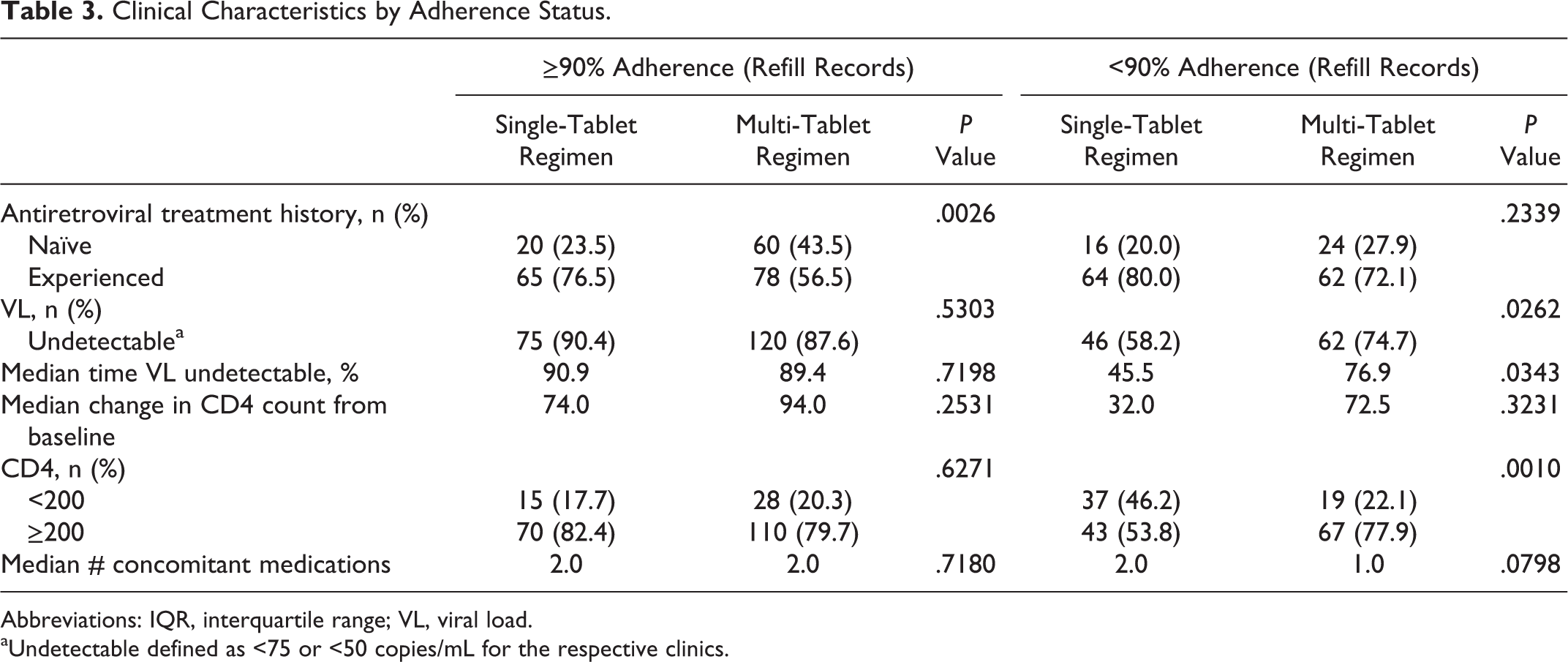
Abbreviations: IQR, interquartile range; VL, viral load.
aUndetectable defined as <75 or <50 copies/mL for the respective clinics.
A multivariable analysis was performed to determine predictors for adherence (Table 4). The greatest predictor of adherence was treatment status. Treatment-experienced patients were 52% less likely than treatment-naive patients to be adherent. The regimen, number of concomitant medications, demographic characteristics, baseline CD4 count, or duration of enrollment was not predictive of adherence in this study.
Predictors for Adherence.a

aSignificance level is set at P < .05.
Predictors for virologic suppression including adherence were evaluated (Table 5). Patients adherent to ART were 3-fold more likely than nonadherent patients to be virologically suppressed (odds ratio [OR], 3.51; 95% confidence interval [CI], 1.98-6.23). Virologic suppression was also related to longer enrollment periods during the study (OR, 1.06; 95% CI, 1.02-1.10). Treatment status, baseline CD4 count, or demographic characteristics did not impact virologic suppression. Interestingly, 1 more concomitant medication resulted in a 14% less possibility of virologic suppression (OR, 0.86; 95% CI, 0.76-0.97).
Predictors for Virologic Suppression.a,b

aSignificance level is set at P < .05.
bVirologic suppression defined as HIV RNA <75 or <50 copies/mL for the respective clinics.
Discussion
It is well established that adherence to ART directly impacts morbidity and mortality.14,15 Nonadherence to ART may further result in the selection of drug-resistant HIV strains.16 –18 Interdisciplinary teams including pharmacists contribute to higher adherence rates.19 –21 Both clinics in this study utilize pharmacists for ART adherence counseling. Direct methods of measuring adherence such as direct observation and therapeutic drug monitoring yield a more reliable assessment of adherence; however, these approaches are often cost-prohibitive. 18 Indirect methods of measuring adherence such as electronic monitoring devices, prescription refill histories, and self-reporting are commonly used adherence measurements.22 –24 Previous studies have reported a moderate to high concordance of self-report and pharmacy refill records.24 –30 Our study revealed a higher mean self-reported adherence rate (95% and 97%) than clinic refill records (84% and 87%), which was consistent between the treatment groups. Of the variables that impact virologic suppression in this study, adherence was found to be the primary factor influencing this clinical outcome (3-fold higher rates). Godwin et al 9 also observed low rates of ADAP utilization, particularly in black, younger patients with a history of alcohol abuse. In their study, the mean MPR was 77%, and only 32% of patients were >90% adherent.
The ARV regimen selected may also affect treatment adherence. Studies evaluating the simplification of regimens by reducing pill burden or changing dosing administration to QD have demonstrated greater adherence rates and comparable or improved virologic outcomes.31 –35 Simplifying regimens from multiple daily doses to QD can positively affect the quality of life.32,36 Reducing pill burden to a QD regimen has also achieved greater optimal (>95% adherence) rates and higher patient satisfaction.33 –38 Single-tablet regimens have demonstrated high adherence rates and noninferiority in maintaining virologic suppression35,38 These outcomes, associated with the simplification of ART, have been largely studied with EFV-based regimens, but PI-based regimens have also been evaluated. Several ATV studies in treatment-naive patients reported very high adherence rates to TDF/emtricitabine/ATV/RTV and positive satisfaction with the regimen.39,40 In a meta-analysis by Parienti et al, adherence was greater for patients who were initiating therapy compared with those who were switching to QD regimens. 31 In our study, both QD regimens had mean adherence rates >84%; Compared to STR, MTR overall had higher rates of adherence (>90%) and specifically in treatment-naive patients.
Studies of virologic failure among ART regimens indicate that different classes of ART appear to have different adherence cutoff rates resulting in regimen failure.41
–43 In a prospective, cohort study evaluating low, moderate, and high adherence rates with ART, moderate levels of adherence to NNRTI-based regimens maintained virologic suppression, and low adherence resulted in virologic failure
41
Comparatively, moderate adherence to unboosted and boosted PI regimens resulted in virologic failure, while an adherence rate of >95% correlated with a 70% lower risk of virologic failure in patients taking PI therapy.
2
Intermediate rates of adherence (70%-90%) with NNRTI appear to be more forgiving.3,42 This is considered to be due to their long half-lives and higher trough concentrations above the level of resistance (inhibitory quotient).
41
Health outcomes in HIV-infected patients in the Deep South are particularly affected by unique barriers related to the rural living circumstances. 44 This population characteristically is more impoverished, has lower levels of education, transportation limitations, poor access to health care resources, preventive health services, and health information and is subject to greater social stigma.10,11,45,46 These aspects present additional challenges to achievement of optimal health outcomes with ART. In our study of patients from 2 clinics in the southern states, the MTR resulted in greater durability of response as compared with the STR. Refill records indicated greater adherence to the MTR (mean adherence rate of 88%). In this more treatment-experienced population, more patients taking the MTR were virologically suppressed regardless of adherence rates with greater durability of response. Greater time of virologic suppression observed with this regimen may have been due to the greater proportion of patients taking the MTR compared with those taking the STR. Of those who were undetectable on ART at baseline, more patients on the MTR remained virologically suppressed during this study. Antiretroviral medications obtained through the ADAP are provided at no cost to the patients, so financial burden would not be a concern with the patients studied. The adherence and clinical outcomes observed in our study favored the MTR rather than the STR as previously reported by Rao and colleagues in a VA population. 7 This may, in part, be attributed to this unique rural population. 47
Not surprisingly, the number of concomitant medications negatively impacted the achievement of virologic suppression. The challenges of multiple medical conditions and treatment adherence can impact clinical outcomes in HIV-infected patients. The impact of concomitant medications was not unexpected in our population from the Deep South among states with the highest incidences of diabetes and cardiovascular disease. 12 Our study did not find a significant impact of STR compared to MTR as a predictor of virologic outcomes. Additional factors that could account for this are patients living in rural areas with a low educational level, poor health literacy, and limited access to care.10,11
Several limitations in the present study warrant discussion. First, this is an observational study conducted in 2 HIV/AIDS clinics in the southeast. It is uncertain whether the results can be generalized to other clinical settings. Second, data were obtained retrospectively from medical charts for self-reporting and refill records. Self-reported adherence records are convenient, but they are subject to recall bias and response bias. Third, there were a greater number of patients on the PI-based MTR. Finally, other factors should be further evaluated as barriers to adherence. These include medication side effects; length of time since diagnosis to evaluate pill fatigue; and barriers unique to a rural population such as transportation, poverty, health literacy, low educational level, and retention in care.
Conclusion
Adherence to ART is critical to the durability of virologic and immunologic response. Treatment-experienced patients receiving ADAP ART are less likely to exhibit >90% adherence, regardless of the regimen. In the Southeastern United States, QD, PI-based MTR may result in comparable adherence to NNRTI-based STR. Evaluation of adherence barriers among ADAP users should continue to be a focus of interdisciplinary teams and state agencies.
Footnotes
Acknowledgment
The authors acknowledge Matthew Caudle, PharmD candidate, for his contributions.
Authors’ Note
Data were presented at the American College of Clinical Pharmacy Annual Meeting, Hollywood, FL, October 21-24, 2012. Abstract #387. At the time of the data collection, Sarah Tennant was a pharmacy student at the South Carolina College of Pharmacy, University of South Carolina.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. Kelly Hester: Speaker’s bureau for ViiV Healthcare. P. Brandon Bookstaver: Advisory board for Gilead.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
