Abstract
Objectives:
We examined the association between adherence to drug-refill visits and virologic outcomes in a cohort of HIV-infected adults on combination antiretroviral therapy (cART) in North Central Nigeria.
Methods:
Retrospectively, 588 HIV-infected, cART-naive adults (aged ≥15 years), initiated on first-line ART between 2009 and 2010 at the Jos University Teaching Hospital, were evaluated. Association between adherence to drug-refill visits, virologic (viral load > 1000 copies/mL), and immunologic failure was assessed using multivariable logistic regression.
Results:
After a median of 12 months on cART, 16% (n = 94) and 10% (n = 59) of patients had virologic and immunologic failures, respectively. In the final multivariable model, suboptimal adherence to drug-refill visits was a significant predictor of both virologic (adjusted odds ratio [AOR] 1.6; 95% confidence interval [CI]:1.2–2.3) and immunologic (AOR 1.92; 95% CI:1.06–3.49) failures.
Conclusion:
Adherence to drug refill is a useful predictor of successful virologic control and could be utilized for routine monitoring of adherence to cART in our clinical setting.
Introduction
Sub-Saharan Africa (SSA) has witnessed a sustained reduction in AIDS-related mortality as a result of increased access to life-saving antiretroviral therapy (ART). The number of people dying of AIDS-related causes was cut by 32% between 2005 and 2011. 1 Antiretroviral therapy also significantly reduced the risk of HIV transmission 2 and the spread of tuberculosis. 3 Nigeria ranks second in the global burden of HIV (next to South Africa), having an estimated 3.5 million people living with HIV and AIDS. 4 Expansion of HIV treatment services in Nigeria started in 2004 with the support of the US President’s Emergency Plan for AIDS Relief (PEPFAR) program. This resulted in more than 3-fold increases in the number of people accessing antiretroviral (ARV) medications from 108 572 to 395 569, between 2006 and 2011. 5
Despite historic gains in the expansion of treatment services, adherence to combination ART (cART) remains a significant challenge to sustaining the benefits of ART. Suboptimal adherence results in virological failure, 6 -9 development of HIV drug resistance, 10 -12 and death. 13 One study showed that, for every 10% decrease in adherence, there is a 16% increase in HIV-related mortality. 7 In another study, there was 28% reduction in the risk of progression to AIDS with each 10% difference in mean adherence. 8 Early identification of patients at risk of poor treatment outcomes through accurate and continuous assessment of adherence is important for effective and efficient treatment planning. This will also inform the provision of targeted adherence support.
Pharmacy refill data could serve as an adherence measure by providing the dates on which ARV medications were dispensed. It is a less intrusive method of measuring adherence than most other measures. A number of studies on the treatment of HIV and other diseases have used pharmacy refill records to assess adherence. 14,15 In a study, patients with regular pharmacy refills for >4 months were more likely than less adherent patients to achieve virologic suppression and immunologic improvement. 16 In another study, a cumulative adherence of <95% to drug-refill visits was associated with virologic and immunologic failure and was comparable to CD4 counts for detecting current virologic failure in the cohort of patients on cART. 17
Although Nigeria contributes significantly to the global HIV burden, 4 very little is known about the virologic outcome of pharmacy refill adherence in public care facilities in Nigeria due to poor availability of viral load (VL) testing. Lack of emphasis on VL testing in ART programs in SSA is related to financial and technical constraints and the public health model adopted for the management of HIV in Africa. 18,19 The current study is aimed at evaluating the impact of adherence to drug-refill schedules on immunological and virological outcomes in a cohort of adult Nigerian patients on cART.
Methods
Setting and Study Population
Jos University Teaching hospital (JUTH) is located in North Central Nigeria. JUTH HIV clinic is an outpatient clinic and is one of the largest providers of treatment, care, and support to people living with HIV in Nigeria. At the time of study, over 13 000 adults were enrolled in care and approximately 9300 patients were on treatment. Since 2004, the JUTH HIV clinic has received additional funding and support from the US PEPFAR program through the Harvard School of Public Health.
Eligibility Criteria
HIV-1-infected (confirmed positive by Western blot) treatment-naive adults (15 years and older) started on first-line ART between January 2009 and December 2010 and followed up through December 2011 were included in a retrospective cohort analysis. Antiretroviral therapy eligibility criteria were based on the Nigerian National guidelines for HIV/AIDS treatment and care in adolescents and adults, 20 which at the time of the study recommended ART for those with a CD4 count <200 cells/mm3 regardless of the symptom or with CD4 count <350 cells/mm3 with HIV-related symptoms.
Standard of Care
Antiretroviral therapy-eligible patients were initiated on nonnucleoside reverse transcriptase inhibitor (NNRTI)-based cART defined as an NNRTI plus 2 nucleoside reverse transcriptase inhibitors (NRTIs), a criterion chosen based on the Nigerian National Adult ART Guidelines and its significance to resource-limited settings (RLSs). 21,22 At ART initiation, patient’s received information on ART and the importance of adherence. Patients were dispensed ART monthly and at scheduled doctor visits. They were also given projected monthly pharmacy refill dates and the date for the next appointment with the doctor. At each refill visit, a pharmacist dispensed ARV pills and entered the dispense data into an electronic pharmacy record (FileMaker Pro, v10, Santa Clara, California), which had been developed as part of the Harvard PEPFAR/APIN Plus program. All the patients had HIV-1 VL (Amplicor HIV-1 Monitor v1.5, Roche Diagnostics, GmbH, Mannheim, Germany, lower detection limit 400 copies/mL) and CD4 count (CyFlow, Partec, Munster, Germany) pre-ART initiation, at 12 weeks, and subsequently every 24 weeks per clinical care protocols.
Data Collection and Ethical Consideration
Data retrieved for analysis from the Harvard PEPFAR/APIN Plus program database included baseline sociodemographic data (age, gender, occupation, and marital status), clinical characteristics pre-ART initiation, drug pickup history (date, number of pills dispensed, and regimen), CD4 count, and VL results pre-ART, at 24, and 48 weeks. The clinical protocols and use of data for studies have been approved by JUTH ethics committee, and all enrolled patients provided informed consent for the use of their data for research.
Outcome Definition
Virologic failure was defined as plasma VL above 1000 copies/mL based on 2 consecutive VL measurements after 6 to 12 months of cART initiation. Lack of responses within the first year was chosen as an outcome because of the clinical importance of initial response to cART on subsequent disease outcomes. 23 Immunological failure was defined as having a CD4 count of equal to or less than CD4 count pre-ART, after 6 months on ART, or a CD4 count of 100 cells/mm3 past 6 months on ART. 24 Drug refill adherence was calculated automatically by the computerized pharmacy appointment system using the total number of days that the patient was behind for the drug-refill visits, based on the following formula:
The number of days behind for drug-refill visit = (last dispense date − date of the prescheduled appointment indicated on pharmacy database).
The results were then added up for all refill visits to obtain the total number of days behind the scheduled visit per patient. Percentage of cumulative adherence was estimated by the following formula:
The total number of days behind schedule/total number of days the patient was assumed to be exposed to ART given the dispensed number of pills × 100.
Optimal and suboptimal adherence was defined as a cumulative adherence to drug-refill visits of ≥95% and <95%, respectively.
Statistical Analysis
Continuous variables were described by means and standard deviation (SD) or median and interquartile range (IQR) as appropriate based on the distribution, while categorical variables were described by frequencies and proportions. Bivariate analyses to evaluate factors associated with virologic and immunologic failure were performed using Pearson chi-square test. Thereafter, variables with a
Results
Patient’s disposition and primary outcomes are shown in Figure 1. A total of 1274 adults were initiated on NNRTI-based cART. Of the 1274 participants, 588 (46%) had sufficient CD4 count and VL data both at baseline and at follow-up to be included in the primary outcome analysis. The median age of the patients included in the analysis was 34 years (interquartile range [IQR] 29–41 years). There were more females (65%) than males and most (53.7%) of the participants were married (Table 1). Median duration on ART was 12 months (IQR 6–12) at study end point.

Patient’s disposition and primary outcome.
Sociodemographic and Clinical Characteristics of 588 Patients on ART in Jos, Nigeria, and Association with Adherence to Pharmacy Drug Refill.

Abbreviations: ART, antiretroviral therapy; 3TC, lamivudine; EFV, efavirenz; NVP, nelfinavir; TDF, tenofovir; WHO, World Health Organization; ZDV, zidovudine. aData not available for 8 patients.
Baseline median (IQR) CD4 count and VL were similar among patients with insufficient follow-up data and those with sufficient follow-up data; (163 [88–264] vs 167 [91–167] cells/mm3 [
Adherence Characteristics
The mean cumulative percentage adherence to drug-refill visits was 94% (95% CI: 94-95). Overall, 250 (42.5%) patients had suboptimal adherence (<95%). In a separate analysis, sociodemographic characteristics were not significantly associated with adherence to drug pickup (Table 1).
Virologic Failure
In all, 94 (16%) of the 588 patients had virologic failure. Median interval of repeat VL test was 6 months (IQR 5–7). Variables associated with virologic failure (Table 2) in the bivariate analysis included sex, age, drug pickup adherence, pre-ART CD4 count, and pre-ART VL. After adjusting for other confounders (Table 3), suboptimal adherence to drug-refill visits was the only significant predictor of virologic failure (adjusted OR [AOR] 1.61; 95% CI: 1.15–2.27).
Demographic and Clinical Characteristics and Bivariate Analysis for the Association with Immunologic and Virologic Failure among 588 Patients on ART in Jos, Nigeria.
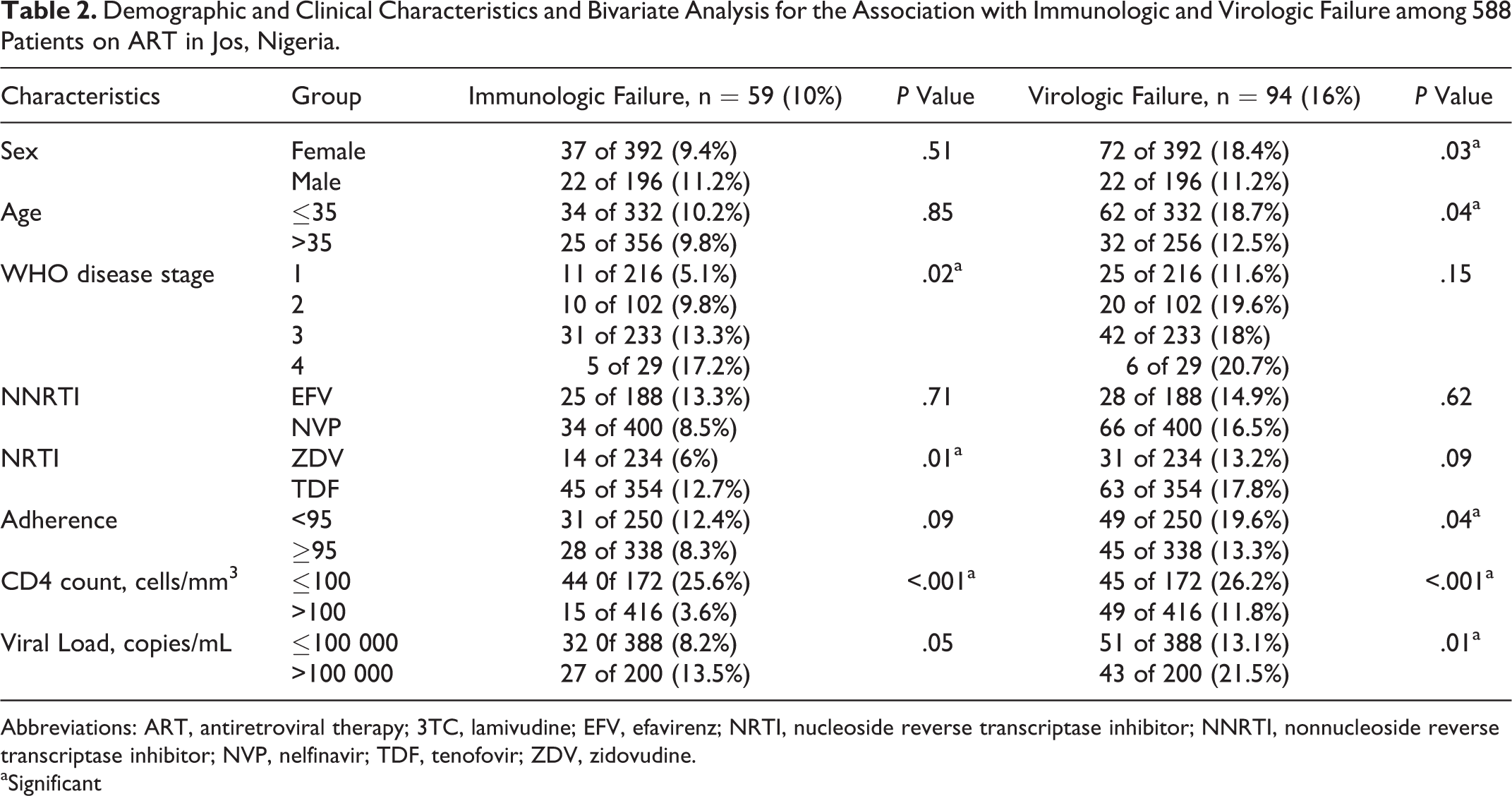
Abbreviations: ART, antiretroviral therapy; 3TC, lamivudine; EFV, efavirenz; NRTI, nucleoside reverse transcriptase inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; NVP, nelfinavir; TDF, tenofovir; ZDV, zidovudine. aSignificant
Adjusted Association Between Characteristics of Patient and Immunologic and Virologic Failure.

Abbreviations: ART, antiretroviral therapy; AOR, adjusted odds ratio; CI, confidence interval; NRTI, nucleoside reverse transcriptase inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor; VL, viral load; WHO, World Health Organization.
aAs defined by WHO.
bTwo repeated VL > 1000 copies/mL.
cSignificant.
Immunological Failure
A total of 59 (10%) of 588 met the criteria for immunological failure. In the bivariate analysis, factors associated with immunological failure included NRTI backbone (
Discussion
This study provides evidence for the usefulness of pharmacy-refill adherence measure in predicting virologic outcomes among patients on cART in a public care facility in Nigeria. Using 2 VL measurements during the first year of treatment, drug-refill adherence was significantly associated with virologic failure in our study participants. Patients with optimal adherence (≥95%) to drug-refill schedules had a better likelihood of achieving maximal viral suppression within the first year of treatment. Clinically, this is useful as systematic monitoring of pharmacy refill adherence, particularly in settings with limited capacity for VL testing, could be used as a surrogate test for virologic failure. The result of this study supports report of similar studies carried out in South Africa 25 and Cameroon. 26
The proportion (57.5%) of our study participants with complete adherence to drug-refill visits was lower than that reported in an earlier study conducted by Taiwo et al 27 in Jos. This reflects the dynamic nature of adherence to chronic therapies. Therefore, monitoring of adherence using validated methods should be part of standard clinical procedures. This will help to identify patients with suboptimal adherence and enhance the provision of targeted adherence interventions. “Unlike other investigators,” 17,26,28,29 we observed rates of viral suppression that were higher than drug pickup adherence. Although pharmacy-refill adherence has been shown to correlate with virologic response and mortality in developed and RLSs, 17,28,29 it gives no description of daily adherence to treatment and is limited in describing patient’s pill-taking behavior. Other investigators have noted discordance between adherence determined using pharmacy records and virologic response. 30 Furthermore, other studies have indicated that moderate adherence levels (70–90%) may be enough to achieve satisfactory virologic control with ARV regimens including boosted protease inhibitors or NNRTIs. 31 However, at the 95% adherence level, we can be quite confident that a very small proportion of individuals on NNRTIs are likely to have virological failure. 32,33 In addition, other measures of adherence, including subjective measures that were not evaluated in this study, 32,33 ought to be examined as should correctness of this approach in patients on non-NNRTI-based cART.
Like other investigators, 17 we found that, independent of adherence, patients initiating treatment at CD4 counts less than <100 cells/mm3 have a higher risk of immunologic failure within the first year of treatment. The benefits of initiating treatment at higher CD4 thresholds have been described in other studies. 34 -37 The most recent treatment guidelines favor treatment initiation at higher CD4 count thresholds. 38
As a result of inconsistent VL data, a large proportion (54%) of participants were excluded from the primary outcome analysis due to our strict inclusion criteria; however, comparison of patients included and excluded did not show any clinically significant differences in the baseline CD4 counts, VLs, and adherence profile.
Conclusion
Virologic suppression within the first year of treatment with an NNRTI-based regimen was significant in our study population. Adherence measured by pharmacy drug refill could serve as a useful predictor of early virologic failure. We, therefore, recommend that it should be used in routine monitoring of adult HIV-infected patients on cART in RLS.
Footnotes
Authors’ Note
This article was presented at the 8th International Conference on HIV Treatment and Prevention Adherence, Miami, Florida, USA, on June 2013. Oral Abstract # 175. We thank Kimberly Scarci for her support in critically reviewing the manuscript. We also thank the patients and staff of the JUTH HIV clinic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by the US Department of Health and Human Services, Health Resources and Services Administration (U51HA02522) and CDC through APIN (PS 001058). The contents are solely the responsibility of the authors and do not represent the official views of the funding institutions.
