Abstract
Background:
Sexually transmitted infections (STIs) significantly increase HIV transmission. Sexually transmitted infections may be asymptomatic and therefore remain undiagnosed in HIV-positive persons. Routine screening and treatment of STIs in HIV-positive high-risk populations in sub-Saharan Africa have not been described previously.
Methods:
We reviewed data from an HIV-positive high-risk population at the Infectious Diseases Institute, Makerere University, a large urban HIV clinic, between July 2011 and April 2012. Our high-risk population cohort included female sex workers, long-distance drivers, barmaids, taxi drivers, commercial motorcycle “boda-boda” riders, soldiers, police officers, prison officers, security guards, prisoners, and fishermen.
Results:
Of 355 participants enrolled in the high-risk population’s program, 21.4% were diagnosed with an STI either clinically or microbiologically. The STIs diagnosed in this population were syphilis, hepatitis B, genital herpes, human papilloma virus infection (condylomata acuminata), nongonococcal urethritis (NGU), and gonorrhea. Rates of syphilis, hepatitis B, genital herpes, condylomata acuminata, NGU, and gonorrhea were 8.5%, 7.0%, 5.4%, 1.4%, 1.4%, and 0.3%, respectively.
Conclusion:
Clinical and microbiologically diagnosed STIs were diagnosed in nearly one-fourth of the HIV-positive high-risk population. HIV care programs should note our high rates of STIs among HIV-positive high-risk populations and consider routine screening and treatment algorithms for these populations in their own settings.
Keywords
Introduction
There is an increased risk of HIV transmission in the presence of sexually transmitted infections (STIs). 1 –3 High-risk populations contribute significantly to the HIV pandemic and are believed to be core drivers of the epidemic in sub-Saharan Africa since they are at increased risk of transmitting HIV. 4 High-risk populations are populations that promote HIV and STI spread because they usually engage in risky sexual behaviors, and these include long-distance drivers, fishermen, men who have sex with men (MSM), barmaids, and female sex workers (FSWs). 5
In resource-limited settings, sexual contacts of HIV-positive high-risk populations are at an increased risk of acquiring HIV and other STIs due to sex with multiple partners and lack of condom use. 6 It is therefore essential to identify and treat STIs among these populations as many have been associated with an increased risk of HIV transmission, for instance genital ulcer disease. 7,8 Many STIs, particularly in women, are asymptomatic and may remain undiagnosed and untreated, 9,10 further increasing the potential for both STI spread and HIV transmission.
Screening algorithms to identify undiagnosed STIs have not been described and introduced for HIV sero-positive high-risk populations. In Uganda and in most of sub-Saharan Africa, no such screening algorithms currently exist for HIV sero-positive high-risk populations. Following the introduction of a high-risk population cohort at the Infectious Diseases Institute (IDI), Kampala, in July 2012, we have successfully screened over 350 HIV sero-positive high-risk persons. This article presents our findings in our high-risk population cohort accessing care at the IDI.
Methods
The IDI established a high-risk population HIV/AIDS cohort to provide holistic HIV care with integrated sexual and reproductive health services. This prospective cohort was established in July 2011, and patients were seen by a dedicated high-risk population’s clinic team that consisted of a counselor, nurse, and medical officer. High-risk populations have been described as populations that engage in risky sexual behavior. 5,6
In the context of IDI, the high-risk population cohort consisted of HIV sero-positive commercial motorcycle “boda-boda” riders, taxi drivers, long-distance drivers, fishermen, prisoners, uniformed persons (police officers, soldiers, prison officers, and security guards), barmaids, and FSWs. Participants in the high-risk population cohort were identified from the general clinic and referred by peer educators, triage nurses, clinic officers, medical officers, and clinic counselors to the dedicated high-risk population’s team (Figure 1). Participants were then followed-up consecutively by the IDI high-risk population clinic team on a monthly basis.

Patient enrollment flow diagram showing the categories of patients enrolled and screened for sexually transmitted infections.
High-Risk Population Clinic Approach
Because of the uniqueness of our high-risk HIV cohort—these populations engage in risky sexual behaviors and have multiple sex partners—it was important that we maintained the confidentiality of our participants as they came to the clinic to access care.
We decided to have the high-risk medical officer sit in the general clinic and utilize one of the usual general consultation clinician rooms to see the high-risk HIV-positive patients interchangeably with the HIV-positive patients from the general cohort so that both the general cohort HIV-positive patients and the clinicians were unable to identify who the high-risk patients were.
This technique was also reciprocated by the counselor— seeing high-risk patients interchangeably with general clinic patients. This procedure made the high-risk patients comfortable enough to continue coming to this specialized clinic and additionally to bring their peers.
Laboratory Monitoring and Treatment Protocols
At the first visit, blood samples of all high-risk population participants were drawn for syphilis and hepatitis B screening (Figure 1). Syphilis infection was established by a positive reactive rapid plasma reagin (RPR) and confirmed with a reactive Treponema pallidum hemagglutination assay (TPHA). Syphilis results were obtained in 15 to 20 minutes and if reactive, the results were given to the patient in the presence of a counselor. Participants were considered to have syphilis when both RPR and TPHA tests were positive. Participants with a reactive RPR and TPHA result received posttest counseling and treatment for syphilis and were asked to refer their sexual partners if possible for evaluation and possible treatment.
At the second visit, the hepatitis B results would be given to the participant still in the presence of a counselor. Hepatitis B infection was based on a positive hepatitis B surface antigen result. Participants with HIV/hepatitis B coinfection were managed according to World Health Organization (WHO) guidelines with a tenofovir/lamivudine (TDF/3TC)-containing regimen regardless of the absolute CD4 counts. 11
In addition to laboratory testing for all high-risk population participants, symptomatic female participants had genital examinations with cervical and high vaginal swabs for microbiological analysis. Symptomatic male participants had genital examinations with urethral swabs for microbiological analysis.
Sexually transmitted infections that were diagnosed clinically by thorough genital examinations, serologically, and microbiologically were treated according to Centers for Disease Control and Prevention STI treatment guidelines. 9,10
At the 6th month visit, CD4 count monitoring was conducted for all participants. Baseline complete blood counts and CD4 counts were done for new participants. All patients with an absolute CD4 count <200 cells/mm3 or classified as WHO clinical stage IV were initiated on antiretroviral therapy (ART), according to WHO ART guidelines. 12 Data were entered into a high-risk population’s database and analyzed using STATA version 11.0.
Results
Between July 2011 and April 2012, 355 people were enrolled in the high-risk population’s program. All 355 persons were screened for syphilis and hepatitis B. Demographic characteristics of the study population are presented in Table 1. Of the 355 participants, 76 (21.4%) were diagnosed with either 1 or more STIs. Although syphilis and hepatitis B were most common, other STIs like human papillomavirus (HPV) infection (condylomata acuminata), genital herpes, gonorrhea, and nongonococcal urethritis (NGU) were also diagnosed in this population.
Characteristics of the Study Population.
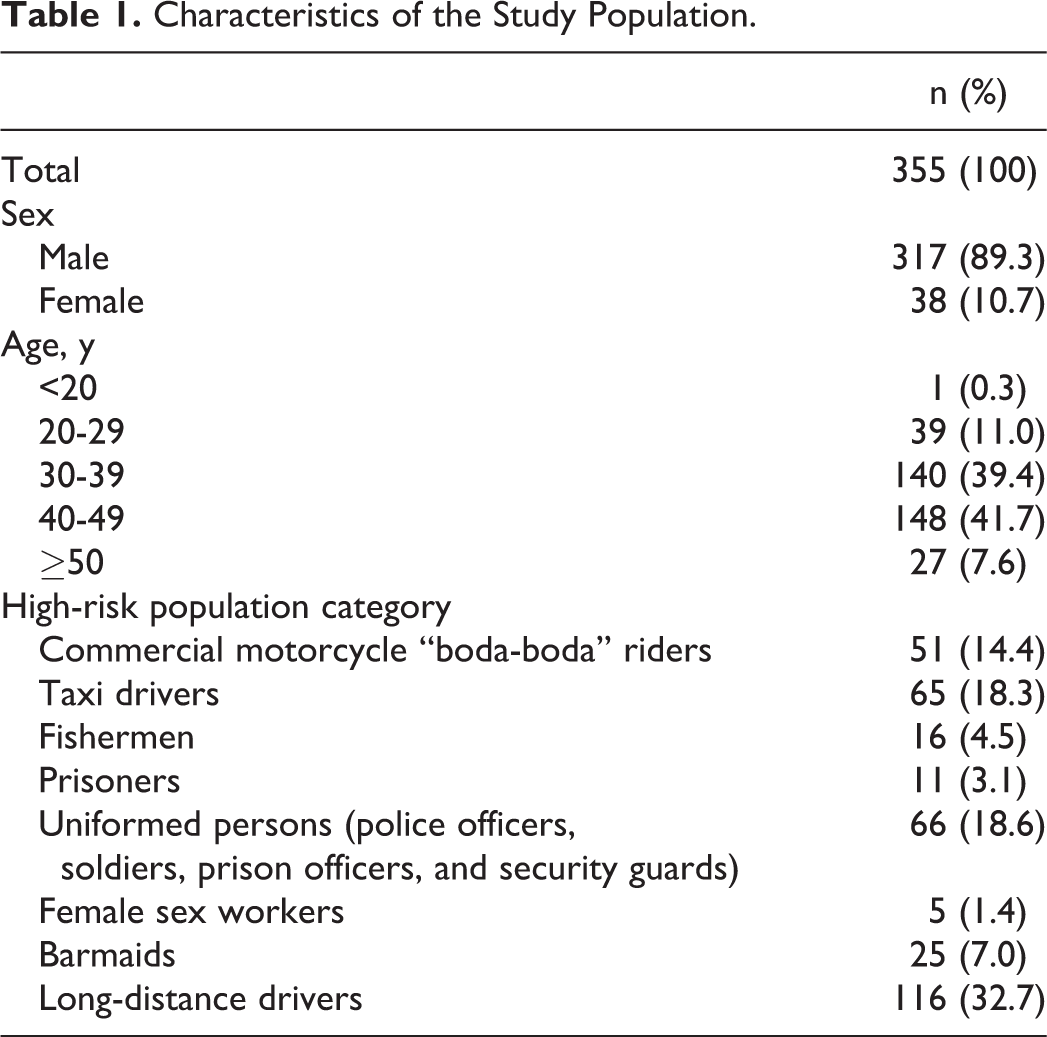
New syphilis infections were found in 30 (8.5%) participants, hepatitis B in 25 (7.0%) participants, genital herpes in 19 (5.4%) participants, HPV (condylomata acuminata) infection in 5 (1.4%) participants, NGU in 5 (1.4%) participants, and gonorrhea in 1 (0.3%) participant.
Syphilis and HPV (condylomata acuminata) infections were found to be highest among prisoners at 18.2% and 9.1%, respectively; hepatitis B among FSWs at 20%; and genital herpes among commercial motorcycle boda-boda riders at 10%. Female sex workers and fishermen had a higher rate of prevalent STIs than the rest of the high-risk population (relative risk [RR] = 1.89; 95% confidence interval [CI]: 0.63-5.64 and RR = 1.82; 95% CI: 0.93-3.54, respectively; Table 2). The 65 HIV-infected taxi drivers and the 11 prisoners additionally had a higher rate of prevalent STIs as compared to the rest of the high-risk cohort (RR = 1.38; 95% CI: 0.88-2.18 and RR = 1.74; 95% CI: 0.77-3.90, respectively; Table 2). The odds of having an STI were reduced by 61% among long-distance truck drivers (P < .001; Table 2).
Prevalence Ratios of STIs among High-Risk Cohort.
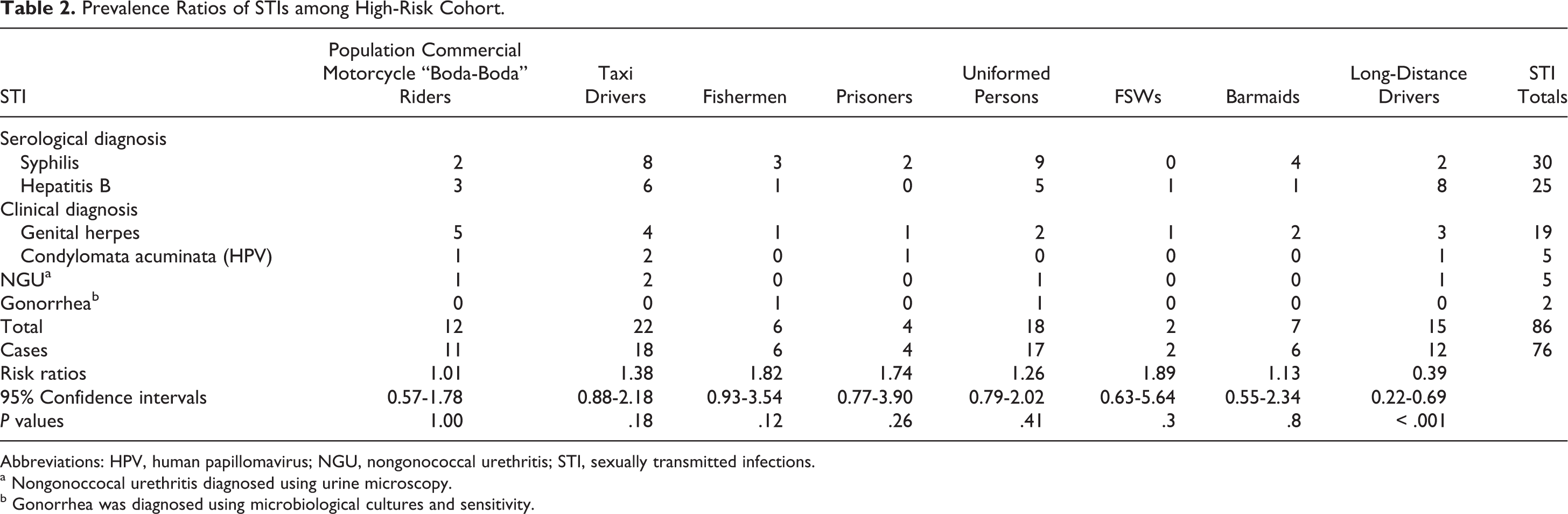
Abbreviations: HPV, human papillomavirus; NGU, nongonococcal urethritis; STI, sexually transmitted infections.
a Nongonoccocal urethritis diagnosed using urine microscopy.
b Gonorrhea was diagnosed using microbiological cultures and sensitivity.
Discussion
Our high-risk population cohort is the largest yet studied to date for the presence of multiple STIs in the setting of HIV coinfection. This is also the first Ugandan study of screening and treating STIs among high-risk populations within an HIV care program. High-risk populations remain the core drivers of the HIV epidemic in sub-Saharan Africa, 4 and several studies have demonstrated the increased risk of HIV transmission in the presence of STIs. 13 –19
Our results suggest that targeted STI detection and treatment programs within HIV programs for high-risk populations may play a vital role in HIV prevention in sub-Saharan Africa. Population-based STI screening among HIV-positive persons in western cohorts have revealed high rates of STIs. 20,21 Screening for STIs in high-risk populations particularly among MSM in Spain and Switzerland and military personnel in the United States has yielded high rates of STIs among asymptomatic patients. 22 –24 Almost a quarter of our high-risk cohort was diagnosed with an STI. These included barmaids, FSWs, prisoners, fishermen, uniformed persons (police officers, soldiers, prison officers, and security guards), commercial motorcycle boda-boda riders, long-distance drivers, and taxi drivers (Table 2).
Commercial drivers in particular are highly mobile, sexually active outside the home, often have money to purchase sex, and are therefore at higher risk of acquiring and transmitting HIV and other STIs than the general population. Prevention specialists have characterized them as “mobile men with money,” and they contribute disproportionately to the spread of STIs and HIV in sub-Saharan Africa. 25,26 Soldiers have also been identified as vulnerable to HIV acquisition and transmission as they are frequently away from home and, as a consequence, may engage more frequently in risky sexual practices than the general population. 27,28 Finally, recent studies in Uganda and Tanzania have indicated that barmaids engage in risky sexual behavior at a greater frequency than the general population. 5,29
Our study demonstrated that the odds of having an STI were significantly reduced by 61% among long-distance truck drivers (P < .001). This is probably because there have been extensive programs with both national and regional commitment targeting long-distance truck drivers. 30 On the contrary, STI risk ratios in the other high-risk groups were not significant.
Recent data have confirmed that high-risk populations have not significantly changed their sexual behavior in response to the HIV/AIDS pandemic. 5 Because of the risk that these groups may pose to their multiple sexual partners, our high-risk population’s program has established a screening algorithm for the diagnosis and treatment of STIs among HIV-positive high-risk persons to decrease the transmission of HIV and STIs (Figure 1).
Our study has several limitations. Herpes simplex virus (HSV) serology was not available for the patients due to the high costs involved and therefore the diagnosis of genital herpes was established on a clinical basis alone. Given the high prevalence of HSV-2 in sub-Saharan Africa as evidenced by HSV seroepidemiologic surveys, 31,32 we may have underdiagnosed HSV infection and, as a consequence, underestimated its contribution to the STI burden in our high-risk cohort.
Other limitations that could have possibly led to underestimation of the STI burden in this population were the absence of bacterial STI testing kits, particularly chlamydia nucleic acid amplification and trichomonas vaginalis testing, which were not available due to resource constraints in our setting. In addition, due to the limited resources we had in our setting, it was only symptomatic participants who had vaginal, cervical, or urethral swabs for microscopy and culture to detect bacterial STIs, which may have further underestimated subclinical infections.
Despite these limitations, our study confirms that STIs are common among certain HIV-positive high-risk groups. HIV care programs can expect that a significant proportion of their high-risk HIV-positive patients will also harbor undiagnosed and untreated STIs. HIV care programs should make it a priority to develop and adopt screening and treating algorithms for STIs for their high-risk populations in order to prevent both HIV and other STI transmission to their current and future sexual partners.
Footnotes
Acknowledgments
The authors acknowledge the high-risk populations’ team and peer educators, triage, and clinic nurses who helped in mobilization. The authors thank the high-risk population’s counselors who helped in mobilization and behavioral change counseling: Julian Nkurayija, Joseph Mawejje, Simon Wejuli, Adelline Twimukye, Rachael Nabbagala, and Phoebe Nabongo. The authors thank the statistics department at the IDI for their invaluable assistance: Agnes Kiragga and Joseph Sempa. Finally, we thank all the participants who have enrolled in the high-risk population’s program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Civil Society Fund (CSF) of Uganda [grant number CSF/003/2010].
