Abstract
Background:
Studies have identified several programmatic and nonprogrammatic indicators that affect adherence to highly active antiretroviral therapy (HAART). Depression has been shown to impact adherence to HAART. This cross-sectional analysis of data collected from Nigeria, Uganda, Zambia, and Tanzania in 2008 examined the relationship between levels of depressive symptoms, clinical progression, and adherence to HAART.
Methods:
A multinational, multicenter, observational, retrospective cross-sectional evaluation of a population of focus comprised randomly selected patients on HAART. The dependent variable was adherence to HAART. The primary variable of interest to be assessed was patients’ level of depressive symptom score. A multivariable logistic regression model was used to examine the relationship between explanatory variables and adherence to HAART.
Results:
A total of 2344 patients were recruited for adherence survey. About 70% of the study sample reported having some level of depression. Logistic regression results show that patients who reported, respectively, low, moderate, and high levels of depressive symptoms are 35% (P < .001), 56% (P < .001), and 64% (P < .001) less likely to adhere to HAART than those who reported having no depressive symptoms. At multivariate analysis, adherence to HAART was independently associated with the levels of depressive symptoms, older age, CD4 count >200 cells/mm3, Truvada (tenofovir [TDF]/emtricitabine [FTC])-based regimens, good knowledge about HAART, and longer period on therapy.
Conclusions:
These results indicate that mental health and clinical parameters are significant factors in determining patients’ adherence to their HAART, which need to be more aggressively addressed as a critical component of care and treatment support.
Introduction
Broad access to highly active antiretroviral therapy (HAART) has drastically diminished the onerous effects that the HIV/AIDS once had on those infected by the virus, as evidenced by patients living longer and leading healthier lives. However, achieving such benefits hinges around optimal adherence to a mutually agreed treatment plan between the patient and the health care provider. Several studies have demonstrated the correlation between adherence and viral suppression. 1 –3
Mental illness is one aspect that is not appropriately addressed in the care of HIV-infected patients in resource-limited settings. 4,5 People infected with HIV are more likely to have depression than those not infected, with studies finding that anywhere from 8% to 50% of HIV-infected individuals have significant depressive symptoms. 5 –10 A study estimated the prevalence of depression and anxiety among people living with HIV/AIDS (PLHIV) at nearly 50% in the United States. 11 In developing countries, depression is highly prevalent among PLHIV, but it is underdiagnosed. 12 The prevalence of depression in low-income settings is between 21% and 63.3%. 4,13 –15 The presence of depression has been shown to be an important patient-related barrier to adequate adherence to HAART. 3,9,16,18 Treatment adherence (defined as making sure to take every pill as prescribed by the health care provider) is critically important to successful treatment of HIV, because missing even a few doses gives the HIV virus an opportunity to develop immunity to the medication. Poor adherence is related to worse medical outcomes, but even a small rate of improvement in adherence may improve these outcomes. 19
Despite the known beneficial effects conferred by optimal adherence, the average rate of adherence to HAART is estimated at 70%. 20 The failure to attain optimal adherence could be explained at least in part by the complexities of factors associated with medication adherence. These factors have been variously grouped under 5 categories, namely treatment regimen, disease characteristics, patient–provider relationship, clinical setting, and patient variables. 21 –26
However, few studies have looked at the prevalence and the association of depression, clinical biomarkers, and nonadherence in people living with HIV in resource-limited settings. To fill the existing gap in a body of literature on managing the mental health of patients who receive HAART and adherence, this research is designed to improve the quality of care of patients in the AIDSRelief program by assessing the association between depression and adherence. The aim of this study was to assess the relationship between depressive symptoms, CD4 count, and adherence to antiretroviral therapy (ART) in 4 countries in sub-Saharan Africa (Nigeria, Uganda, Zambia, and Tanzania).
Methods
Study Setting
Nigeria, Tanzania, Uganda, and Zambia are some of the countries in Africa, where the US Presidents’ Emergency Plan for AIDSRelief (PEPFAR) is being implemented. To ensure that optimal care was provided, continuous quality improvement measures were carried out at fairly regular intervals. One of these measures was the patient-level outcome that involved a randomly selected number of patients who had been on treatment for at least 9 months. These selected patients were administered an adherence survey by a trained professional. All patients received a consent form and were explained the nature of the evaluation, the benefits, the risks, and the option of opting out without negatively impacting their treatment. A chart abstraction was also performed on each of the selected patients. The analysis was conducted on 2344 randomly selected eligible adult patients who had been initiated into HAART between August 2004 and April 2005.
Study Design
The study design involved a multinational, multicenter, observational, retrospective cross-sectional evaluation of a population of focus comprising randomly selected patients on HAART enrolled in the AIDSRelief program in selected antiretroviral treatment sites, which cover the majority of the cases and which were set up in rural Nigeria, Tanzania, Uganda, and Zambia in 2008.
Study Variables
The primary variable of interest was adherence to HAART. Explanatory variables were depressive symptoms, CD4 count, age, gender, baseline CD4 count, and new opportunistic infections, and several other covariates were also used.
Data Abstraction
For medical chart abstraction, information including patients’ age, gender, baseline CD4 count, regimen type and combination, and the total number of active and new opportunistic infections since HAART initiation was collected on the standardized chart abstraction forms.
Measurements
Depression Symptom Severity
We used the 5-item depression subscale from the Beck depression scale as screening questions for depression and mental health. The Beck Depression Inventory (BDI) has been used in other studies to assess both somatic and cognitive aspects of depression in PLHIV. 10,27 In this survey, depressive symptoms were measured from 5 questions related to mental health issues: (1) In the past month, have you had persistent feelings of sadness/hopelessness that you just can’t control? (2) How often do you feel the need to be alone? (3) In the last month, have you lost interest in what used to give you pleasure? (4) In the past month, how often have you felt confused or not yourself? (5) How often do you feel that life is too difficult for you to go on? The responses for all these 5 questions were always, sometimes, and never. The data coded responses of always as 2, sometimes 1, and never as 0. A summative score was created ranging from 0 and 10. The total scores were then categorized into 4 categories—no depression, low depression, medium depression, and higher depression. The reliability of the scale has a Cronbach α of .74.
Adherence
In this cross-sectional study, ART adherence was obtained from the combined indictors for both dose adherence and schedule instructions. When a person is said to be adherent to schedule, he or she always follows scheduling instructions; and a person is said to be dose adherent, if he or she took ≥95% of the prescribed doses correctly in the past 7 days.
Highly Active Antiretroviral Therapy Knowledge Score
The respondents were asked 14 questions related to HIV, use of HAART, and consequences of stopping the medication.
Statistical Analysis
Data were entered into an access database and analyzed using STATA 9. 28 The statistical analyses included descriptive and analytical procedures. For descriptive purposes, the frequencies of the variables were examined first to explore the data. Proportions were calculated for categorical variables. For analytical purposes, univariate and multivariable logistic regression models were estimated, and odds ratios were used to characterize the association between levels of adherence with depressive symptoms and CD4 count while controlling for other covariates. For all analyses, P < .05 was considered statistically significant.
Ethics Statement
The study protocol was approved by the institutional review board (IRB) at the University of Maryland School of Medicine. National IRB approval was also requested from each country that was involved in the study. Written consent was obtained from each eligible adult participant. Patient names and identifiers were not collected.
Result
A total of 2344 randomly selected patients agreed and responded to adherence survey questions. The sample had more women than men (68% versus 32%). The patients were of a mean age of 38 years, had a mean baseline CD4 count of 227 cells/mm3, were all on first-line HAART regimen with equal proportions on stavudine (D4T)-, zidovudine (ZDV)-, and Truvada (tenofovir [TDF]/emtricitabine [FTC])-based regimens. A good proportion (77%) of patients showed a good adherence to HAART. About 77% of the patients had at least some level of depressive symptoms (low depressive symptoms 30%, medium depressive symptoms 22%, and high depressive symptoms 15%). Most of the patients had a good knowledge of HAART. Similarly, on average, the majority of the cases had at least 2 opportunistic infections and had been on treatment for 2 years (Table 1).
Characteristics of the Study Samples from Nigeria, Tanzania, Uganda, and Zambia, in 2008.

Abbreviations: FTC, emtricitabine; HAART, highly active antiretroviral therapy; SD, standard deviation; TDF, tenofovir.
Predictors of Adherence to HAART
At multivariate analysis, adherence to HAART was independently associated with the low levels of depressive symptoms (adherence reducing with the increasing levels of depressive symptoms). Other factors independently associated with adherence were age (older patients adhering better than younger ones), CD4 counts >200 cells/mm3 with better adherence levels, type of regimen (those on D4T- and ZDV-based regimens are less likely to adhere than those on TDF/FTC-based regimens), knowledge about HAART, and number of months on therapy (Table 2).
Crude and Adjusted Odds Ratios for the Logistic Regression Estimates for the Relationship between Depressive Symptoms and Adherence to HAART in Samples from Nigeria, Tanzania, Uganda, and Zambia, in 2008.
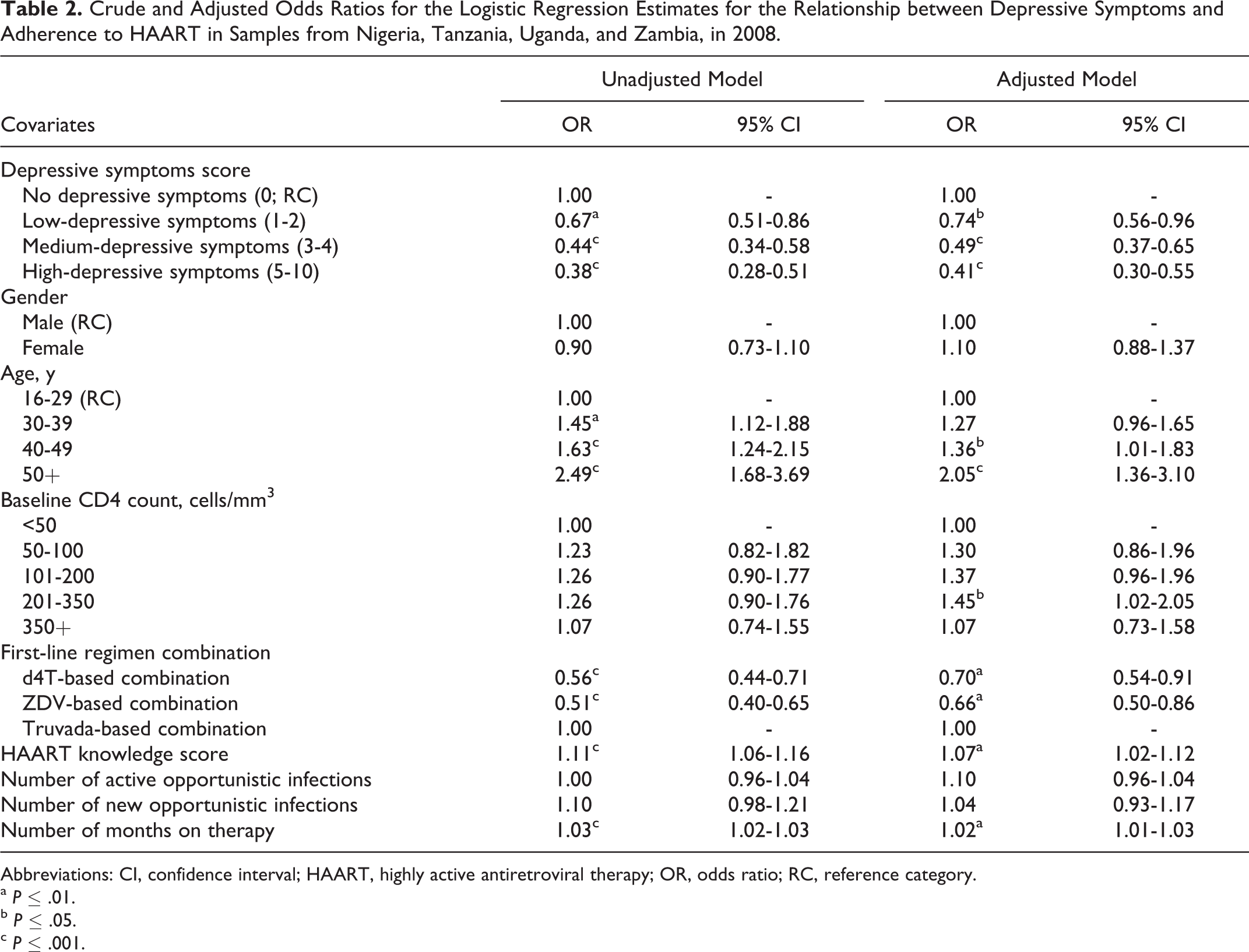
Abbreviations: CI, confidence interval; HAART, highly active antiretroviral therapy; OR, odds ratio; RC, reference category.
a P ≤ .01.
b P ≤ .05.
c P ≤ .001.
Discussion
This study found a high rate of adherence (77%). Despite the critical need for strong HIV treatment adherence, research indicates that many patients have difficulty in realizing this goal. A meta-analysis of 59 studies conducted in North America and Africa reported that only 55% of North American patients demonstrated high levels of HAART adherence. 29 Another study conducted in East Africa showed that the percentage of African patients who achieved high adherence was more favorable and consistent with our findings. 30,31 However, this proportion could decline over time as patients initiating therapy encounter the challenges of maintaining long-term adherence and as treatment availability in these nations expands beyond those with early access to HAART.
Our study showed that the prevalence of depressive symptoms was very high (77%) when compared to other studies, and it was found to be between 20% and 63%. 4,6 –11,13 –15 Our study found that adherence reduced with increasing levels of depressive symptoms among patients on HAART. This confirms the earlier findings that also showed depression was associated with poor adherence. 10 –13,19,27,32 –34 Similarly, a study by Wagner et al also found decreasing levels of adherence with increased severity of depression, 12 although their study had a smaller sample size (1374) than that of our study. Similarly, in one of the meta-analysis studies, the estimated odds of a depressed patient being nonadherent is 1.76 times the odds of a nondepressed patient being nonadherent. 36 In a study in Tanzania involving 150 adult participants who had received HAART for at least 6 months, depression was not associated with incomplete adherence, although this might have been due to the small sample size. 36
Depression has been linked to poor HIV outcomes due to nonadherence, increasing HIV progression and doubling the risk of mortality. 13,31,37 –39 PLHIV with depression are prone to nonconsistent drug intake and likely to discontinue treatment resulting in nonvirologic suppression. 34 This is because PLHIV with depression have negative thoughts and feelings of hopelessness, which discourage them from perfect adherence to treatment. 4,19,39 Optimal clinical benefits associated with HAART, however, demand rigorous adherence to medication dosing schedules and other aspects of treatment. Research has identified high adherence to ART regimens as the most important predictor of viral suppression, improved CD4 count, delayed progression to AIDS, and patient survival. 40 Although more potent medication regimens can allow for effective viral suppression at moderate levels of adherence, improving adherence to any degree can only increase the likelihood of suppressing the virus and postponing disease progression. 41 Striving for the highest possible level of adherence therefore remains essential for optimizing HIV treatment outcomes.
Furthermore, depression increases social isolation, creating a gap between PLHIV and their support networks that would ideally encourage adherence. Other studies have found social support to be linked to keeping to medical appointments in HIV care. 34 This suggests that improvements in depressive symptoms could lead to better adherence and virologic outcomes. Antelman suggests that while interventions for depressed PLHIV exist, there are other barriers to accessing these services such as stigma, lack of confidentiality, and other logistical constraints. 37 Thus, for supportive interventions to be effective, they must address these barriers.
Although most studies have mostly examined the association between depression and nonadherence, their samples have been relatively small compared to our study that had a large sample size. We are only aware of 1 previous study that considered how the different severity levels of depression may differentially influence adherence, 42 and our study took this into account and has demonstrated that adherence reduces with the increasing severity of depressive symptoms.
Our study also showed a direct relationship between adherence, age, and CD4 counts. Younger patients aged 16 to 29 years were less likely to adhere than their older counterparts. This could be due to factors such as stigma and denial. 31 However, other studies have shown that age does not significantly affect adherence. 43,44 Similarly having CD4 counts lower than 200 cells/mm3 was associated with less adherence to HAART. Being a cross-sectional study that precludes causality, this association can be viewed to mean that either low adherence leads to less viral suppression, thus lower immunity, or those just starting HAART (with low CD4 counts) are less likely to adhere to treatment than those who are on HAART for a long time and attained immune recovery. The direct relationship between duration on HAART and adherence has further confirmed this hypothesis. Patients who are on HAART for a longer duration have continuous counseling support and are also less likely to experience drug side effects such as nausea and vomiting usually observed at HAART initiation, thus reinforcing their adherence. The study by Gordillo et al showed similar findings except for the duration of HAART. 10
The study has the following limitations: first, the study used a cross-sectional design that did not take into consideration the fact that adherence and depressive symptoms are dynamic, and study results are correlations limiting causal inferences. Second, this is a self-reported measure of depressive symptoms and the measures may be biased and not completely true. Third, although BDI has been used in other similar settings, 45 –47 it has not been validated in HIV-positive patients initiating therapy in these 4 countries. Further research is consequently required so as to ensure the reliability and validity of the BDI in HIV-positive settings. Fourth, our study did not look into intercountry or regional differences. Despite this, the study had a large sample size and was conducted in a multinational sample.
Implications for Practice
This analysis provides evidence that depressive symptoms are associated with poor adherence to HAART medication. Although this study cannot assess causality, it supports the importance that must be placed on depression in studies that assess adherence and attempt to improve it. The study demonstrates that counseling should be intensified for PLHIV on HAART, for those younger than 30 years of age, and for those with a CD4 count of less than 200 cells/mm3 as they are at greater risk of nonadherence. More research in these groups needs to be done to find out the factors associated with poor adherence to therapy so as to tailor interventions. There is a need to sensitize health workers to identify depressive symptoms among PLHIV and empower them to respond appropriately. Designing a simple tool for screening to identify depressed PLHIV in care is important in integrating depression treatment and informing decisions about HAART initiation in patients with depressive symptoms.
Conclusions
This study confirms the relationship between adherence and depressive symptoms and highlights that adherence is more difficult with increased severity of depressive symptoms. Therefore, there is a need to strengthen mental health services within HIV care programs with clear budget lines for mental health. Improvements in effective diagnosis and treatment of depression will ensure maximum benefits of HIV treatment. Operations research within HIV programs should be encouraged to promote gathering evidence on ways of improving service delivery and promoting holistic care.
Footnotes
Acknowledgments
We are thankful to Dr Robert Redfield for his stewardship. We are grateful to all patients and health care providers who were involved in this quality improvement process. The views expressed in this article are solely the responsibility of the named authors and do not necessarily reflect the the US Presidents’ Emergency Plan for AIDSRelief (PEPFAR) decisions or stated policy.
Authors’ Note
PM and CS coordinated the study and drafted the manuscript, MEM designed the study and drafted the manuscript, SA designed and coordinated the study, MBH designed the study and performed the statistical analysis, and SB interpreted the data, helped in drafting the manuscript, and critically reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AIDSRelief is supported by the US government and funded under the Presidential Emergency Plan for AIDS Relief.
