Abstract
Background:
It is unclear whether HIV-related factors modify risk of hypertension (HTN). In a cohort of patients with AIDS, the authors determined HTN incidence and prevalence and assessed associated traditional, HIV-specific, and retinal vasculature factors.
Methods:
Prospective observational cohort included 2390 patients with AIDS (1998-2011). Univariate analysis was used to assess the impact of traditional- and AIDS-related risk factors for HTN prevalence and incidence. Multivariate regression analyses were used to evaluate the adjusted impact of these factors.
Results:
Hypertension prevalence was 22% (95% confidence interval [CI] 21%-24%) and was associated with traditional HTN risk factors (age, black race, and higher weight) as well as diabetes, hyperlipidemia, time since AIDS diagnosis, and higher CD4 counts. Hypertension incidence was 64.1 per 1000 person-years (95% CI 58.7/1000-69.9/1000). Age, race, weight, and diabetes were associated with incident HTN but HIV-specific factors were not.
Conclusions:
Hypertension, a prevalent cardiovascular risk factor in patients with AIDS, is associated with traditional and metabolic risk factors.
Introduction
In the era of highly active antiretroviral therapy (HAART), chronic, noninfectious medical conditions, such as cardiovascular (CV) disease, are emerging as the significant causes of morbidity and mortality among HIV-positive patients. 1 –10 In this context, convincing evidence suggests that HAART should be prescribed for patients without interruption and at increasingly higher CD4 thresholds, despite its association with metabolic and CV risk and disease. 6, 11 –17 Hypertension (HTN) is among the traditional risk factors that contribute to CV disease in adults with HIV infection. 18 –20 The availability of effective antihypertensive medications allows the adverse consequences of HTN to be mitigated. 21 Efforts to bring the effect of CV risk reduction in HIV-positive adults on antiretroviral therapy (ART) should therefore target HTN as a known, modifiable risk factor.
To date, a small, diverse literature has identified traditional risk factors for HTN in HIV-positive adults. These factors include older age, male sex, African American race, higher body mass index (BMI), and elevated cholesterol. 22 –24 These studies also associate several HIV-related factors with HTN, including HAART initiation in treatment-naive patients, duration of antiretroviral (ARV) medication use, and longer time since HIV diagnosis. 22, 24 Inconsistent data exist about the effect of nonnucleoside reverse transcriptase inhibitors (NNRTIs) on HTN in HIV-infected adults. 24, 25 These studies encompass patients both with and without a diagnosis of AIDS. Because of the increased risk of mortality among HIV-positive patients with AIDS, it is unclear whether these patients have the same risk of developing HTN as do other HIV-positive patients. 26
Clinical markers of HTN are also important to be identified in HIV-positive patients in order to determine the HTN risk and modify disease burden. Retinal vasculature provides a noninvasive source of end-organ information about CV changes associated with HTN. In the general population, several studies have demonstrated an association between the development and the progression of HTN and narrower retinal arteriolar diameters. 27 –31 An association between prevalent HTN and baseline retinal vascular caliber among HIV-positive patients in the cohort examined in this study has been described briefly. 32 To the authors' knowledge, no studies have examined the association between retinal arteriolar diameters and incident HTN in patients with AIDS.
The aim of this study was to assess traditional, HIV-related, and retinal vascular factors associated with prevalent and incident HTN in a large, prospective cohort of patients with AIDS in the era of HAART. The authors hypothesized that traditional HTN risk factors and duration of HIV infection would be significantly associated with both prevalent and incident HTN. The authors further hypothesized in secondary analysis of patients without a history of ocular opportunistic infections (OOIs) that central retinal arteriolar equivalent (CRAE) narrowing would be associated with incident HTN.
Methods
The Longitudinal Study of the Ocular Complications of AIDS (LSOCA) is a prospective observational study of patients with AIDS that began in September 1998. The study is designed to document the prevalence, incidence, and course of AIDS-related ocular complications in the era of HAART. Eligible participants are ≥13 years of age and have an AIDS diagnosis according to the 1993 US Centers for Disease Control and Prevention Case Surveillance definition of AIDS. 33
Participant recruitment and study design have been described in detail elsewhere. 34, 35 Briefly, recruitment was performed at 19 AIDS ophthalmology centers across the United States. Participants with or without opportunistic ocular infections and across a spectrum of immunologic function were included in the cohort on a rolling basis. The study and the protocol were approved by institutional review boards at all participating centers, and enrolled participants provided written informed consent.
At enrollment and follow-up, participants provided standardized medical histories to trained study personnel. According to the study protocol, this data collection was primarily by self-report via interview, with review of participants’ medical chart or contact of medical care providers as necessary to clarify or confirm elements of the history. The medical history included details about HIV course, AIDS-related illnesses, and ART. Data on non-AIDS medical comorbidities (HTN, diabetes, and hyperlipidemia) were also collected. Beginning in 2005, cigarette smoking and CV events were collected at baseline. Blood pressure medication use was recorded as of 2006.
Participants underwent baseline and follow-up ophthalmologic examination. Retinal photographs were taken at study entry, every 6 months thereafter for participants with OOI or every 5 years thereafter in participants without OOI. Retinal microvascular measurements and indices were determined in participants without OOI in a semiautomated manner by certified graders at the University of Wisconsin Fundus Photograph Reading Center (IVAN software, Department of Ophthalmology and Visual Science, University of Wisconsin, Madison, Wisconsin) using a standardized protocol described previously. 36 These analyses resulted in 2 specific measurements of the retinal microvasculature, the projected caliber sizes of the central retinal artery (CRAE) and the central retinal vein (central retinal vein equivalent [CRVE]). 37 –39
Beginning in 2005, participants had direct weight and 1-time height measurements recorded, with subsequent 1-time BMI calculations. Also beginning in 2005, the participants had direct blood pressure measurements. Using a standardized method, blood pressure was taken once in a seated position at each study encounter. Laboratory tests performed at enrollment include CD4 and quantitative plasma HIV RNA levels (HIV viral load). Follow-up visits occur every 3 to 6 months for participants with an OOI, such as cytomegalovirus (CMV) retinitis, and every 6 months for those without an OOI.
Outcomes
Presence of HTN was determined by data collected per study protocol during the standardized baseline and follow-up medical history interviews as described previously. A question was added to the medical history data collection forms of September 30, 2005, specifically asking whether the patient was taking any HTN treatments. Prior to that date, ascertainment of hypertensive treatment was obtained from an open-ended question asking about the use of non-HIV/AIDS-related treatments. Among 478 participants enrolled after September 30, 2005, 96% had agreement between their responses to taking any HTN treatment and reported HTN obtained during the study interview (κ statistic = .90; 95% confidence interval [CI] 0.85-0.94). Because of this high agreement, the presence of HTN was defined for all the participants as either reported HTN diagnosis or report of taking hypertensive treatment using the open-ended question. Participants who reported a history of high blood pressure requiring medication or reported taking any antihypertensive medications were therefore categorized as having prevalent disease. Those who reported neither at baseline, but did subsequently in follow-up, were coded as having incident HTN. Given the limited numbers, the blood pressure measurements were not included in the outcome.
Predictors
The primary predictors of prevalent and incident HTN were traditional HTN risk factors (age, gender, race, and weight), baseline indicators of AIDS duration, severity and disease control (mean number of years since AIDS diagnosis, median CD4 count, mean HIV viral load, median nadir CD4 count, and mean peak HIV viral load), and HIV treatment history (HAART use and duration, protease inhibitor [PI], or NNRTI exposure at enrollment).
Secondary Analysis
To test the hypotheses about CRAE and HTN and to control for OOI effect on retinal vessels, the authors performed a secondary analysis in participants without a documented history of OOI. Models used in this analysis included the same variables used to assess HTN incidence and prevalence in the general cohort, with the addition of CRAE and CVRE measurements as predictors.
Covariates
The authors adjusted all the models for sociodemographic covariates, components of metabolic syndrome (hyperlipidemia and diabetes), CMV retinitis, and hepatitis C coinfection.
Statistical Analyses
Data obtained and keyed into the database as of December 31, 2011 were included. Unadjusted associations of enrollment characteristics with prevalence of self-reported HTN or antihypertensive use were assessed using the
Results
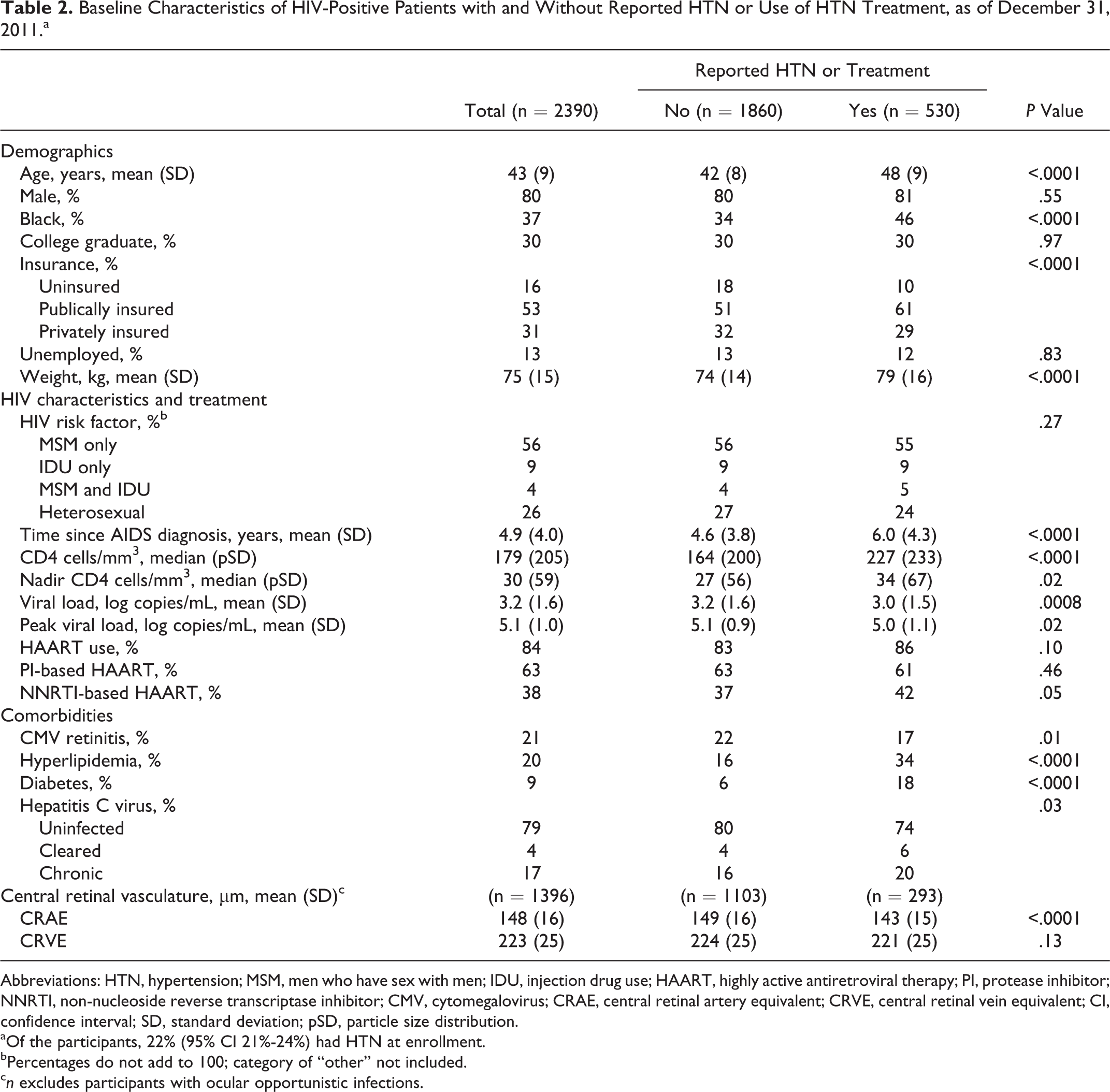
As of December 31, 2011, there were 2390 participants enrolled in LSOCA (Table 1). Overall, 80% of the participants were male, 45% white, non-Hispanic, 37% black, non-Hispanic, and 14% Hispanic. Approximately two-third of the participants were 40 to 59 years old. Among all the participants, 530 reported having high blood pressure requiring medication or reported using antihypertensive agents at enrollment, for a baseline HTN prevalence of 22% (95% CI 21%-24%, Table 2).
Characteristics of Overall Study Population, Longitudinal Study of the Ocular Complications of AIDS (LSOCA), as of December 31, 2011 (n = 2390).
Baseline Characteristics of HIV-Positive Patients with and Without Reported HTN or Use of HTN Treatment, as of December 31, 2011.a
Abbreviations: HTN, hypertension; MSM, men who have sex with men; IDU, injection drug use; HAART, highly active antiretroviral therapy; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; CMV, cytomegalovirus; CRAE, central retinal artery equivalent; CRVE, central retinal vein equivalent; CI, confidence interval; SD, standard deviation; pSD, particle size distribution.
aOf the participants, 22% (95% CI 21%-24%) had HTN at enrollment.
bPercentages do not add to 100; category of “other” not included.
c
Prevalent HTN
In adjusted analysis, prevalent HTN was associated with older age (odds ratio [OR] 2.05/10 years of age, 95% CI 1.77-2.37; Table 3), black race (OR 2.03, 95% CI 1.57-2.63), heavier weight (OR 1.21/10 kg, 95% CI 1.12-1.31), and lack of a college degree (OR for college graduation 0.74, 95% CI 0.57-0.98). Diabetes (OR 1.82, 95% CI 1.28-2.57) and hyperlipidemia (OR 2.18, 95% CI 1.66-2.87) were also significantly associated with HTN. Patients with prevalent HTN had longer times since AIDS diagnoses (OR 1.04/year, 95% CI 1.02-1.08) and higher CD4 counts (OR 1.09/100 cells/mm3, 95% CI 1.02-1.16). HIV viral load (baseline and peak), nadir CD4 counts, HAART, ARV class, hepatitis C coinfection, and CMV retinitis were not significantly associated.
Unadjusted and Adjusted Associations between Enrollment Characteristics and Prevalent Reported Hypertension or Use of Hypertensive Treatment at Baseline.a

Abbreviations: MSM, men who have sex with men; HAART, highly active antiretroviral therapy; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; CMV, cytomegalovirus; CI, confidence interval.
an = 2390 participants. There were 2104 complete cases. 12% of data with missing weights imputed with average weight (75 kg) and 14% of data with missing hepatitis C virus determinations imputed as uninfected.
Incident HTN
The median follow-up in this study was 6.5 years (0.1-13.3 years). Self-reported HTN or initiation of antihypertensive medication in the LSOCA cohort was 64.1 per 1000 person-years (PY, 95% CI 58.7/1000-69.9/1000 PY, Table 4). In the adjusted model for incident HTN among all participants, older age (hazard ratio [HR] 1.38/10 years, 95% CI 1.22-1.57, Table 5), black race (HR 1.42, 95% CI 1.15-1.75), and higher weight (HR 1.18/10 kg, 95% CI 1.10-1.27) were associated with an increased risk. Diabetes (HR 1.97, 95% CI 1.45-2.67) was also associated with incident HTN, whereas hyperlipidemia was not. Time since AIDS diagnosis, HIV viral load (baseline and peak), CD4 (baseline and nadir), HAART use and ARV class, CMV retinitis, and hepatitis C coinfection were not significantly associated with incident HTN in this model.
Unadjusted and Adjusted Associations between Enrollment Characteristics and Incident Reported Hypertension or Use of Hypertensive Treatment among Participants Without Ocular Opportunistic Infections.a

Abbreviations: MSM, men who have sex with men; HAART, highly active antiretroviral therapy; PI, protease inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; CMV, cytomegalovirus; CI, confidence interval; PY, person-years.
aHypertension incidence was 64.1/1000 PY (95% CI 58.7/1000-69.9/1000 PY). n = 1783 participants without baseline hypertension and with available follow-up data. All variables selected for multiple Cox regression model. There were 1576 complete cases. In all, 12% of the data with missing weights imputed with average weight (75 kg) and 15% of data with missing hepatitis C virus determinations imputed as uninfected.
Unadjusted and Adjusted Associations between Retinal Vasculature Measurements and Prevalent Reported Hypertension or Use of Hypertensive Treatment at Baseline, Participants Without Ocular Opportunistic Infections.a

Abbreviations: CI, confidence interval; HAART, highly active antiretroviral therapy; NNRTI, non-nucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
an = 1255 participants without ocular opportunistic infection and with available retinal vasculature measurements.
Secondary Analyses: Retinal Microvasculature
The majority (78%, Table 1) of the participants did not have OOI at enrollment. In unadjusted analysis of these participants, those with baseline HTN had smaller mean CRAE measurements compared to participants without HTN (OR 2.30, first versus fourth quartile, 95% CI 1.55-3.43; Table 5), which remained significant in adjusted analysis (OR 2.16 first versus fourth quartile, 95% CI 1.23-3.78). In adjusted analyses, incident HTN was also associated with smaller baseline CRAE (HR 2.87 first versus fourth quartile, 95% CI 1.86-4.42; Table 6). There were no significant associations between CRVE and either prevalent or incident HTN.
Unadjusted and Adjusted Associations between Retinal Vasculature Measurements and Incident Reported Hypertension or Use of Hypertensive Treatment at Baseline, Participants Without Ocular Opportunistic Infections.a

Abbreviations: CI, confidence interval; HAART, highly active antiretroviral therapy; NNRTI, non-nucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
an = 938 participants without ocular opportunistic infection or baseline hypertension and with available retinal vasculature measurements.
bAdjusted for demographics (age, sex, race, education, insurance status, employment status, and weight), HIV characteristics and treatment (HIV risk factor. Time since AIDS diagnosis, CD4: baseline and nadir, HIV viral load: baseline and peak, HAART, PI-based HAART, and NNRTI-based HAART), and comorbidities (hyperlipidemia, diabetes, and hepatitis C infection: cleared and chronic).
Discussion
In this large, prospective study of patients with AIDS attending AIDS ophthalmology centers in the era of HAART, self-reported HTN was present at enrollment in 22% of the patients. Factors associated with prevalent HTN were traditional HTN risk factors and metabolic comorbidities (diabetes and hyperlipidemia), in addition to longer time since AIDS diagnosis and higher CD4 count. Incident HTN among participants was also associated with traditional risk factors and diabetes. In secondary analysis, narrower CRAE was associated with both prevalent and incident HTN. These findings identify HTN as a significant chronic condition in patients with AIDS. Furthermore, risk factors for HTN in this population mirror those in the general population. The results of this study highlight the importance of screening for HTN among patients with AIDS.
The prevalence of HTN (22%) among LSOCA participants falls within the range identified in other studies of HIV-infected adults, and generally overlaps with prevalence in HIV-negative populations. 40, 41 To the authors' knowledge, only one other longitudinal study has described HTN incidence among HIV-infected adults, and no other analyses are exclusively in patients with AIDS. The Data Collection of Adverse Events of anti-HIV Drugs Study (D:A:D) reported HTN incidence of 72.1 per 1000 PY (95% CI: 68.2-76.0). 24 This rate is similar to that in LSOCA, which was 64 per 1000 PY (95% CI 58.7/1000-69.9/1000 PY), with overlapping CIs. The D:A:D study outcomes included measured blood pressure, compared to this largely self-reported outcome, which may, in part, explain the small variation in raw incidence rates between these 2 studies.
Traditional risk factors for HTN in HIV-negative adults are well established and include black race, hyperlipidemia, obesity, and age. 21, 42 –44 This study confirms that these factors are also associated with HTN in patients with AIDS. Numerous public health interventions target HTN prevention for the general population, and the overlap of HTN risk factors in patients with AIDS and HIV-negative patients suggests that these interventions are applicable to this population as well. 21, 45 In addition, HIV-related factors have been previously associated with HTN, including indicators of infection duration. 22 A variety of studies suggest the effects of chronic inflammation on endothelial function, the CV system, and potentially on CV risk in individuals with HIV, regardless of the viral suppression. 46 –48 Presumably, the longer the duration of HIV infection, the greater the risk of lasting endovascular damage that might result in HTN. The discrete mechanisms underlying these processes in HIV infection is an ongoing area of investigation, but their relationship to CV comorbidities remains important. 49 In this study, the authors also found an association between prevalent HTN and time since AIDS diagnosis. Time since AIDS diagnosis may be considered as a marker of overall infection duration and suggests that a potential HIV-specific mechanism for HTN in these participants might also be related to chronic inflammation. In addition, baseline CD4 counts were higher in participants with prevalent HTN. This finding might also suggest that participants with more functional immune systems and potentially greater inflammatory responses might experience long-term vascular effects of chronic inflammation. Finally, central retinal microvasculature has been identified as a predictor of incident HTN in the general population. 27, 29 –31 In this study, narrower CRAE measurements were also associated with incident HTN in HIV-positive patients without OOI. Central retinal arteriolar equivalent narrowing has been associated with CV risk and morbidity in certain groups of HIV-negative adults as well as with mortality in HIV-positive patients. 27, 32, 50 Further work might elucidate the mechanisms underlying this relationship in the context of exposure to prolonged inflammation.
Longitudinal analysis of the D:A:D study data demonstrated a decreased risk of incident HTN with cumulative exposure to NNRTIs. 24 In a smaller, cross-sectional study of patients attending a sexual health clinic in London, Wilson et al found an association between NNRTI use and increased blood pressure. 25 In this study, neither HAART nor NNRTI-based HAART use at enrollment were associated with either prevalent or incident HTN, further suggesting that HIV treatment may not, in and of itself, contribute to HTN.
Although these data were collected from a cohort with a primary aim of assessing ocular complications in patients with AIDS, since its inception, LSOCA has been designed to additionally assess the nonocular outcomes, including mortality and visceral CMV. Furthermore, LSOCA enrollment included participants both with (22%) and without (78%) a history of OOI, and the authors found no increased risk of prevalent or incident HTN in patients with CMV retinitis, the nearly exclusive form of OOI, compared to those without it.
Several additional limitations of this study merit comment. First, the authors do not have specific information on the degree to which reported diagnoses were additionally confirmed by study personnel via chart review and/or medical providers (as was possible given study protocol). A conservative approach, therefore, would be to assume that HTN was largely self-reported and therefore subject to recall bias.
Second, the final analysis did not exclude participants who were on antihypertensive agents, did not explicitly report a diagnosis of HTN but were determined to have another diagnosis for which an antihypertensive agent might be prescribed (for example β-blockers in patients with coronary artery disease). The authors did perform a sensitivity analysis using only reported HTN as the outcome, excluding reported antihypertensive medication use. In this analysis, baseline differences between patients with and without HTN were similar compared to the final analysis reported in the results of this study. The prevalence of a reported HTN diagnosis was 19% (versus 22% in the final composite diagnosis and medication outcome). Hypertension incidence rate using only reported HTN was lower (26/1000 PYs versus 64/1000 PYs), suggesting that some of the HTN incidence the authors report with the composite outcome may be due to antihypertensive medication use for other indications. Factors associated with prevalent and incident HTN remained largely the same, with the exception of borderline significant associations between prevalent HTN and baseline PI exposure (OR: 1.06, 95% CI 0.60-0.97) and incident reported diagnosis of HTN and shorter time since AIDS diagnosis (OR: 0.94, 95% CI 0.90-0.98), as well as lower peak HIV viral load (OR: 0.86, 95% CI: 0.76-0.98).
Finally, the authors did not have sufficient observations on smoking status or BMI to include them in these regression models. In adjusted analyses, however, the authors demonstrated an association between heavier weight and HTN.
Cardiovascular disease poses a comorbid burden to HIV-infected adults on HAART and likely results from a variety of factors, including aging, traditional risk factors, HIV itself, and medications. 13 –17, 51 Convincing evidence relates continuous, early initiation of HAART to better outcomes; therefore, the benefits of HAART outweigh the costs related to its role in comorbid disease development. 11–12, 19 Furthermore, effects of chronic inflammation may persist despite appropriate HIV control with HAART, resulting in ongoing CV disease risk. 49 Identifying components of CV risk and disease that are modifiable is therefore of increasing importance in patients with HIV and AIDS. Diagnosis and control of HTN in general have been demonstrated as essential to improving CV disease outcomes. 21 This study demonstrates that HTN is a prevalent comorbidity in HIV-positive patients in the era of HAART and suggests the value of screening for HTN as a potential means to decreasing CV risk in this population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Jabs has been a consultant to Abbott Pharmaceuticals, Alcon Laboratories, Allergan, Inc, Corcept Therapeutics, Genentech, Genzyme Corporation, GlaxoSmithKline, and Roche Pharmaceuticals, and serves on Data and Safety Monitoring Committees for Applied Genetic Technologies Corporation and Novartis Pharmaceutical Corporation. The remaining authors have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by cooperative agreements from the National Eye Institute to The Mount Sinai School of Medicine (U10 EY 08052), The Johns Hopkins University Bloomberg School of Public Health (U10 EY 08057), and the University of Wisconsin, Madison School of Medicine (U10 EY 08067).
