Abstract
The aim of this study was to compare the retention in medical care and antiretroviral (ARV) treatment of individuals living with HIV and AIDS to antiretroviral therapy in southern Brazil according to their “race” or skin color. This study is part of a 225-day prospective trial, comprising 7 interviews, in which an intervention designed to improve adherence to treatment was tested. A convenience sample of 73 individuals living with HIV and/or AIDS enrolled in this follow-up procedure. The mean length of continuance in treatment was 161.5 (standard deviation [SD] = 18.6; 95% confidence interval [CI] = 125-198) and 138.4 (SD = 14.1; 95% C.I. = 111-166) days in the “nonwhite” and “white” categories, respectively. There was no significant difference between the 2 categories, χ2(1, n = 72) = 0.76, P = .38, which include similar levels of retention in medical care and treatment with ARV medications between groups of individuals categorized as white and nonwhite in this sample.
Keywords
Introduction
The most recent figures on HIV and AIDS in Brazil, issued by the Joint United Nations Programme on HIV/AIDS, estimated that around 0.5% of the Brazilian population is infected by HIV. 1 Thus, considering that the Brazilian population is approximately 200 million, there are 1.3 million Brazilians living with this condition, although recent figures may show different results. In Brazil there is universal access to antiretroviral (ARV) drugs, and highly active antiretroviral therapy (HAART) has been publicly funded since 1996. 2 This treatment modality has transformed AIDS into a chronic infectious disease, making adherence the main challenge for reducing the rate of disease progression and mortality. 3,4
Adherence to complex medical regimens entails an interplay among patients (critical skills, incorporating the regimen into daily life), disease processes, treatment components (number of pills, side effects), medical care (reliability, establishing and maintaining a therapeutic alliance), social support, and outcome. 5,6 Research evidence indicates that absence of or limited adherence to treatment regimens is the main cause for the failure of HAART. 7 In addition, failure to comply with HAART may worsen patients’ health status 8 as well as increase drug resistance. 9 Despite the fact that adherence rates must be superior to 95% to produce effective HIV replication suppression, 10 in Brazil adherence to antiretroviral therapy (ART) has been estimated to be between 41% 11 and 56.9%. 12 The levels of viral resistance to HAART are still relatively low in Brazil (6.6%), when compared with the rates in the United States (15%-26%), Spain (26%), and United Kingdom (14%). 13 Several factors are known to reduce adherence to HAART, including psychological distress, 14 substance use, 15 and individual characteristics, such as young age, 12 limited income, 16 and low educational levels. 17
There is an increasing tendency to expand the levels of care to people living with HIV and AIDS (PLWHA) by transcending the concept of adherence to ARV medications to also focus on engagement in medical care as an important outcome. 18 One of the benefits of uninterrupted adherence to treatment relates to the preventive potential of sustained suppression of plasma viral load to curb the growth of the HIV epidemic. 19,20 Different measures of retention in treatment may have variable degrees of utility according to variable settings and circumstances. A study in which 6 different retention measures were evaluated further confirmed the fact that currently “there is no clear gold standard to measure retention in HIV care.” 21 Although patients’ retention in care has been conceptualized and assessed in different ways, the measures based on missed appointments remain the most commonly used parameter. 22
In Brazil, the variable “race” or color of skin started to be recorded in the national databases related to HIV and AIDS in 1996. 23 However, the interpretation of results of race and ethnicity within the context of HIV and AIDS has been considered problematic, given that, by 2000, less than 4% of the cases had been classified according to race. 23 Furthermore, there were inconsistencies between the self-classification and heteroclassification systems. 23 Although doctors and other health care professionals used the heteroclassification system, the prevalence rates of HIV and AIDS were established on the basis of self-classification. 24–26
It has been well documented that the AIDS epidemic has expanded in Brazil to mainly affect the poorest and least educated segments of the population, especially the women and the unemployed. 27 –29 Although there is a higher proportion of black and “pardo,” or brown, which are overrepresented categories among the poorest segments of the population, recent epidemiologic data do not support the claim that there is a significant association between the “black population” and AIDS in Brazil. 23 Yet, the putative “racial” differences in the context of HIV and AIDS have not been investigated formally in this country, particularly the rates of adherence to ART.
The objective of this study is to compare, prospectively, the rates of adherence to ARV medications between groups of white and nonwhite individuals living with HIV and AIDS in Southern Brazil.
Methods
Sample
In this study, the authors evaluated a convenience sample of 73 individuals living with HIV and/or AIDS who were prescribed ARV medications. Research participants were recruited from the outpatient and the inpatient units associated with the Department of Infectious Diseases within the medical campus and teaching hospital of the University of Caxias do Sul (UCS) in southern Brazil.
The inclusion criteria included (1) being older than 18 years of age, (2) being enrolled in clinical follow-up for HIV and AIDS in the health care center associated with this study, and (3) having the ability to understand the aim of the study as well as the content of the questions in all questionnaires used in this study. The HIV ambulatory clinic associated with this study is a major regional public health care provider within the Brazilian universal health system (Sistema único de Saúde [SUS]). Exclusion criteria included detainment in correctional facilities, residence in cities other than the municipality where this study was conducted (Caxias do Sul), and incapacity to properly complete the questionnaires and forms as required in this study due to cognitive impairment.
Informed Consent and Recruitment Process
This study was granted approval by the UCS Human Research and Ethics Committees. Treatment-seeking individuals attending the inpatient and outpatient facilities mentioned previously were verbally invited to participate in this study. Prospective participants were invited to take part in the study irrespective of their socioeconomic background, level of educational attainment, health insurance coverage statuses, or any other specific personal characteristic.
Volunteers were provided with a written protocol pertaining to the study and a verbal explanation about the purposes of this investigation. They were also informed that participation was voluntary, confidential, and anonymous. Volunteers were also told that they could withdraw from this study at any time without any repercussion to their treatments. Upon their agreement to participate in this study, research volunteers were then asked to sign an informed consent document. All recruitment and research procedures were conducted in Portuguese. Research participants were compensated for each attendance to research interviews with a food staple basket (Cesta Básica Nacional) according to official regional parameters established by the Departamento Intersindical de Estatística e Estudos Sócio-Econômicos in Brazil.
Study Design
The current study is part of a prospective trial in which participants were enrolled in a longitudinal follow-up procedure of 225 days in which 7 interviews were programmed to take place in order to test an intervention designed to improve adherence to ART. 30 The assessments were conducted during an initial interview (time zero) and then subsequently after 15, 45, 75, 105, 135, and 225 days. In this study, the authors compared the rates of retention in medical care and ARTs for research participants categorized according to the Brazilian Institute of Geography and Statistics (IBGE) racial classification system. 31
In this study patients retained in treatment were considered as those who attended all scheduled appointments. Only scheduled visits to the UCS HIV ambulatory clinic were considered as regular attendance medical health care for the purpose of retention in medical care. Other procedures to obtain care, including attendance at undesignated walk-in services, urgent care, or supportive services were not considered as a formal attendance in the trial for the purpose of retention in care.
The original trial was registered in 2 research trial registries as follows: the Latin American Ongoing Clinical Trial Register (#BRA89) and the University Hospital Medical Information Network (#UMIN000001092).
Data Collection Procedures
All research and assessment forms were completed within a pen-and-paper format. Immediately after termination of the interview session, research documents were conveyed to the research central office. Research data were transcribed into an electronic databank by a team of research assistants who revised the data storage procedures for accuracy and formatting purposes.
Demographic data collected in this study included age, sex, marital or relationship status, educational level, and employment status, as well as residential and living arrangements. Skin color was classified according to parameters of the IBGE. Socioeconomic status was measured according to the Critério de Classificação Econômica Brasil that takes into consideration familial monthly average income. 32 Additional variables of interest were also assessed via different research tools.
The IBGE is the governmental organization responsible for formulating and collecting censuses in Brazil. The Brazilian census uses the term cor (literally “color”) rather than raça (race). Although censuses have been conducted for over a century in Brazil, the official racial categories have rarely been changed. Since 1950, IBGE has used the categories Branco (white), Pardo (brown), Preto (black), and Amarelo (yellow). In 1991, the category Indígena (indigenous) was added. 33
Interview Process
A team of investigators was trained to independently interview research participants. Researchers had different academic backgrounds in the health and medical sciences and were all well versed on issues related to HIV biology and transmission, medication adherence, and research ethics. The research team was supervised by senior investigators during regular meetings in order to clarify pertinent issues and to ensure the study was being conducted according to guidelines outlined in the research protocol. In order to keep a potential in-group bias in check, 34 researchers also self-attributed their racial categories that were consensually endorsed by other members of the research group during a team meeting.
Assessment sessions included both interview-administered questions and self-report questionnaires. Particular care was taken with those with limited educational background in order to ensure satisfactory understanding of the research questions. Interviews took place in a suitable room at the outpatient clinic building following routine medical appointments coordinated by the department of infectious diseases.
In all non-MATI (Medication Adherence Training Instrument) interviews, the researchers followed a predetermined list of topics to be addressed during medical reviews. Participants allocated to the MATI group reviewed adherence to treatment as part of the structured MATI interview in all follow-up interviews. The measurement of retention in medical care and treatment was based on sustained attendance to clinical appointments for both the groups, as the trial progressed from enrollment to the final interview of the 225-day trial after enrollment. These parameters were confirmed further by measures of adherence mentioned below.
Adherence to Treatment
The topics included overall physical and mental status, the presence of adverse reactions to medications, and adherence to treatment. Participants allocated to the MATI group reviewed adherence to treatment as part of the structured MATI interview in all follow-up interviews. The measurement of retention or survival in the treatment was based on sustained attendance to clinical appointments for both the groups as the trial progressed from enrollment to the final interview of the 225-day trial after enrollment.
In addition, 3 other modalities of assessment were used to confirm adherence to treatment in this study. The AIDS Clinical Trials Group (ACTG) adherence questionnaire—baseline and follow-up versions—was also used in this trial. The ACTG has been used extensively in HIV research trials. 18 The 5-item follow-up version was used to assess adherence during interviews that took place 105 and 225 days after enrollment in the trial. Adherence to treatment was also assessed via the Simplified Medication Adherence Questionnaire (SMAQ). This 6-item instrument has been validated for the assessment of adherence to HAART in clinical settings. 21 The SMAQ proved to be effective when used alone and in combination with indirect methods also in the context of other medial specialties. 35 The SMAQ was used to assess adherence during interviews talking place 105 days after enrollment of participants in the trial.
Finally, the clinical status of research participants was also evaluated via blood tests including viral load and CD4 count every 4 months. A closer review of one’s adherence to treatment was recommended in the context of the 3 clinical events: (1) viral load >400 or 50 copies/mL after 24 and 48 weeks of HAART, respectively; (2) confirmation of a reduction of at least 25% in CD4 count; and (3) occurrence of significant adverse clinical events including onset or recurrence of infectious diseases.
Statistical Analysis
Measures of central distribution and dispersion of data were calculated for all demographic characteristics. Cohen κ coefficients were calculated to determine interannotator agreement for racial categories. A Kaplan-Meier survival analysis was conducted to evaluate the null hypothesis that in this population, the 2 survival functions, based on the white and nonwhite categories according to the IBGE, were equal. One participant did not complete the questions related to race or color of the skin (n = 72). A similar survival analysis was conducted taking into consideration 2 categories of educational background (n = 73), namely, those with less than 8 years of formal education, which corresponds to primary school, and those with more than 8 years of education, including secondary and tertiary institutions. For the sake of the survival analyses mentioned previously, research participants who completed the study, those who were deceased during follow-up, and those who did not turn up for their appointments due to other reasons (incarceration during the trial, move to a different city, etc) were considered as censored data. Participants who withdrew from the study and those who lost contact or were untraceable were considered as uncensored data (events of noncompliance). A Mantel-Cox Log-Rank test was used to test the null hypothesis that the 2 survival functions in this population are equal.
Considering that a significant fraction of the sample chose not to complete or completed partially or improperly the scales of socioeconomic status (SES), instead of a survival analysis for this variable, a chi-square (χ 2 ) test was conducted to compare those who completed the trial—attended the final follow-up appointment and completed the final set of questionnaires. A series of χ 2 analyses were used to explore differences in sex, SES status, and educational background of the participants classified as white and nonwhite, whereas a Mann-Whitney U test was used in regard to age range. All statistical analyses were conducted using PASW Statistics, version 18.0.
Results
Sociodemographic Data
Table 1 presents the main sociodemographic characteristics of the sample. The results revealed that the typical patient in this sample was a white man, in his late 30s, from a low socioeconomic stratum, who completed primary schooling. There was no difference in terms of distribution of participants according to self-classification to different racial categories (P = .45). Similarly, no significant differences in terms of educational background (P = .18), socioeconomic standards (P = .31), and sex (P = .20) were observed.
Sociodemographic Characteristics according to the Race or Color of Skin (n = 58).
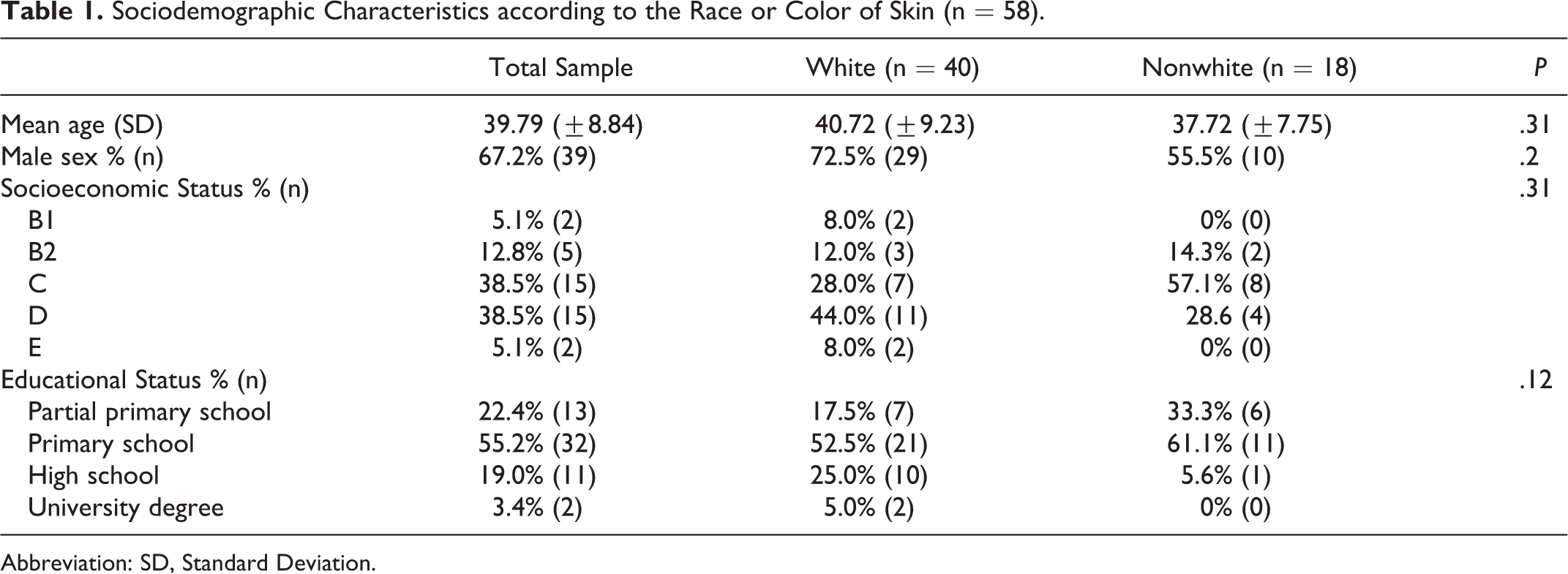
Abbreviation: SD, Standard Deviation.

The Kaplan-Meier survival plot depicts the continuance in treatment of research participants classified as white and nonwhite.
Racial Classification Methodology
In 15 cases, there was no information on color of skin according to self-classification or heteroclassification. In addition, only 1 volunteer self-classified as yellow. Cohen κ coefficients were therefore calculated with 57 research participants who had commensurate response patterns. A κ coefficient of 0.79 was observed for the 2 methods of classification mentioned previously, which indicate a high accuracy in prediction.
A 2-way contingency table analysis was conducted to investigate whether the interviewer’s color of skin may have influenced significantly the rates of disagreement between racial self- and heteroclassification by the research participants. The interviewer’s color of skin and the rate of disagreement between the 2 racial classification methods were found to have no significant correlation, with Pearson χ 2 (2, 57) = 0.59, P = .74, Cramér V = 0.1.
Survival in Treatment
Color of skin
The plot in Figure 1 demonstrates the continuance in treatment of participants who self-classified as white and those aggregated in the category Others, representing all nonwhite racial alternatives. The mean length of continuance in treatment was 138.4 (standard deviation [SD] = 14.1; 95% confidence interval [CI] = 111-166) and 161.5 (SD = 18.6; 95% C.I. = 125-198) days in the white and nonwhite categories, respectively. A Mantel-Cox log-rank test comparing both the groups according to the number of days remained in treatment revealed that there was no significant difference between the 2 groups, χ2 (1, n = 72) = 0.76, P = .38.
There were no differences in continuance of treatment when all the 4 categories (white, black, brown, and others) were compared separately as revealed by a Mantel-Cox log-rank test comparing all 4 groups according to the number of days remained in treatment, χ2 (1, n = 72) = 0.99, P = .8.
Educational Background
The mean length of continuance in treatment was 149.2 (SD = 20.7; 95% CI = 108-189) and 145.8 (SD = 13.5; 95% CI = 119-172) days in the less than 8 years of education (up to complete primary school education) and more than 8 years of education (at least any level of secondary education and including tertiary education). A Mantel-Cox log-rank test comparing both groups according to the number of days remained in treatment revealed that there was no significant difference between the 2 categories, χ2 (1, n = 73) = 0.02, P = .87.
Socioeconomic Status
Forty (of 73) research participants completed the scales related to SES . Of these, 23 have reached the final stage of the trial (completers who fulfilled questionnaires of the 225th day of follow-up), of whom 17 were classified in the lower SES classes (corresponding to C, D, and E), and 6 in the higher SES classes (A and B). There was no significant difference between these 2 groups (P = .4).
Comparison between Trial Completers and Noncompleters
There was no significant correlation between educational background and permanence in the study until the 225th day after the baseline assessment when those who completed at least on questionnaire (completers, n = 23) and those who left the trial prior to its completion (noncompleters, n = 50) were compared, with Pearson χ2 (3, 73) = 5.75, P = .11. Similarly, there was no significant difference between completers and noncompleters when compared by means of SES (P = .6) and color of skin (P = .72).
Discussion
The demographic characteristics of the sample evaluated in this study are generally similar to the demographic composition of the local population as reported in the Brazilian 2010 Census, 36 in that there are more white than nonwhite residents in Caxias do Sul, Brazil.
The results of this study also revealed significant agreement between self-classification and heteroclassification methods. This consistency persisted even when the interviewer and interviewee were not classified within the same racial category. This significant correspondence suggests that the racial affiliation of the observer does not interfere (absence of in-group bias) with the classification of research participants according to the IBGE classification system based on color of skin. In 1995, a survey conducted by Datafolha revealed discrepancies between self- and heteroclassification methods of race in Brazil, with a trend toward a greater assimilation into the whiter categories of the skin color spectrum. 37 However, results from a similar survey conducted in 2008 revealed a progressive increment in self-identification as brown (or pardo in Portuguese) and black. 38 It is possible that recent affirmative action policies as well as media campaigns to confront racism and to promote black pride in Brazil may have influenced progressive changes in Brazilian society. 39
The results of this study reveal no difference in retention in medical care and ART, as represented by medical follow-up review procedures, between white and nonwhite research participants. Therefore, racial affiliation per se does not explain poorer levels of retention in medical care and ARV treatment in this sample. Furthermore, the lack of significant difference in continuation or survival in treatment among different racial groups persevered when different racial groups were analyzed separately. These findings are substantiated further by the fact that there were no significant differences between completers and noncompleters (withdrawal bias) in terms of educational background, SES, and color of skin.
Only a limited number of studies conducted in Brazil have analyzed the association between racial categories and retention in ARV treatment. A cross-sectional study which compared 35 participants who were considered nonadherent with 70 age-matched adherent individuals revealed that those who self-classified as white and black were significantly overrepresented in the nonadherent group, whereas there were significantly more brown participants in the adherent group. 40 The interplay between adherence to treatment and racial categories in Brazil has also been investigated in studies from different medical specialties. A cross-sectional study developed in Salvador in which 200 hypertensive outpatients from low SES were interviewed and had their blood pressure measured (double-blind) revealed that compliance with medical recommendations was significantly superior among white (53.6%) when compared with black (19.7%) Brazilians. 41 A prospective, randomized controlled trial conducted in Sao Paulo to evaluate rates of adherence of patients with heart failure to a disease management program involving health education and telephone monitoring revealed a lack of significant difference between blacks and whites, in that the proposed intervention was equally successful for both the groups. 42
The results of this study are therefore in agreement with previous investigations on the association between color of skin and adherence to treatment in Brazil. However, there are significant regional variations in the demographic makeup of the Brazilian population. The highest proportion of nonblack populations in Brazil is concentrated in the southernmost part of the country where this study was conducted. 36 Therefore, the comparison of studies involving racial categories among different regions in Brazil may be prone to inconsistencies. In addition, the results revealed in this prospective study may not be comparable with data obtained from cross-sectional investigations.
It has been well documented that low levels of educational attainment may predict limited adherence to HAART and consequently poor prognosis in the context of HIV and AIDS. 17,40,43 –46 In this study, patients who received formal education for a longer period of time (more than 8 years of formal education) did not remain in follow-up treatment longer than the less formally educated research participants. In the same way, levels of formal education (primary/secondary versus high school/college) did not influence the rates of adherence to ART in a randomized clinical trial conducted to test an intervention aimed at increasing compliance to treatment. 47 In fact, the percentage of research participants in the current study with more than 8 years of formal education is lower than that for the sample investigated in the trial just mentioned (22.4% versus 52.3%). 47
Considering that only 54.8% of the research participants completed the SES questionnaires, inferences about the association between adherence to ARV medications and SES in this sample cannot be made with sufficient accuracy. Yet, evidence from several studies developed in Brazil indicate that adherence to ARV medication is significantly associated with family income 40 and employment status. 40,48
The lack of a significant difference among research participants according to their skin color appears to suggest that this variable was possibly not a major determinant in compliance. Consistently, the results of one of the few studies investigating this aspect in Brazil indicated that race was not a determining factor to explain poor adherence to ART. 45
In addition to shortcomings mentioned previously, the authors acknowledge potential limitations in this study. Considering that convenience sampling was used, generalizations to a wider population may not necessarily prove to be scientifically valid. However, given that the public health care facility in which this study was conducted provides similar standards of care and treatment to the only other HIV and AIDS treatment center in this region, it is plausible to infer that this particular convenience sample should not respond or behave differently than a random sample from the same population. Finally, another possibility to explain the results may be related to the limited sample size in this study. Although our study was initially powered to attain a larger sample size, a series of contextual obstacles may have hindered the enrollment of the number of participants needed to guarantee an ideal sample. In addition to an enlargement of sample size, increasing the number of observed outcome events also reduced the probability of a type II error. 49 Therefore, despite the efforts put forward to guarantee an optimal execution of this pioneering endeavor in this region of Brazil, the authors cannot exclude the possibility of a β-type error to account for the negative nature of the findings reported in this study.
The follow-up period in this study involved 7 medical appointments during the course of 225 days. It has been stated that “even in the very best designed and conducted studies,” it is unusual to conclude a study with a data set fully usable for every participant, apart from the fact that follow-up studies are always exposed to some degree of attrition. 50 On the other hand, the fact that both established clinic patients and patients being started on specialized HIV medical care were recruited represents a strength of this study. Evidence obtained from studies conducted solely with patients with regular attendance may have limited validity due to the fact that established patients may be more likely to be adherent to ART irrespective of their retention status. 21
Finally, there are remarkable worldwide differences in terms of health care coverage for PLWHA and as such “being linked to care is not enough.” 51 In the United States HIV ambulatory clinics, follow-up care may be given by multiple providers and “whether or not a patient is seen in the same clinic may not be important.” 22 In Brazil, a 3-level—federal, state, and municipal—public approach is provided as part of the SUS public health system. 35 In Brazil, patients are usually followed-up in designated HIV ambulatory clinics by a regular team of health care providers. Therefore, measures of adherence to treatment and retention in medical care can vary significantly across the different nations.
In Brazil, free access to HAART is guaranteed to all PLWHA in need of treatment since 1996, despite the rising cost of AIDS treatment. Brazil’s public health program on HIV and AIDS has led to sustained improvement in a series of health outcomes. This situation is in contrast to the provision of health care in the United States, for instance, where still “too many people living with HIV do not have access to the medical care that they need.” 51 Given the singular status of the public health program for PLWHA in Brazil, research evidence obtained in this country may not be readily generalizable to other economies. 52
Conclusion
The results of this prospective study suggest that color of skin, as measured by the IBGE criteria, and educational background are not associated with retention in medical care and treatment with ARV drugs in this specific sample. Additional prospective trials on HIV and AIDS are required in Brazil in order to determine, in a more definite manner, the predominant factors that may influence adherence to ART in Brazil.
Footnotes
Authors’ Note
This study was conducted at the University of Caxias do Sul Caxias do Sul, Brazil.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Grant CSV 302/06 of the National Program on Sexually-Transmitted Diseases and AIDS (Brazil).
