Abstract
In the context of emerging evidence related to preexposure prophylaxis and HIV treatment as prevention, an evidence summit was held in mid-2012 to discuss the current state of the science and to provide a platform for consensus building around whether and how these prevention strategies might be implemented globally. Health care providers, researchers, policy makers, people living with HIV/AIDS, and representatives of government authorities, donor agencies, pharmaceutical companies, advocacy organizations, and professional associations attended from 52 countries. An international advisory committee was convened to identify key messages and recommendations based upon the data presented and discussed at the summit. The advisory committee further worked to develop this consensus statement meant to assist relevant stakeholders in taking stock and mapping out a route forward to enhance the HIV prevention armamentarium.
Background
The development of effective antiretroviral therapy (ART) and the expansion of access to this life-saving and enhancing medical interventions across the world have been critical successes in HIV treatment. Since 1996, ART has added 14 million years of life in low- and middle-income countries alone; more than 9 million of those are in sub-Saharan Africa. 1
In contrast, despite the diligent work advanced by many, prevention efforts have experienced slower progress. Although HIV incidence has declined globally, it remains at unacceptably high levels in many developed and developing world countries. Biobehavioral prevention (biomedical interventions that require ongoing self-administration to be effective)—both treatment as prevention (TasP; the provision to and use of ART by HIV-infected individuals to reduce morbidity and mortality as well as the risk of onward HIV transmission through durable viral suppression) and preexposure prophylaxis (PrEP; the use of oral ART or topical microbicides by HIV-uninfected individuals before and after potential sexual exposure to HIV to reduce the risk of acquisition of the virus)—now offers new options for preventing HIV infection and the potential to end the HIV pandemic. Over the past year, TasP and PrEP have reached important milestones.
Treatment as Prevention
HTPN 052, a study of 1763 serodiscordant (also referred to as serodifferent) couples in which the HIV-infected partner had a CD4 count of 350 to 550 cells/mm3, was stopped early when the independent data and safety monitoring board saw a 96% reduction of transmission from the HIV-infected partner to the uninfected partner in couples who initiated ART when they entered the study. 2 On the basis of this result, Science selected TasP as the scientific breakthrough of the year for 2011. 3 In April 2012, the World Health Organization published guidance on testing and counseling of HIV serodiscordant couples which includes the use of ART to reduce the risk of transmission. 4 Earlier epidemiological studies suggested that expanded ART coverage played a role in decreasing HIV infection rates in San Francisco, British Columbia, and Denmark, 5–7 as well as mathematical models 8–11 support the theory that expanding access to TasP to the community level will decrease the HIV incidence. Although expanding access to treatment has clearly had significant impact, ongoing and planned research will further examine the impact of earlier initiation of ART on HIV incidence as part of a combination prevention strategy, taking into account the diversity of the HIV epidemic in different parts of the world. Despite the promising findings from HPTN 052 and other studies, initiation of ART upon diagnosis with HIV irrespective of CD4 count for those who want to initiate treatment will require resources and political will.
Preexposure Prophylaxis
The iPrEx study in men who have sex with men (MSM) provided with daily oral emitricitabine-tenofovir (FTC-TDF) reported a 44% reduction (95% confidence interval [CI]: 15%-63%) in new HIV infections; the result in participants with detectable drug levels in plasma was a 92% reduction (95% CI: 40%-99%). 12 After these results were released, the US Centers for Disease Control and Prevention issued interim guidance on FTC-TDF use for PrEP in high-risk MSM. 13 The partners PrEP study of serodiscordant couples showed a 75% reduction (95% CI: 55%-87%) in new HIV infections with daily oral FTC-TDF and 67% (95% CI: 44%-81%) with TDF, again with better results (90% for FTC-TDF and 86% for TDF) in patients with detectable drug levels. 14 On July 16, 2012, the US Food and Drug Administration (FDA) approved FTC-TDF for PrEP for HIV-uninfected MSM, HIV-uninfected partners in serodiscordant couples, and other individuals at risk of acquiring HIV through sexual activity. Two other PrEP studies had equivocal results, perhaps due to difficulties with adherence. 15–17 Research continues with studies of alternatives to daily dosing (eg, twice weekly and sex-dependent regimens); studies of different antiretroviral (ARV) agents (including nonnucleoside reverse transcriptase inhibitors such as dapivirine, entry inhibitors such as maraviroc, and integrase inhibitors); and studies of non-oral PrEP technologies such as vaginal and rectal microbicides, intravaginal rings, and injectable agents.
The IAPAC Evidence Summit
In the context of this important emerging evidence, the International Association of Physicians in AIDS Care (IAPAC), now known as the International Association of Providers of AIDS Care, same acronym, hosted an evidence summit June 11-12, 2012, in London to discuss the current state of TasP and PrEP science and to provide a platform for consensus building around whether and how these novel prevention strategies might be introduced globally. Cohosted in partnership with the British HIV Association (BHIVA), the summit was organized as 1.5 days discussing TasP and a half day on PrEP. Health care providers, researchers, policy makers, people living with HIV/AIDS (PLWHA), and representatives of government authorities, donor agencies, pharmaceutical companies, advocacy organizations, and professional associations attended from 52 countries. Slide presentations from the summit are available on both the IAPAC and BHIVA Web sites (www.iapac.org and www.bhiva.org).
The summit organizers used an audience response system (ARS) to collect responses to a set of survey questions asked to the summit attendees at the conclusion of the TasP and PrEP sections of the program. Responses were tabulated using the system’s polling software. This process was an effort to take the pulse of a heterogeneous group of experts closely involved in HIV prevention, care, treatment, research, and advocacy on areas of consensus or disagreement as well as those areas requiring additional research. Subsequently, IAPAC convened an international advisory committee to identify key messages and recommendations based upon the data presented and discussed at the summit as well as the ARS-facilitated survey results. The advisory committee further worked to develop a consensus statement meant to assist relevant stakeholders in taking stock and mapping out a route forward to enhance our HIV prevention armamentarium.
Treatment as Prevention
Treatment as prevention is at a crossroads similar to that witnessed at the 1996 advent of highly active ART (HAART), when the results of only 2 early-phase HAART trials were known, yet questions remained unanswered about its effectiveness and the resources required for its wide-scale implementation, especially in resource-limited settings and challenges to ensuring equitable access. Early adoption of HAART transformed an HIV diagnosis from “death sentence” to a manageable chronic condition, saving and enhancing the lives of countless PLWHA. The use of HAART for TasP should move forward so that a similar impact on prevention might occur.
Given the clear-cut results from HPTN 052 and the corroborative observational and ecological studies, no further trials are considered necessary to demonstrate TasP’s efficacy. Further studies are needed, however, to assess the effectiveness and cost-effectiveness of immediate initiation of HAART in different populations and settings. To address these questions, several studies have started or will start shortly in Botswana, South Africa, Tanzania, Uganda, and Zambia to evaluate the effectiveness of TasP as part of combination prevention strategies that also include the following essential services:
Universal voluntary HIV testing and counseling
Linkage to care and treatment support for those testing HIV positive
Male medical circumcision for HIV-negative men
Counseling and condom provision
Prevention of mother-to-child transmission with ART or ARV drugs
Treatment of sexually transmitted infections
During the summit, the question was raised of whether all these elements are needed to effectively control HIV transmission. Following proof of concept for sustained HIV control within communities in which combination prevention strategies are implemented, further research may be warranted to investigate implementation approaches that maximize the efficiency and minimize the cost of these interventions.
Mathematical models of the effect of universal testing and treatment on HIV incidence also support the use of TasP. Although the models differ in their assumptions and thus their outcomes, a significant consensus exists among them that TasP could significantly reduce HIV incidence. Figure 1 shows the potential effect of universal HIV testing on HIV incidence and TasP projected by one model. 8
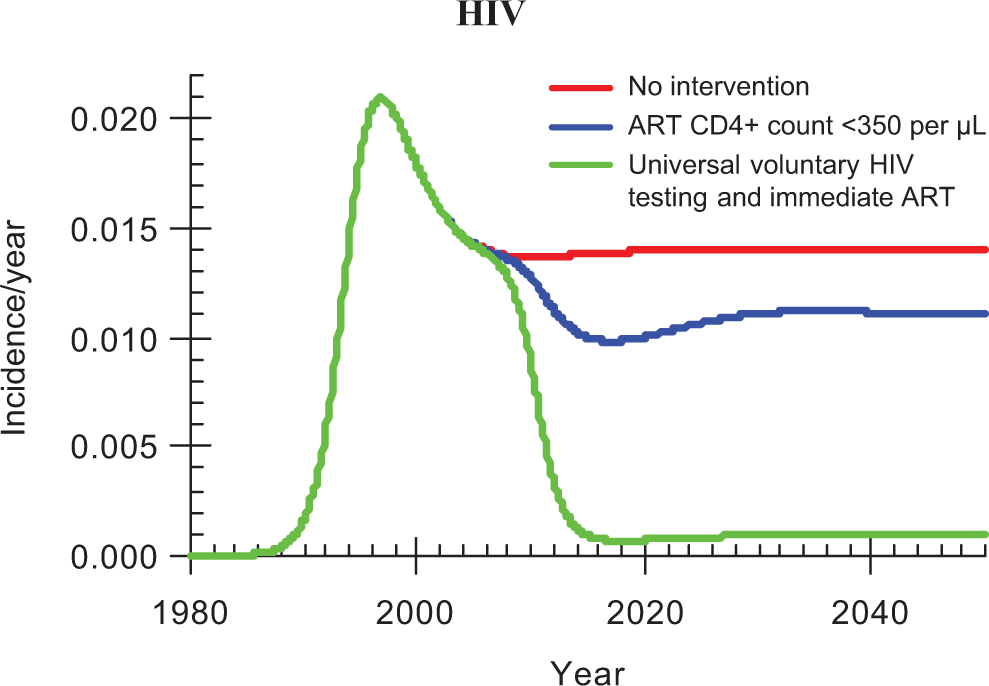
Deterministic transmission model. Reprinted with permission from Granich et al.8
A panel of pharmaceutical industry executives considered the cost and sustainability of medications currently available for TasP. They posed questions around whether current ARV agents are sufficient for TasP or whether new ones are needed to address cost, convenience, and tolerability concerns. Also discussed was how to scale-up access to TasP in resource-limited countries.
TasP should not present a conflict between individual health and public health, since data suggest that early initiation of HAART benefits PLWHA. Some feel, however, that the data for individuals with CD4 counts greater than 500 cells/mm3 are not sufficiently robust; the START study continues in order to address this question. 18 On the other hand, national guidance in many developed countries increasingly recommends initiation of treatment when individuals are first found to be HIV infected. Treatment can provide significant benefit to both HIV-infected and uninfected individuals but not without a great deal more work and careful communication in the coming years. Implementation will require:
Greater community education about the benefits of early ART
Operational research to establish TasP’s acceptability and feasibility in diverse settings
Clinician and allied health professional education
Anticipation of higher demand for services
Adaptation of treatment discussions to focus on patient health and choice and prevention
Public acceptance of the right of individuals to seek earlier treatment and/or forgo treatment
Access to treatment for all populations
Current inadequate levels of HIV diagnosis and linkage to and retention in care of HIV-infected individuals are the primary threat to TasP’s ability to significantly reduce HIV incidence. Figure 2 illustrates gaps in the continuum of HIV care in the United States that exemplify this threat. 19

Number of HIV-infected people engaged in selected stages of the continuum of HIV care in the United States. Reprinted with permission from Gardner et al.19
Addressing and narrowing these gaps across each aspect of the continuum is essential to achieving the goals of TasP. Reconceptualizing treatment as essential for staying healthy rather than an intervention that is used only for people who are severely ill may help address the need for earlier diagnosis as well as access to and retention on treatment. Of note, the magnitude of this challenge may be very different in resourced versus resource-limited settings.
Although the benefits in terms of individual and public health are clear, introduction of earlier access to ART presents a number of other challenges, particularly in resource-limited settings:
Initial funding and financial sustainability, including competition for limited ART resources with groups already defined as clinically eligible for and thus requiring ART
Global availability of ART regimens that are less toxic, better tolerated, and more convenient to use
Human rights and ethical issues, including the tension between patient choice and clinician responsibility, potential for coercion, and the need to ensure equity of access
Stigma related to HIV testing and against individuals who choose to forgo HIV treatment once diagnosed
Initial overload of health care systems
Training and continuing education of the health workforce
Changes to testing and counseling programs
Suboptimal adherence leading to virologic failure and resistance; it was noted that recently published guidelines for improving entry into and retention in care and ART adherence may be of particular use to mitigate this challenge 20
Potential for behavioral changes in current prevention practices that may offset risk-reduction benefits in the context of overestimated protection against HIV
The question remains whether sufficient ART coverage can be achieved to significantly curb the HIV pandemic. This challenge will be particularly acute in settings where ART coverage for HIV-infected patients with CD4 counts less than 350 cells/mm3 continues to be a large unmet need.
A novel approach to measure ART coverage and evaluation of the impact of TasP on the national and global levels was proposed. The concept of individual viral load suppression as a key factor for a healthy and long life for an HIV-infected individual has been long established. Data from HPTN 052 and other earlier studies, as well as ecological data, indicate that suppressed individual viral load also leads to decreased transmission among couples. The concept of community viral load has emerged as a way to monitor access to HIV testing and treatment. 5 , 21 The concepts of national viral load and global viral load were introduced at the summit as potentially useful tools to guide the decisions of policy makers and funders at these levels. This approach shows a much larger coverage gap than merely measuring ART provision does. The measurement of community viral load—and thus of national or global viral load—also has limitations since in many settings the individuals who are most likely to transmit HIV may not know they are infected and so are not engaged in care or using ART. Thus, estimation of viral load is only as accurate as a community’s or a nation’s ability to identify all HIV-infected individuals. Another key indicator to consider is the national or local proportion of people estimated to have HIV who are virally suppressed. Nonetheless, these measurements may prove useful in demonstrating increased ART coverage and viral suppression.
Summit Attendee Response
Summit attendees largely reported a high (54%) or moderate (41%) level of confidence in the scientific support for TasP. Most attendees felt that the efficacy and safety research supported immediate implementation of TasP; only 16% said that additional research was required before implementation should proceed. Just over three-fourths of the attendees said that TasP should be offered to all patients. In contrast, just over half said medical providers in their countries might be unsure about TasP and require more data; only 40% reported that providers were in agreement with TasP and 9% reported that providers were opposed to TasP. Resources and funding for TasP were a concern for many attendees. Attendees indicated that a recommendation to offer TasP to all HIV-positive individuals would result in a significantly higher proportion of newly diagnosed patients on ART; 35% of the attendees said that the increase would be 50% or more. Not surprisingly, given these increases, attendees worried about the resource constraints posed by TasP. More than half of attendees cited funding as the greatest barrier to implementation of TasP; stakeholder acceptance and workforce shortages ranked a distant second and third.
Over half (54%) of the attendees stated that TasP would limit resources for ART as currently indicated.
Key Messages
The paradigm for use of ART has shifted; treatment and prevention have converged.
The evidence for TasP’s efficacy justifies its use in patients who wish to start ART early.
Further research is needed to investigate the effectiveness and cost-effectiveness of TasP, its feasibility and acceptability on a community and national level, and its role as one component of a comprehensive prevention strategy.
Successful TasP will require higher levels of HIV testing, enhanced linkage to and retention in care, access to quality treatment, adherence support, and new ways to monitor coverage and impact.
There are numerous challenges to implementation of TasP including financial and resource limitations, quality and appropriateness of available drugs, ethical and human rights issues, stigma, health system and workforce capacity, and potential increases in risk from suboptimal adherence and/or risk compensation behavior.
Preexposure Prophylaxis
The proof of concept of daily oral PrEP efficacy has been achieved, and research should move on to examining questions of effectiveness. Figure 3 shows the effect size seen in various oral PrEP studies in the context of other HIV prevention technologies. 22

HIV prevention technologies shown to be effective in reducing HIV incidence in randomized clinical trials. Reprinted with permission from Abdool Karim and Abdool Karim.22
It should be noted, however, that although there have been 4 successful trials supporting the efficacy of oral and topical TDF-based compounds, 12 , 14 , 23 , 24 the FEM-PrEP study of oral FTC-TDF and the TDF tablet, TDF gel, and placebo gel arms of the VOICE study were discontinued early because of lack of efficacy. 22–24 Suboptimal adherence clearly played a role in the failure of these interventions to demonstrate efficacy but there may have been other factors as well.
Figure 4 shows a potential model of the relationship between plasma drug concentration and efficacy. 25 Of note, pharmacological analysis shows a clear relationship between plasma drug concentrations and decreased HIV incidence in completed oral PrEP studies. Use of oral PrEP at nearly daily rates appeared to be critical to achieving effective concentrations, and nonuse of study drug was evident in most of the individuals who had seroconverted in these studies. The relationship is less clear for topical microbicides. Recent studies have found that rectal cells from participants exposed to daily TDF gel in MTN 007 exhibited downregulation of a variety of genes. 26 These findings raise the question of whether daily use of a gel could be more detrimental to mucosal integrity than a sex-dependent regimen, since the latter provides mucosa an opportunity to reestablish optimal homeostasis between gel applications.

Preexposure prophylaxis concentration–response curve. Reprinted from Hendrix.25
The safety of FTC-TDF and TDF PrEP for HIV-uninfected persons is also well supported. No serious drug-related adverse events were observed in about 4500 individuals who received TDF or FTC-TDF in 2 pivotal trials and 1 small supportive safety trial, and there was no effect on pregnancy outcomes. 12 , 14 , 27 Small increases in serum creatinine elevation were seen in some patients, but these elevations reversed when drug was stopped and did not appear to correlate with clinical events or other consistent laboratory abnormalities. Small but significant reductions in bone mineral density were also seen but were not associated with clinical findings. Renal and bone parameters and pregnancy intentions should be closely monitored in individuals using PrEP.
Selection for drug resistance with oral PrEP has been rarely seen in the trials to date, but these trials were conducted in controlled environments with monthly monitoring to promptly identify new infections, thus minimizing opportunities to select for resistance. Potential for the selection of or development of resistance is higher with inconsistent use or acute infection at the time of PrEP initiation. HIV testing (particularly testing for acute infection at initiation), education for patients and providers, and adherence support are the keys to preventing resistance. The frequency of HIV testing required for people using PrEP and the feasibility of frequent testing in the context of a public health program have not been established. Demonstration projects in almost a dozen countries are expected to enroll over 32 000 participants to investigate these questions. 28 The potential risks of PrEP adversely affecting public health—specifically selection of and transmission of resistant HIV strains and risk compensation behavior—can be further mitigated by support for safe behavior (including appropriate prescribing), monitoring the results of access and implementation studies and implementation of the risk evaluation and mitigation strategies mandated by the FDA (such as the one developed by Gilead Sciences for its recently approved FTC-TDF PrEP indication). Important issues such as testing frequency and careful exclusion of seroconversion at PrEP initiation must be addressed.
Studies of oral PrEP continue, addressing the following questions:
Efficacy of intermittent dosing regimens
Efficacy, safety, and tolerability of new ARV agents, including maraviroc, dapivirine, and rilpivirine
Long-term safety
Effects of use on sexual behavior and changes in HIV prevention practices
Effects of PrEP safety and efficacy information on adherence and risk behaviors outside of the clinical trial environment
Optimization of delivery (recruitment, retention, adherence promotion, prevention of risk compensation behavior)
Studies are also continuing to investigate nonoral PrEP:
Efficacy and safety of nonoral TDF PrEP formulations (vaginal and rectal gels, intravaginal rings, films, depot injections)
Confirmation in diverse populations of South African women of reduction in HIV and Herpes simplex virus-2 by 1% TDF gel used vaginally before and after sex
Safety and acceptability of use of 1% TDF gel used before and after sex in 16- and 17-year-old girls in South Africa
The interaction between oral and topical PrEP and HIV vaccines is also being studied. Additional research is required in the following areas:
Characterization of rates and patterns of PrEP uptake and adherence and factors influencing each
Methods for monitoring of PrEP use and adherence
Efficacy of sex-dependent use of PrEP
Optimal management of an individual sexually exposed to HIV after interruption of PrEP
Although PrEP is generally thought of as a biomedical intervention, it is also essential to think about it as a behavioral intervention; adherence and maintenance of healthy behaviors are essential to safe and effective use of PrEP. Providers will need to support adherence and safe cycling (HIV testing before reinitiation of PrEP) and be vigilant for development of beliefs of invulnerability or patients’ overestimation of PrEP’s protective effect. To promote PrEP’s success, providers must be prepared to:
Discuss PrEP efficacy and effects of inadequate adherence openly
Explain how to restart PrEP and why this is recommended
Support adherence and provide needed and responsive services; as with TasP, it was noted that recently published guidelines for improving HIV treatment adherence may also be useful in the context of PrEP 20
Frame PrEP use as one of several factors to consider for reducing risk
Explain limitations of PrEP in protection from other sexually transmitted infections
Invite the patient to contribute to the decision and respect the patient’s autonomy
Cost-effectiveness of PrEP is an important consideration for policy makers and funders. A number of models with various assumptions and characteristics have been used to examine the cost-effectiveness of FTC-TDF PrEP in the United States and in South Africa. 29–33 In all the models, the cost-effectiveness ratio is lowest when groups at high risk are the focus of attention and improves substantially if effectiveness improves or cost decreases. Priorities for future study include assessment of new PrEP formulations in clinical trials, evaluation of implementation, and modeling cost-effectiveness of combination interventions such as TasP and PrEP.
Implementation Challenges for PrEP are Numerous
Addressing funding and financial sustainability, including competition for limited ART resources with groups already defined as requiring ART
Identifying the populations at high risk of HIV infection and for whom PrEP use would be most effective and linking and retaining them in care
Identifying the optimal cader of clinicians, as well as locations and ancillary services, for PrEP provision
Advocating equity of access for high risk as well as medically indigent populations
Decreasing stigma against individuals who use PrEP and those who elect not to do so
Addressing overloaded health systems, specifically with respect to retention monitoring and additional laboratory testing and clinic visits
Training and providing continuing education to the health workforce
Changing testing and counseling programs to include PrEP provision as part of the risk reduction package
Developing culturally tailored strategies to support adherence and promote continued use of multiple prevention strategies
As with TasP, implementation of PrEP will be a greater challenge in countries where significant gaps exist in ART coverage among groups of clinically eligible individuals for whom it is currently recommended.
It bears noting that postexposure prophylaxis (PEP), which has been shown to be helpful when individuals are unexpectedly exposed to HIV (eg, sexual assault, needle-stick injury), is an established pillar of biomedical prevention programs. Although there are generally supportive data to suggest that PEP after HIV exposure is effective, 34 randomized controlled clinical trial data are lacking because HIV transmission is inefficient and the presumption of PEP is that exposure was a one-off event. Scaling up programs for the provision of PEP may assist in protecting individuals from acute exposure to HIV and in helping individuals learn how to avoid HIV exposure in the future. PEP may also help to identify individuals who are reexposed to HIV and might benefit from PrEP.
Summit Attendee Response
The majority (82%) of attendees felt that no additional research is needed before using daily oral PrEP. Most (91%) attendees said that their country would require formal PrEP guidelines. Most (70%) also said that in their country PrEP will be used in only select cases, and 24% said PrEP would be met with reluctance in their country.
About one-fourth thought that PrEP should be offered only to 1 high-risk group (MSM, injection drug users, women, or serodiscordant couples) but most thought PrEP should be offered to more than one (33%) or all (43%) of these groups.
Resources and funding for PrEP were a concern for many attendees. Over half (56%) of the attendees chose funding as the largest barrier to implementation of PrEP. Stakeholder acceptance (18%) was another significant barrier. As with TasP, over half (56%) of the attendees stated that PrEP would limit resources for ART as currently indicated. When presented with various breakdowns of national AIDS budget expenditures, some (20%) attendees favored no expenditure on PrEP or 10% expenditure for HIV treatment (ART) and 90% for PrEP (66%).
Key Messages
The evidence of daily oral PrEP efficacy and safety generally supports use in high-risk groups now.
The early discontinuation of the FEM-PrEP study and 2 arms of the VOICE study raise the question of what level of adherence is sufficient to achieve efficacy.
Safety monitoring of individuals taking PrEP as well as of PrEP’s public health effects is required.
PrEP should be used as part of a comprehensive risk reduction package.
PrEP is a biobehavioral intervention that requires support for adherence and other risk reduction strategies.
There are numerous challenges to utilization of PrEP including financial and resource limitations, need for national/global guidelines, identification of high-risk populations and ways to reach them, stigma, health system and workforce capacity, and potential increases in risk from risk compensation in the context of suboptimal adherence.
Conclusions
IAPAC embraces TasP and PrEP, their promise to curb the HIV epidemic, and their injection of new energy into the intertwined HIV treatment and prevention agendas. With sufficient evidence in hand, the time has come to integrate these new prevention approaches into the long-established practices of condom use, male medical circumcision, prevention of mother-to-child transmission, and treatment of sexually transmitted infections.
TasP requires more research into its effectiveness on the population level as well as significant political will, new resources, community involvement, provider support, and individual commitment to provide the increased levels of HIV testing, linkage to and retention in care, access to quality treatment, and adherence—all of which are critical to achieving TasP’s promise.
PrEP requires perhaps even more effort to realize its potential. Although the science of PrEP has achieved a great deal over the last 2 years, much more work is required to establish the optimal drugs, regimens, and delivery for PrEP. Furthermore, demonstration projects must be expanded, linkage to and retention in care for the high-risk populations must be achieved, and strategies for the key challenges of adherence and adoption of health-protecting behaviors must be developed.
Successful TasP and PrEP will require engagement by every single stakeholder but most notably people at risk for HIV/AIDS and PLWHA, their health care providers, their advocates, representative institutions, and the myriad communities in which they live. We are convinced that by fully integrating these 2 biobehavioral interventions into our current armamentarium, we stand a chance of further bending the HIV incidence and AIDS-related mortality curves in a way only imagined years ago and, perhaps—as many have advocated—of welcoming an AIDS-free generation within our lifetimes.
Footnotes
Authors’ Note
The Controlling the HIV Epidemic with Antiretrovirals evidence summit was supported by the following institutional and commercial supporters: IAPAC, BHIVA, Gilead Sciences, and ViiV Healthcare. Neither Gilead Sciences nor ViiV Healthcare had any involvement in the summit’s content or the selection of presenters, panelists, and moderators. They also had no input into the content or involvement in the development of this consensus statement. Additionally, the views reflected in this consensus statement are those of its individual authors and should by no means be construed as having been endorsed by the institutions with which they are affiliated (except in the case of IAPAC representatives).
Acknowledgments
The authors acknowledge the following individuals, who were instrumental to the development of this consensus statement: Project Coordination: Angela Knudson (IAPAC, Washington, DC, USA); Medical Writer: Anne McDonough, MPH (McDonough Clinical Research, London, UK).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
