Abstract
Background:
Factors associated with initial methicillin-resistant Staphylococcus aureus (MRSA) skin and soft-tissue infections (SSTIs) and their recurrence have not been fully elucidated among HIV-infected persons.
Methods:
We retrospectively evaluated a large cohort of HIV-infected patients from 1993 to 2010 for culture-proven MRSA SSTIs. Separate logistic regression models evaluated factors associated with initial and recurrent infections.
Results:
Of the 794 patients, 63 (8%) developed an initial infection (19.8 infections/1000 person years [PY]); risk factors included CD4 count <500 cells/mm3 and HIV RNA level ≥400 copies/mL (P < .01), US Centers for Disease Control and Prevention (CDC) stage C versus A/B (P < .01), and injection drug use (IDU, P < .01). In all, 27% developed recurrence (206 infections/1000 PY); risk factors included hospital admission (P = .02). Minocycline for treatment of the initial infection was associated with an 80% decreased odds for recurrence (P = .03).
Conclusion:
HIV control and avoidance of IDU may be useful in reducing rates of MRSA SSTIs among HIV-infected persons.
Introduction
During the past decade, the incidence of methicillin-resistant Staphylococcus aureus (MRSA) skin and soft tissue infections (SSTIs) has increased dramatically in the inpatient and outpatient settings. 1 –4 Community-acquired strains of MRSA (CA-MRSA) have become the predominant cause of SSTIs in the United States, 5 and outbreaks have been reported worldwide among many diverse groups.6 –9 Recent studies, however, have suggested that the incidence of MRSA infections may now be declining,10,11 but further data are needed.
HIV-infected persons are at a heightened risk of MRSA SSTIs compared with the general population. 4,12 The reasons for the increased risk among HIV-infected persons remain incompletely understood and are likely multifactorial. 13 Poor immune status (eg, low CD4 count and elevated HIV RNA level), recent use of β-lactam antibiotics, absence of trimethoprim-sulfamethoxazole (TMP-SMX) prophylaxis, injection drug use (IDU), and high-risk sexual behaviors (ie, men who have sex with men, public bath use, and multiple sexual partners) have been reported as risk factors. 12,14 –16
HIV-infected persons also appear to be at increased risk of recurrent MRSA SSTIs with recurrence rates of 21% to 71% reported. 13,17,18 However, factors associated with recurrent SSTIs are largely unknown and strategies for the prevention of recurrent events have not been completely devised. In particular, the impact of the initial treatment on recurrent disease requires further study. Additionally, the relationship between MRSA recurrence with CD4 counts and HIV RNA levels has not been fully delineated. Hence, we performed a study to examine the trends in the incidence of MRSA SSTIs among HIV-infected persons over an 18-year period and to determine the clinical characteristics of and factors associated with initial and recurrent MRSA SSTIs.
Methods
We conducted a retrospective study among all HIV-infected patients attending a large HIV clinic from January 1, 1993 to December 31, 2010. The study population included active duty military personnel, dependents, and retirees. HIV-infected military beneficiaries have free access to medical care including HIV medications. Active duty members undergo routine HIV testing (approximately every 2 years) and hence patients were diagnosed early in the course of infection.
The MRSA SSTIs were identified by reviewing all wound culture results utilizing medical and computerized laboratory records. For each positive MRSA wound culture, the medical chart was evaluated to confirm the presence of an infection and to record the site and type of infection. The MRSA was defined using standard microbiologic techniques including a minimal inhibitory concentration of ≥4 mg/L on oxacillin screening.
Clinical data were collected including demographics (age, sex, and self-reported race/ethnicity); body mass index (BMI); history of IDU; relevant medical conditions (eg, hypertension, diabetes mellitus, eczema, and cancer); history of sexually transmitted infections (STIs; eg, syphilis and herpes simplex virus 2 [HSV-2]); duration of HIV infection; current and nadir CD4 counts; current and maximum HIV RNA levels; US Centers for Disease Control and Prevention (CDC) stage; receipt of highly active antiretroviral therapy (HAART); prior receipt of TMP-SMX; and follow-up time. The aforementioned data were collected from the time of HIV diagnosis or first clinic visit (whichever was later) to time of MRSA infection or last clinic visit (whichever occurred first).
Cases of MRSA SSTIs were further investigated to collect data at the time of initial infection and included the site, type, and location of infection; need for hospitalization; CD4 count and HIV RNA level; CDC stage; BMI; and treatments administered (eg, type and duration of antibiotics, incision and drainage). Antibiotics prescribed and procedures performed for the treatment of MRSA SSTIs were done at the discretion of the provider. A recurrent infection was defined as a culture-proven MRSA SSTI at least 30 days following the initial infection or at a distinct anatomical location occurring at least 2 weeks after the initial infection.
Descriptive statistics evaluating the characteristics of the study population and MRSA SSTIs were performed and presented as medians (interquartile ranges [IQRs]) for continuous variables and frequencies (proportions) for categorical variables. The incidence of MRSA SSTIs was calculated for 2-year time intervals, and a chi-square test was used to determine differences in the incidence among time periods. Factors associated with the development of an initial MRSA SSTI were examined using logistic regression models. Full multivariate logistic regression models included factors with a P value ≤.05 in the univariate analyses as well as age and years of follow-up to control for potential confounding. The final multivariate model was derived using stepwise backward elimination with assessment for confounding. In a separate regression analysis, factors associated with the development of a recurrent MRSA SSTI were examined among patients who developed an initial MRSA infection. Odds ratios (ORs), 95% confidence intervals, and P values are reported. Goodness-of-fit of multivariate models was assessed using the Hosmer-Lemshow test; a P value >.10 indicated a good fit.
In order to examine whether CD4 count around the time of the initial MRSA infection affects the development of a recurrent infection, a longitudinal linear mixed effects model was fit using random intercepts associated with each patient as well as a first-order autoregressive covariance matrix for the random error. All patients with at least 1 CD4 count baseline measurement within the 6 months preceding the initial MRSA SSTI and at least 1 follow-up measurement within the 6 months after the initial MRSA SSTI were included in the linear-mixed effects model analysis. A separate linear mixed effects model was fit using log10 HIV RNA level as the dependent variable and similar methods, but with a heterogeneous first-order autoregressive covariance matrix for the random error. The final linear mixed-effects models were derived using the likelihood ratio test for the fixed effects. Least-squares means associated with the categorical factor recurrence of a MRSA SSTI were computed.
All P values were 2-sided and statistically significant at P < .05. All analyses were performed using SAS version 9.2 (SAS Institute, Cary, North Carolina). The current study was approved by the institutional review boards of Naval Medical Center San Diego and Uniformed Services University of the Health Sciences.
Results
Study Population Characteristics
A total of 794 HIV-infected persons were evaluated with a median age of 30 years (IQR 25-37 years); 94% were men, and 46% were white and 30% African American (Table 1). In all, 5% had diabetes and 4% had IDU. The median duration of HIV infection was 7.0 years (IQR 2.5-15.0 years), median current CD4 count was 569 cells/mm3 (IQR 420-743 cells/mm3), median nadir CD4 count was 294 cells/mm3 (IQR 187-396 cells/mm3), 75% had an HIV RNA level <400 copies/mL, and 80% were currently receiving HAART.
Study Population Characteristics and Univariate Analysis of Factors Associated with Initial MRSA SSTIs among HIV-Infected Personsa

Abbreviations: MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection; BMI, body mass index; CI, confidence interval; HSV-2, herpes simplex virus 2; HAART, highly active antiretroviral therapy; TMP-SMX, trimethoprim-sulfamethoxazole; CDC, US Centers for Disease Control and Prevention.
aDependent variable categorized as developed or did not develop a MRSA skin and soft-tissue infection. Data presented represent numbers (percentages) for categorical variables and medians (interquartile ranges) for continuous variables. All data presented are n = 794, except there were missing data for hypertension (n = 2), diabetes mellitus (n = 2), eczema (n = 2), cancer (n = 2), injection drug use (n = 2), nadir CD4 count (n = 1), current CD4 count (n = 4), current HIV RNA level (n = 4), maximum HIV RNA level (n = 5), current CDC stage (n = 2), and current BMI (n = 51).
bOdds ratio, per 10 years.
cCurrent indicates at last follow-up for patients without a MRSA SSTI; for patients with a MRSA SSTI, current indicates the most recent value before the initial infection.
dOdds ratio, per 100 cells/mm3.
Incidence of MRSA SSTIs
In all, 63 (8%) patients developed a total of 108 MRSA infections during 5466 person years (PY) of follow-up (mean 6.9 [standard deviation, 5.9] years of follow-up per person), representing an overall incidence of 19.8 infections/1000 PY. There were no documented cases of culture-positive MRSA SSTIs during the years 1993 to 2002, with the first case occurring in 2003. The incidence of MRSA SSTIs increased significantly from 2003-2004 to 2005-2006 (Figure 1; 16.8 infections/1000 PY vs 40.0 infections/1000 PY; P < .01). The incidence of MRSA SSTIs declined thereafter during the years 2007 to 2010.
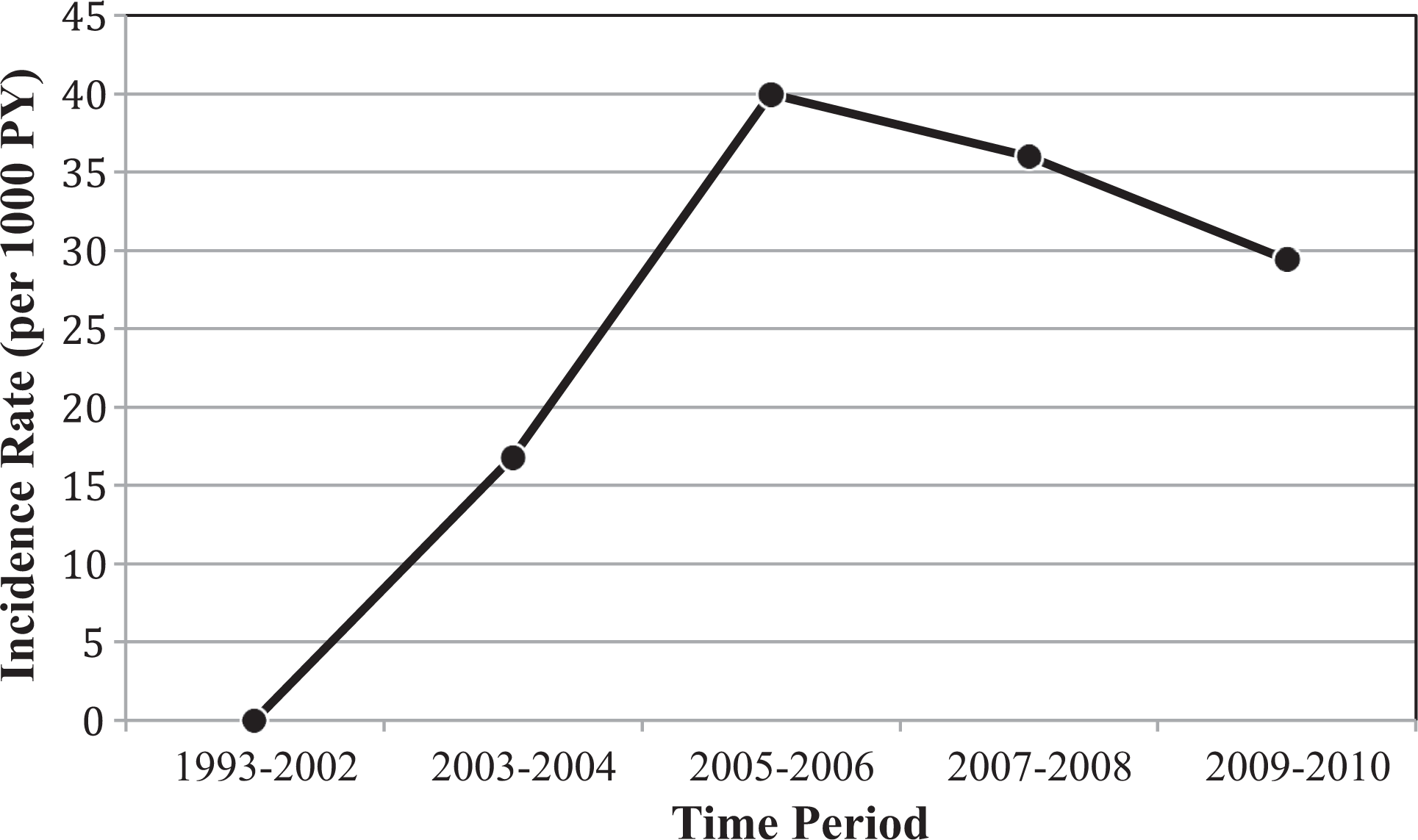
The incidence of MRSA SSTIs among HIV-infected persons, 1993 to 2010. MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection.
Clinical Characteristics of Initial MRSA SSTI
The most common type of initial SSTI was an abscess (44%) followed by wound infection (27%) and cellulitis (21%). The location of the initial MRSA infection was the lower extremity in 28%, buttocks/scrotum in 27%, head/face in 18%, upper extremity in 17%, and trunk in 12%; 2 patients presented with MRSA SSTIs at multiple body sites, including one at the head/face and lower extremity, and another at the upper and lower extremities. All isolates from initial MRSA SSTIs (63 of 63) were susceptible to vancomycin, and the majority were susceptible to TMP-SMX (61 of 63; 97%) and tetracyclines (57 of 63, 91%). In all, 68% (15 of 22) of isolates tested were sensitive to fluoroquinolones (of which 3 had intermediate susceptibility to levofloxacin), and 80% (8 of 10) of isolates tested were sensitive to clindamycin.
Factors Associated with the Development of an Initial MRSA SSTI
Patients who developed a MRSA SSTI compared with those who did not were more likely to have a current CD4 count <350 cells/mm3 (OR 4.1; P < .01), nadir CD4 count <200 cells/mm3 (OR 2.5; P < .01), and current HIV RNA level ≥400 copies/mL (OR 2.7; P < .01) in the univariate models (Table 1). Furthermore, cases of MRSA were more likely to be classified as CDC stage C versus A/B (OR 3.1, P <.01) and IDU (OR 4.7, P <.01). Sex, race, duration of HIV infection, current use of HAART, history of syphilis or HSV-2, use of TMP-SMX, BMI, and other medical history variables (including hypertension, diabetes, eczema, and cancer) were not associated with the development of a MRSA SSTI in the univariate models.
In the multivariate model, current CD4 count and HIV RNA level were combined into 1 variable as these variables were highly correlated (P < .01). The final multivariate model included age at first clinic visit, current CD4 count and HIV RNA level, CDC stage, IDU, and years of follow-up (Table 2). The odds of developing a MRSA SSTI were reduced in patients with optimal CD4 counts and HIV RNA levels (CD4 count ≥500 cells/mm3 and HIV RNA level <400 copies/mL) compared with those with lower CD4 counts (<500 cells/mm3) or higher HIV RNA levels (≥400 copies/mL). Patients with the greatest odds of developing a MRSA SSTI were those who had both a low CD4 count (<500 cells/mm3) and a high HIV RNA level (≥400 copies/mL) (OR 5.2; P < .01). Finally, patients with current CDC stage C versus A/B (OR 5.4; P < .01) and those with a history of IDU (OR 4.9; P < .01) were more likely to experience a MRSA SSTI. Additional analyses were performed excluding patients followed prior to 2003 (data not shown); and, similar results were obtained.
Final Multivariate Logistic Regression Model of Factors Associated with Initial MRSA SSTIs among HIV-Infected Persons (n = 788)a

Abbreviations: CI, confidence interval; MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection; CDC, US Centers for Disease Control and Prevention; df, degrees of freedom; χ2, chi-square; VL, viral load.
aDependent variable categorized as developed or did not develop a MRSA skin and soft-tissue infection. Hosmer-Lemeshow goodness-of-fit test results: χ2 = 11.00, df = 8, P value = .20 > .10, which indicates model is a good fit.
bCurrent indicates at last follow-up for patients without a MRSA SSTI; for patients with a MRSA SSTI, current indicates the most recent value before the initial infection.
Factors Associated with the Development of a Recurrent MRSA SSTI
A total of 17 (27%) HIV-infected patients with an initial MRSA SSTI developed a recurrent SSTI. Regarding the first recurrent event, 65% (11 of 17) occurred at a distinct anatomical location from the initial infection. The total number of recurrent MRSA SSTIs was 45 for an incidence of 206 recurrent infections per 1000 PY among those with an initial infection. The median number of recurrent infections was 1 (range 1-20) and the median time to first recurrence was 4.7 months (range, 1-80 months).
Patients who developed a recurrent MRSA SSTI compared with those who did not were older (42 years vs 36 years, P = .03), had a lower CD4 count at initial MRSA SSTI (337 vs 475 cells/mm3, P = .02), lower nadir CD4 count (82 vs 264 cells/mm3, P < .01), higher maximum log10 HIV RNA level (5.3 vs 5.0 copies/mL, P = .02), history of an AIDS-defining illness (ie, CDC stage C; OR 4.2, P = .02), history of cancer (OR 5.7, P = .02), and IDU (OR 6.0, P = .03) in the univariate models (Table 3). Regarding management of the initial MRSA SSTI, receipt of minocycline as the initial antibiotic was marginally associated with a lower odds of recurrence (OR 0.3, P = .05); patients who received minocycline were more likely to be white (P = .02) and have a longer duration of antibiotic therapy (P <.01). Patients who had been hospitalized for the initial MRSA infection had a higher odds of recurrence (OR 5.0, P = .01). Additionally, patients with a recurrent SSTI were more likely to have had a hospital admission (OR 3.6, P = .05) or surgery (OR 3.8, P = .03) after the initial infection. Factors not associated with the development of a recurrent MRSA SSTI included receipt of decolonization medications (ie, mupirocin and hexachlorophene), treatment with other specific antibiotic classes (ie, TMP-SMX, vancomycin, β-lactam antibiotic, fluoroquinolone), incision and drainage, use of combination antibiotics for empiric therapy, receipt of an empiric antibiotic effective based on the MRSA isolate sensitivity pattern, or duration of antibiotic therapy.
Univariate Analysis of Factors Associated with Recurrent MRSA SSTIs among HIV-Infected Personsa
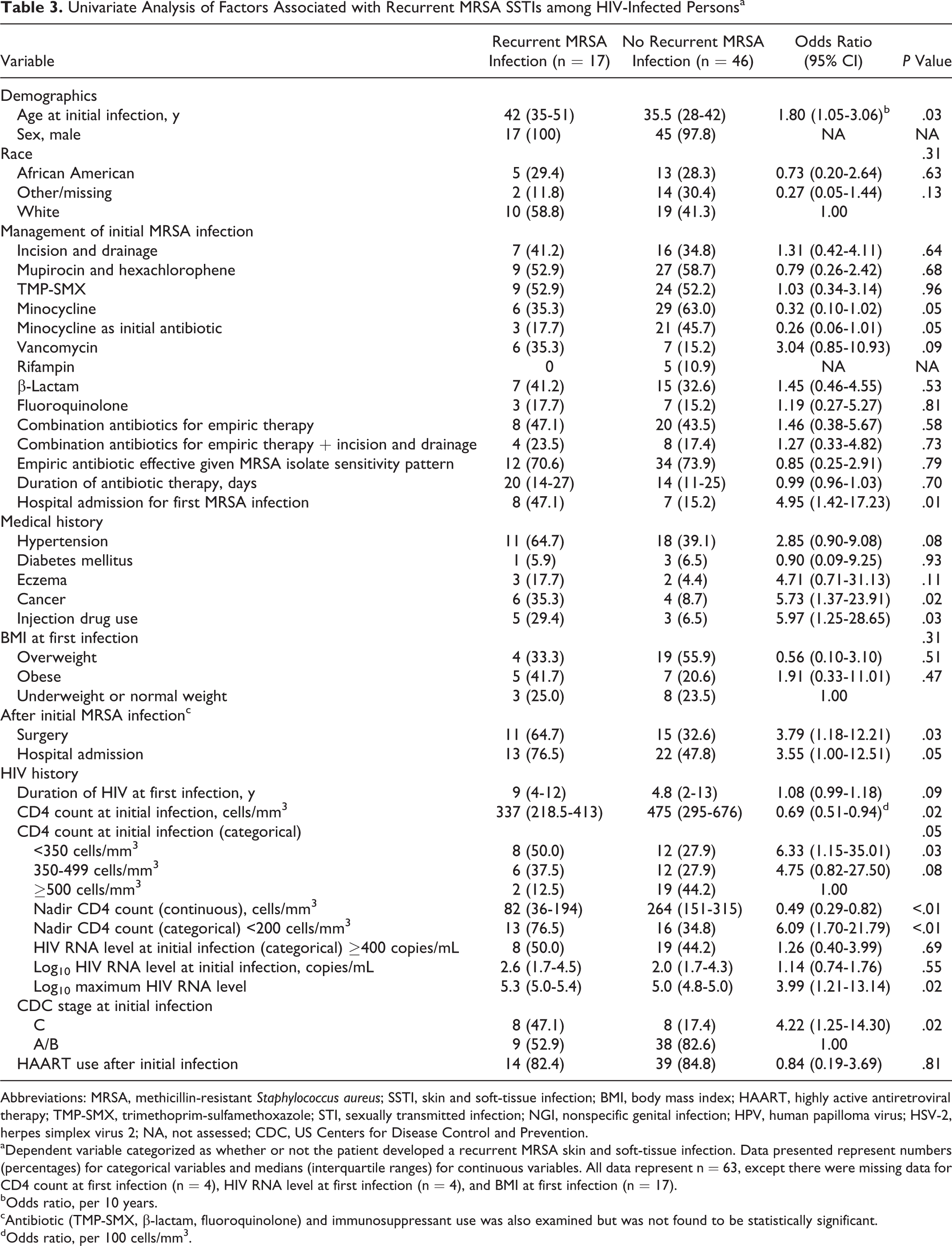
Abbreviations: MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection; BMI, body mass index; HAART, highly active antiretroviral therapy; TMP-SMX, trimethoprim-sulfamethoxazole; STI, sexually transmitted infection; NGI, nonspecific genital infection; HPV, human papilloma virus; HSV-2, herpes simplex virus 2; NA, not assessed; CDC, US Centers for Disease Control and Prevention.
aDependent variable categorized as whether or not the patient developed a recurrent MRSA skin and soft-tissue infection. Data presented represent numbers (percentages) for categorical variables and medians (interquartile ranges) for continuous variables. All data represent n = 63, except there were missing data for CD4 count at first infection (n = 4), HIV RNA level at first infection (n = 4), and BMI at first infection (n = 17).
bOdds ratio, per 10 years.
cAntibiotic (TMP-SMX, β-lactam, fluoroquinolone) and immunosuppressant use was also examined but was not found to be statistically significant.
dOdds ratio, per 100 cells/mm3.
In the final multivariate model, receipt of minocycline as the initial antibiotic for treatment of the initial infection was associated with a lower odds for recurrent disease (OR 0.2; P = .03; Table 4). Furthermore, hospital admission after the initial infection (OR 7.4; P = .02) was significantly associated with the development of a recurrent MRSA SSTI. A lower CD4 count at the time of the initial MRSA infection was marginally significantly associated with a recurrent infection (OR 0.7, P = .07).
Final Multivariate Model of Factors Associated with Recurrent MRSA SSTIs among HIV-Infected Persons (n = 59)a

Abbreviations: CI, confidence interval; MRSA, methicillin-resistant Staphylococcus aureus; SSTI, skin and soft-tissue infection; df, degrees of freedom; χ2, chi-square.
aDependent variable categorized as developed or did not develop a recurrent MRSA skin and soft-tissue infection. Hosmer-Lemeshow goodness-of-fit test results: χ2 = 5.26, df = 8, P value = .73 > .10, which indicates model is a good fit.
bOdds ratio, per 10 years.
cOdds ratio, per 100 cells/mm3.
Impact of CD4 Count and HIV Viral Load on Recurrence
Using longitudinal linear mixed effects modeling, patients with HIV who experienced a recurrent MRSA SSTI had on average a CD4 count of 151 cells/mm3 lower in the 6-month window before and after the initial MRSA SSTI than patients without recurrence (P = .03). For patients with recurrence, the lowest median CD4 count was observed 6 months after the initial MRSA SSTI (data not shown). In a separate linear mixed effects model, there was no significant association between log10 HIV RNA level and recurrence (P = .34).
Discussion
This study provides one of the most comprehensive evaluations of the trends and factors associated with MRSA SSTIs among a cohort of HIV-infected persons. Despite a rapid rise of MRSA SSTIs among HIV-infected persons prior to 2007, our study demonstrates that these infections may now be decreasing. Additionally, our study confirms the importance of HIV control in potentially reducing both initial and recurrent MRSA SSTIs among HIV-infected persons. Further, we found that the use of minocycline for initial MRSA SSTI treatment was associated with a reduced risk of recurrence. These data provide important potential strategies to reduce the burden of MRSA infections among HIV-infected persons.
Compared with MRSA infection incidence rates of 0.2 to 1.6 infections per 1000 PYs in the general population, our study reported a higher overall incidence rate of 19.8 infections per 1000 PYs, consistent with the observation that HIV-infected persons are at increased risk of MRSA infections compared with persons not infected with HIV. 1,4,19 In comparison with another study among the general military population served at the same medical facility as the current study, in which an incidence of 2.3 infections/1000 PYs was observed, this represents an approximate 9-fold increase in risk. 1 Over a 4-year period (2003-2006), we documented a 2.4-fold increase in the incidence of MRSA SSTIs in our HIV cohort, consistent with trends noted in other studies and likely the result of the emergence of the MRSA USA300 strain in community-acquired infections. 4,16 Since 2007, however, we documented a 27% decrease in the incidence of MRSA SSTIs, consistent with findings reported in a recent study among HIV-infected outpatients.10 The reasons for this observed decreasing trend in incidence are unclear, but may be due to improved infection control practices or HIV-control with broadening antiretroviral use. Additionally, temporal variations in MRSA incidence may mirror trends in the general population.
The overall proportion of patients with HIV who developed a MRSA SSTI in our study was 8%. IDU and poor HIV control (ie, lower CD4 counts, higher HIV RNA levels, and CDC stage C disease) were associated with the development of an initial MRSA SSTI. Studies among other HIV-infected cohorts have found that low CD4 count is a risk factor for MRSA infection,10,16,20 while others have implicated high HIV RNA levels as a risk factor. 16 These associations are likely related to poor host immune responses (including neutrophil and Th17 cell functioning) to S. aureus infection in the setting of low CD4 count and uncontrolled HIV replication. 21
The recurrence rate of MRSA infections in our cohort was substantial— 27% developed a novel infection within a median of 4.7 months. Compared with a study among the general population served at our medical facility, our recurrence rate was higher (27% vs 9%), suggesting that HIV-infected persons are at increased risk of recurrent infections compared to HIV-uninfected persons. 1 Determining factors associated with recurrent disease is important, but few data exist within the literature. Our study noted that subsequent hospitalization after the initial MRSA infection was associated with recurrence. Hospitalization may facilitate the spread of MRSA colonization via contaminated surfaces or direct contact with patients/health care providers carrying MRSA or be related to procedures (eg, surgery, line placement) that increase the risk of subsequent infection. Further studies are needed to precisely define the relationship between hospitalization and recurrence, including the molecular characterization of the initial and recurrent strains.
Similar to the initial MRSA infection, CD4 counts were additionally important in the development of recurrent disease—those with lower CD4 counts around the time of the initial infection were more likely to experience recurrence. Previously, the association between CD4 count and recurrent MRSA infections had not been fully elucidated; our linear mixed effects analysis is novel and has the important advantage of taking into account multiple CD4 count measurements over time.
Despite the wide variety of antimicrobial options available for the treatment of MRSA infections, guidelines for the optimal management of MRSA SSTIs have not been devised.22 Irrespective of the prescribed regimen, cure rates of initial MRSA SSTIs in the general population (80%-90%) and among HIV-infected persons (93%) are relatively high, 18,22 but little data exist on the role of antimicrobial type and prevention of recurrence.
To our knowledge, this study is the first to show that receipt of minocycline as the initial antibiotic may be protective against recurrent MRSA SSTIs. This finding is novel and suggests that minocycline may be an important part of the armamentarium against MRSA SSTIs. Of note, the majority (91%) of MRSA isolates at the initial infection were sensitive to this antibiotic. The rationale for the clinical superiority of minocycline in our study may be multifactorial: minocycline has excellent skin/soft tissue penetration and efficacy at eradicating S. aureus skin infections; administration is relatively simple leading to favorable antibiotic completion rates; and minocycline may reduce concurrent colonization that may be critical in preventing future recurrence events. 23 –25 In addition to the antibacterial characteristics of minocycline, an in vitro investigation recently found that minocycline produced a dose-dependent reduction in HIV replication and a decrease in immune activation (ie, decreased expression of activation [CD25] and proliferation [Ki-67] markers, and reduced the secretion of cytokines such as interleukin 2, interferon-γ, and tumor necrosis factor-α). 26 Finally, minocycline is an attractive option for MRSA treatment as it is safe and inexpensive. Since our number of treated cases was small and data on the exact size and severity of the SSTIs treated were not available, further studies are needed to evaluate the role of minocycline in recurrent disease.
Our study had some limitations. First, due to the retrospective nature of the study, causality could not be established between factors of interest and the development of initial and recurrent MRSA SSTIs. Second, although our study evaluated a large HIV-positive cohort, we did not collect data on HIV-uninfected persons and therefore direct comparisons could not be performed; however, we did utilize data collected from prior reports among HIV-uninfected persons at our medical facility. 1 Although we reported an association between low CD4 counts and elevated HIV RNA levels with MRSA infections, it is unclear whether HIV control itself is related or whether this is a marker for other conditions or health behaviors. Third, we only analyzed HIV-infected military beneficiaries, which are a unique population with a low prevalence of IDU and comorbidities, male predominance, mandated follow-up, and unique occupational exposures among active duty members; therefore, our results may not be generalizable to other HIV-infected populations. Our study did not collect data on sexual behaviors or orientation. Finally, we do not have data on the molecular characteristics of the isolates over time and could not determine whether the same strain was responsible for both the initial and the recurrent infections.
Strengths of our study included the long duration of the study (over an 18-year period) and the comprehensive data collection including demographic, HIV-related, and clinical factors. Furthermore, we evaluated factors associated with recurrent infections, including antibiotic management and immune status, which to date had not been fully elucidated among HIV-infected persons.
In summary, HIV-infected persons have a high rate of both initial and recurrent MRSA SSTIs. In recent years, however, the incidence rate of MRSA SSTIs may be decreasing in the HIV-infected community. HIV control and avoidance of IDU are important for the prevention of MRSA infections. Additionally, minocycline may be the preferable initial antibiotic for MRSA SSTIs and patients who experience recurrent infections; future randomized, controlled trials should be conducted to evaluate the optimal antibiotics for MRSA infections among HIV-infected persons.
Footnotes
Authors’ Note
The authors have no financial interest in this work. All authors contributed to the content of the manuscript and concurred with the decision to submit it for publication. This work is original and has not been published elsewhere. The content and views expressed in this publication is the sole responsibility of the authors and does not necessarily reflect the views or policies of the NIH or the Department of Health and Human Services, the DoD or the Departments of the Army, Navy, Air Force, Department of Defense, nor the US Government. Mention of trade names, commercial products, or organizations does not imply endorsement by the US Government. Some data contained in this manuscript were presented at the XIX International AIDS Conference: “Trends and Factors Associated with Initial and Recurrent Methicillin-Resistant Staphylococcus aureus (MRSA) Skin and Soft-Tissue Infections among HIV-Infected Persons: An Eighteen-Year Study”. Washington DC, USA, July, 22-27, 2012. Abstract #17685.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this work (IDCRP-003) was provided by the Infectious Disease Clinical Research Program (IDCRP), a Department of Defense (DoD) program executed through the Uniformed Services University of the Health Sciences. This project has been funded in whole, or in part, with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), under Inter-Agency Agreement Y1-AI-5072. The study sponsors were not involved in the study process or during manuscript preparation.
