Abstract
Noni (Morinda citrifolia) is a widely plant-based herbal product with purported antioxidant, anti-inflammatory, and immunomodulatory properties and is widely available in oral, liquid, and topical formulations. Hepatotoxicity from juice and oral formulations are infrequent and liver injury associated with topical noni exposure has not previously been described. We report the case of a 60-year-old male with a history of hypertension and hypothyroidism who presented with 5 days of jaundice, generalized pruritus, dark urine, and acholic stools. Laboratory evaluation showed elevated liver enzymes in a cholestatic pattern. The patient reported prolonged use of a topical noni-containing analgesic cream for approximately 8 months, which had been discontinued 3 months prior to symptom onset. A through diagnostic evaluation excluded viral, autoimmune, and metabolic etiologies of the liver disease. Given the exposure history, clinical presentation, and exclusion of alternative diagnoses, the drug-induced liver injury was attributed to the topical noni product. The management included close outpatient follow-up until resolution of liver function tests 6 months after discharge after the discontinuation of the product. The precise mechanism for noni toxicity remains unclear, but a potential proposed pathway includes anthraquinone-mediated oxidative stress with subsequent mitochondrial dysfunction, but further investigation is warranted to better characterize systemic absorption and hepatotoxic risk associated with topical noni formulations.
Introduction
Drug-induced liver injury (DILI) represents a significant diagnostic and therapeutic challenge, with dietary supplements and herbal products being increasingly recognized as culprits. Noni (Morinda citrifolia) is a popular and widely marketed health supplement for its purported health benefits, and has been implicated in both hepatocellular and cholestatic liver injury in several cases.1-4 While much attention has been paid to link between oral noni consumption and liver, the potential for hepatotoxicity through prolonged topical use has been less explored. This case entails the hepatotoxic effects of long-term noni cream application in a patient presenting with cholestatic jaundice.
Case Presentation
Demographic and History of Present Illness
A 60-year-old male with a medical history of hypertension (controlled with amlodipine 5 mg daily) and hypothyroidism (managed with levothyroxine 150 μg daily) presented to the emergency department in December with complaints of yellow discoloration of the skin, generalized pruritus, dark yellow urine, and acholic stools. He denied any abdominal pain, fever, or unintentional weight loss. Notably, he reported fever, chills, and vomiting 5 days before the onset of jaundice. The patient had traveled to Mexico in February but denied recent alcohol consumption, or intravenous drug use.
The patient had been using a noni-based topical analgesic cream for a shoulder injury twice daily for 8 months. He discontinued the cream 3 months prior to presentation, believing it was no longer necessary. He denied any oral consumption of noni or recent use of hepatotoxic medications. His family history was significant for hypertension, hyperthyroidism, and hypothyroidism. The patient weighed 73.5 kg (BMI 24.6 kg/m2) at follow-up. He reported applying a generous fingertip-sized amount of the topical noni cream twice daily to both shoulders and the upper back for 8 months. He denied any open wounds, rashes, or dermatologic conditions during the time of use, and examination at presentation revealed intact, healthy skin. There was no oral use of noni and no concurrent hepatotoxic medications. The product was identified as the Karla Caribbean Cosmetics Noni Rejuvenation Face Cream Set, purchased online through an international vendor (NinthAvenue, Israel). According to the manufacturer’s public product page, the cream contains Morinda citrifolia extract, eucalyptus oil, menthol, lanolin, and capsaicin. Precise concentrations and regulatory standards were not available.
Physical Examination
On examination, the patient appeared jaundiced but was alert and oriented, with no signs of acute distress. Scleral icterus was evident. Cardiovascular and respiratory examinations were normal, with no murmurs or abnormal breath sounds. Abdominal examination revealed a soft, nontender, and nondistended abdomen with no hepatosplenomegaly. Skin examination confirmed generalized jaundice but was otherwise unremarkable, with no rashes, or lesions.
Laboratory Results
Initial laboratory revealed a cholestatic pattern of liver injury:
Alkaline phosphatase (ALP): 368 U/L.
Aspartate aminotransferase (AST): 208 U/L.
Alanine aminotransferase (ALT): 499 U/L.
Total bilirubin: 12.4 mg/dL.
Direct bilirubin: 9.4 mg/dL.
Gamma-glutamyl transferase: 471 U/L.
Other notable findings included a negative viral hepatitis panel (HAV, HBV, HCV, HEV), negative autoimmune markers (ANA, ASMA, AMA), and no evidence of Wilson’s disease or α1-antitrypsin deficiency. Imaging studies, including a computed tomography (CT) abdomen and pelvis and magnetic resonance cholangiopancreatography (MRCP), were unremarkable, ruling out structural or obstructive causes of cholestasis.2,3,5,6
Discussion
Noni (Morinda citrifolia) has gained popularity for its purported health benefits, but increasing evidence links it to hepatotoxicity. The hepatotoxicity of noni products is attributed to several mechanisms, primarily involving the anthraquinones present in the plant. Anthraquinones, such as damnacanthal, are known to induce oxidative stress and mitochondrial dysfunction, leading to hepatocyte injury. 3 Chronic exposure, even through the skin, can result in cumulative toxicity as these compounds are absorbed into systemic circulation over time. 1 Anthraquinones are lipophilic and may accumulate in adipose tissue, potentially releasing slowly into the bloodstream over time. While formal pharmacokinetics for topical noni are unavailable, this could explain the latency of several months between discontinuation and symptom onset.7-10 A delayed steady state or prolonged elimination half-life due to lipid storage may underlie this temporal gap.4,11
As shown in Figure 1 the patient demonstrates the combination of elevated liver function tests in a cholestatic pattern. Although elevated, the patient’s AST and ALT levels (208 and 499 U/L, respectively) did not approach fulminant thresholds often seen in acute toxic hepatitis. However, the marked hyperbilirubinemia (total bilirubin: 12.4 mg/dL) and ALP elevation (425 U/L) were notable, supporting a mixed hepatocellular-cholestatic pattern of injury consistent with DILI. Noni has immunomodulatory properties that may trigger immune-mediated liver injury in susceptible individuals.12,13 For example, the activation of inflammatory pathways, including the production of cytokines such as tumor necrosis factor-alpha, has been observed in studies involving noni products. 14 The immune response could exacerbate liver injury, especially in patients with underlying comorbidities such as hypothyroidism as in this patient, which may alter immune regulation. Figure 2 shows the schematic of proposed toxicity where anthraquinones generate reactive oxygen species and disrupt mitochondrial respiration, impairing energy production in hepatocytes that lead to cell apoptosis or necrosis.3,14,15

Trend of liver function tests over a 6-month period following discontinuation of topical noni (Morinda citrifolia) cream. Initial laboratory evaluation demonstrated a cholestatic pattern of injury with markedly elevated alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin, and direct bilirubin. Following cessation of the topical product and supportive management, all values progressively declined and normalized by approximately 6 months, consistent with drug-induced liver injury.
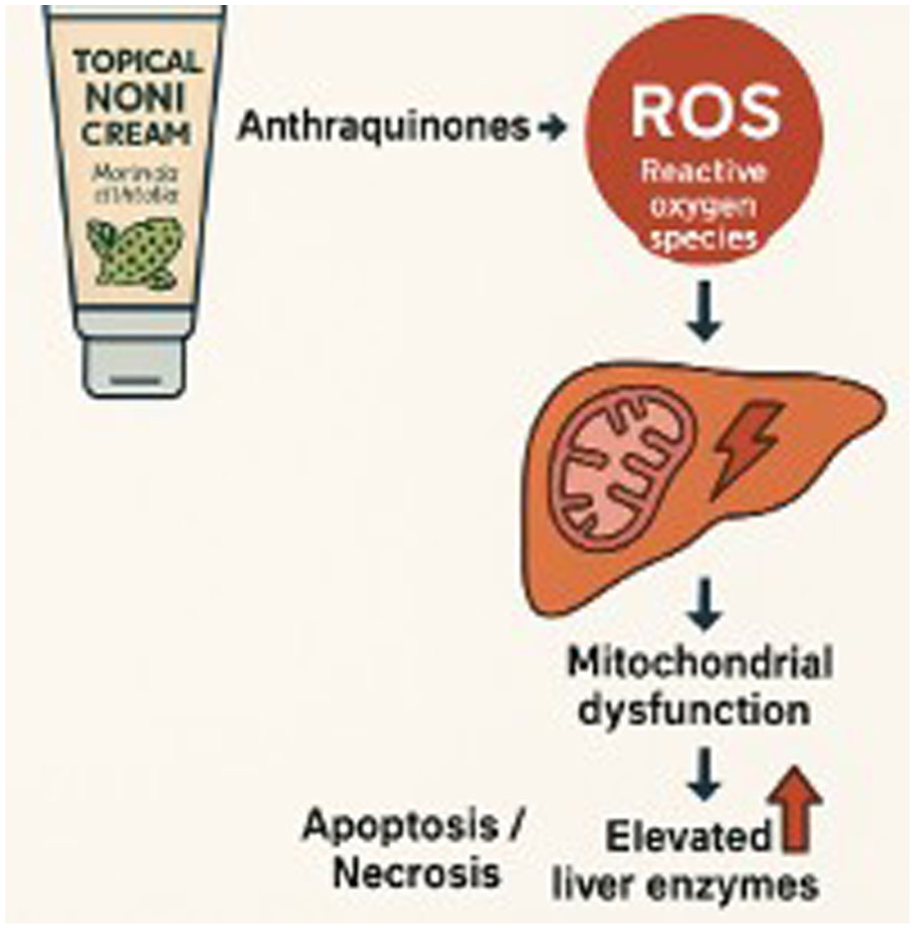
Proposed mechanism of hepatotoxicity associated with noni (Morinda citrifolia). Anthraquinone compounds generate reactive oxygen species, leading to mitochondrial dysfunction, impaired hepatocellular energy production, and subsequent apoptosis or necrosis, resulting in elevated liver enzyme levels.
Although the patient discontinued the topical noni cream 3 months before the onset of symptoms, the prolonged use over 8 months likely contributed to cumulative toxicity. Lipophilic compounds like anthraquinones can remain stored in adipose tissue and release slowly into the bloodstream, sustaining their hepatotoxic effects long after exposure ceases.16,17 This delayed presentation is consistent with reports of idiosyncratic DILI from other herbal products. 5
Despite claims that noni products have hepatoprotective effects, the scientific evidence remains controversial.18-21 Most reports of noni-associated hepatotoxicity involve products marketed as “natural” remedies, undermining the risks of unregulated supplements and highlighted that the lack of premarket testing and quality control for herbal products increases the risk of adverse reactions. 3
This patient’s clinical presentation is consistent with previously reported cases of noni-associated liver injury. Stadlbauer et al reported 2 cases of acute hepatitis and subacute hepatic failure following noni juice consumption, with 1 case requiring liver transplantation. 17 Similarly, Mrzljak et al 2 described hepatocellular injury from noni juice in a patient on concurrent phenobarbital therapy, emphasizing the synergistic effects of noni and other hepatotoxic agents. Although this patient used noni in a topical form, systemic absorption through the skin is plausible, particularly with prolonged use, and could cause the observed liver injury.
The diagnosis of DILI is often challenging and mostly done by exclusion, requiring thorough evaluation to rule out alternative causes such as viral hepatitis, autoimmune hepatitis, and metabolic liver diseases. The latency period between exposure and symptom onset in this patient aligns with the idiosyncratic nature of DILI caused by noni, which can manifest weeks to months after initial exposure. 5
Conclusion
Healthcare providers should maintain a high index of suspicion for DILI in patients with unexplained liver dysfunction, particularly when herbal products are involved. Public education is essential to inform patients about the misconception that mot all “natural” remedies are inherently safe. This case indicates the hepatotoxic potential of noni (Morinda citrifolia), even with topical use. The delayed onset of symptoms and the idiosyncratic nature of DILI highlight the need for through interview with patients in identifying possible herbal exposures. Clinicians should routinely inquire about the use of supplements and herbal products when evaluating patients with liver dysfunction.
Footnotes
Author Note
Prior presentation of abstract statement: This work was previously presented in abstract form at the American College of Gastroenterology (ACG) Annual Scientific Meeting, Phoenix, Arizona, October 24-29, 2025.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
