Abstract
Acute promyelocytic leukemia (APML) is a rare hematologic emergency with a high mortality rate due to bleeding diathesis, which is due to a disseminated intravascular coagulation-like coagulopathy; APML is complicated enough to treat on its own and becomes particularly challenging when it occurs during pregnancy due to the complexities in managing both maternal and fetal health. APML is associated with a challenging therapeutic dilemma for pregnant women, and there is a risk of fetal malformations and developmental abnormalities caused by exposure to chemotherapy. A 31-year-old woman at 29 weeks of gestation presented with a 3-week history of fatigue. Complete blood count revealed pancytopenia, and further evaluation confirmed a diagnosis of APML. Due to her severe thrombocytopenia and associated pregnancy risks, ATRA therapy was initiated, and a primary Cesarean section was performed at 31 weeks 3 days of gestation to mitigate maternal and fetal complications. After delivery, arsenic trioxide was added to the treatment regimen, resulting in a favorable response. In this case report, we discuss clinical decisions and therapeutic interventions and compare our patient’s case with those found in the literature. This case highlights the importance of prenatal care and early intervention in improving outcomes for both mother and child.
Keywords
Introduction
Acute myeloid leukemia (AML) is the clonal proliferation of myeloid progenitor cells. These cells acquire genetic mutations that prevent them from differentiating into mature blood cells, and instead accumulate, leading to bone marrow failure and subsequent cytopenias. Acute promyelocytic leukemia (APML) is a distinct subset of AML characterized by the arrest of myeloid differentiation in the promyelocyte stage of development. The hallmark of this cancer is a translocation between the promyelocytic leukemia (PML) gene on chromosome 15 with the retinoic acid receptor alpha (RARA) gene on chromosome 17. 1
Under normal circumstances, the RARA gene on chromosome 17 encodes a nuclear receptor that allows for the regulation of gene expression in response to retinoic acid, which is derived from vitamin A. The RARA protein forms a heterodimer with the retinoid X receptor (RXR) and binds to sequences of DNA called retinoic acid response elements. This allows for activation of gene expression that promotes the differentiation of myeloid precursors into mature granulocytes. The PML protein is a tumor suppressor that activates p53 to promote the transcription of pro-apoptotic genes to cause programmed cell death in response to stress or DNA damage. 2
In APML, the t(15,17) translocation results in a chimeric, fusion PML-RARA protein that retains domains from both the PML and RARA proteins, altering its function. This new protein forms homodimers or heterodimers with RXR but binds to the DNA sequences with higher affinity. 3 This binding has a dominant negative effect because it recruits co-repressor complexes like nuclear corepressor (NCoR) and histone deacetylases (HDACs) to the DNA. Together, these complexes cause transcriptional repression of genes necessary for myeloid differentiation through alteration of the chromatin. In addition, this fusion protein disrupts the normal function of the PML nuclear bodies, which impairs apoptotic pathways and allows for the survival of abnormal promyelocytes. 4
The mainstay of treatment in APML is all-trans retinoic acid (ATRA). ATRA binds the altered RARA portion of the fusion protein, promoting the release of the NCoRs and HDACs. In turn, this release allows for the transcriptional activation of differentiation genes, thereby causing promyelocytes to mature into functional granulocytes. 5 The introduction of ATRA fundamentally changed the prognosis of APML, transforming a previously fatal leukemia into one of the most curable forms of acute leukemia. Tallman et al 6 demonstrated, in a landmark New England Journal of Medicine study, that ATRA, used in combination with anthracycline-based chemotherapy, produced complete remission rates exceeding 90% and long-term survival rates approaching 75% to 85%, establishing ATRA as the cornerstone of APML treatment. Similarly, Hoffman et al 7 reported one of the earliest successful uses of ATRA in a pregnant woman with APML, achieving complete remission and demonstrating that remission induction with ATRA during pregnancy could be both feasible and safe when carefully timed with delivery. Their report, along with subsequent cases, underscored the potential for balancing maternal treatment and fetal safety through coordinated multidisciplinary management.
This report of a pregnant 31-year-old female with APML contributes to the limited literature regarding the description and treatment of APML in pregnancy, especially in the third trimester. Cases of this condition diagnosed in the third trimester, such as this one, allow for the delivery of the fetus via c-section in the early stages of the disease. Maternal treatment with ATRA before delivery allows for a rapid reduction in the number of abnormal promyelocytes, which mitigates the risk of bleeding complications that could endanger the mother and fetus. The induction of arsenic trioxide (ATO) post-delivery allows for effective treatment without harming the fetus.1,8,9 The studies done by Fei et al 10 and Li et al 11 reinforce that the third trimester provides an optimal period for delivery, allowing for immediate postpartum treatment initiation for the mother while maintaining safety for the fetus. Both of these studies reported favorable outcomes when the timing of delivery corresponded with APML-specific therapy. This case aligns with those findings, underscoring that for a third-trimester diagnosis, prompt cesarean delivery followed by targeted APML therapy is a viable strategy for optimizing maternal and fetal outcomes.
This report provides a demonstration of effective interdisciplinary collaboration between obstetrics, hematology, and neonatology, as this coordinated approach to care was essential for managing the APML while mitigating risks to both mother and fetus. The recognition of the vague symptoms along with vigilant prenatal care via routine blood testing facilitated early diagnosis. In turn, this early detection allowed for proactive and early interventions, preventing severe APML-related complications such as coagulopathy and hemorrhage. 5 Finally, this report highlights the importance of ongoing research that allows for the development of standardized guidelines to refine APML management during pregnancy. Enhanced guidelines would provide a valuable framework by which clinicians could balance aggressive maternal treatment and fetal safety in future cases.
Case Presentation
A 31-year-old female G2P1001 presented to the hematology clinic at 28 weeks of gestation after a recent outpatient primary prenatal visit, revealing a new finding of generalized pancytopenia. At that time, she endorsed a 3-week history of episodes of lightheadedness and dizziness. She denied shortness of breath, chest pain, palpitations, joint pain, fever, chills, night sweats, sore throat, autoimmune disorders, hematochezia, and hemoptysis. She reported a 3-pound weight loss and significant bleeding from her gums and nose. She has always had easy bruising with contact in the past, but in the weeks before presentation, she reported having bruising without an identifiable cause. Her child had been very sick the week before her presentation, but a viral respiratory panel came back negative. She endorsed streaks of blood in her vomit for the 2 weeks prior to presentation, more consistent with the bleeding in her gums than concerns for a gastrointestinal source.
Initial differential diagnoses for the patient’s presentation included anemia of pregnancy, infection (parvovirus B19), leukemia, and drug reaction. The patient was ordered labs, which included a complete blood count (CBC) that showed pancytopenia; WBC 0.62, RBC 2.95, Hgb 9.4, Hct 25.5, MCV 86.4, MCH 31.9, MCHC 36.9, RDW 14.6, Plts 19, and MPV 10.3. A peripheral smear was done, which demonstrated reduced RBCs, marked leukopenia, and occasional blasts/promyelocytes. A bone marrow biopsy was then completed, which showed hypercellular marrow with 70% atypical promyelocytes. Analysis of the obtained sample with fluorescence in situ hybridization (FISH) showed a PML-RARA rearrangement in 69% of cells, confirming the diagnosis of APML. Finally, flow cytometry with immunophenotyping demonstrated strong positivity for CD13 and CD33 (dim), weak expression of CD45, and increased CD71, with absent CD34 and HLA-DR. These findings are all consistent with a diagnosis of APML. Morphologically, the abnormal promyelocytes displayed folded or bilobed nuclei with Auer rods and granular cytoplasm (Figures 1a and 1b). CD34 staining was negative (Figure 2), while CD117 was positive in the majority of abnormal promyelocytes (Figure 3), further supporting a diagnosis of APML. Ecadherin staining (Figure 4) showed membranous positivity in leukemic promyelocytes, and terminal deoxynucleotidyl transferase was negative (Figure 5a and b), confirming the absence of lymphoid lineage involvement. A final diagnosis of APML with PML-RARA rearrangement was made based on these bone marrow findings and FISH analysis. Because of this diagnosis, treatment with ATRA was started at week 28 of pregnancy.

(a, b) Hematoxylin and eosin (H&E) stain of the initial bone marrow biopsy showing a hypercellular marrow with abnormal promyelocytes with folded nuclei and prominent Auer rods.

CD34 immunostaining shows a lack of CD34 expression in the abnormal promyelocytes, a characteristic feature of APML. APML, acute promyelocytic leukemia.

CD117 on initial bone marrow biopsy, highlighting strong cytoplasmic positivity in abnormal promyelocytes. This supports a diagnosis of APML. APML, acute promyelocytic leukemia.
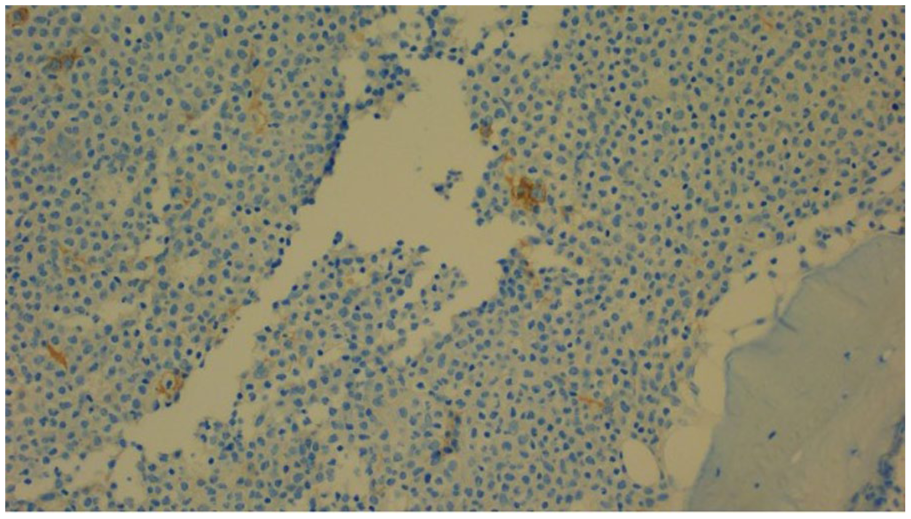
E-cadherin stain on the initial bone marrow biopsy shows membranous positivity in leukemic promyelocytes, consistent with aberrant expression sometimes observed in APML. APML, acute promyelocytic leukemia.

(a, b) Terminal deoxynucleotidyl transferase (TdT or TP) staining is negative, ruling out lymphoid lineage involvement.
The dosage of 40 mg was given twice daily from September 28 to October 28 (45 mg/m2 total daily). On October 24, 2024, the patient reported a persistent headache with blurry vision. She was evaluated by neuro-ophthalmology, who found bilateral disc edema with many hemorrhages surrounding the disc and mild cranial nerve VI palsy consistent with idiopathic intracranial hypertension (IIH). The ATRA was held on October 28 due to the patient developing a side effect of IIH. The headaches resolved when the ATRA was stopped. A magnetic resonance imaging brain and magnetic resonance venography were completed and ruled out intracranial lesions or dural venous sinus thrombosis, as well as demonstrated no evidence of myeloid cells and flow cytometry on cerebrospinal fluid.
The patient underwent C-section delivery on October 14, 2024 at 31 weeks 3 days gestation. After this, treatment with ATO was started at a dosage of 0.15 mg/kg/day (rounded up to 13 mg for curative intent). It was given from October 16, 2024 to June 23, 2025. Diamox 250 mg by mouth daily was started on October 25, 2024. Acyclovir 400 mg by mouth daily was started on October 23 for the risk of opportunistic infection. For the same reason, Micafungin 100 mg IV daily given the high risk of the QT interval on EKG prolongation with Diflucan.
The patient was discharged and received a right port-a-cath under ultrasound and fluoroscopic guidance about a month later. She underwent the following targeted therapy: ATO (Trisenox) as follows: cycles 1, 3, 5, 7: 0.15 mg/kg IV over 1 to 2 hours once per day on days 1 to 5, 8 to 12, 15 to 19, and 22 to 26. ATRA 40 mg/m2 PO twice per day on days 1 to 14, followed by 14 days of not taking the medication. She had a 28-day cycle for 7 cycles. Her last dose of ATO was June 23, 2025. Post-treatment bone marrow biopsy (Figures 6 and 7) showed a marked reduction in abnormal promyelocytes with a return toward normal trilineage hematopoiesis. CD34 and CD117 staining demonstrated rare residual blasts, confirming morphologic and immunophenotypic remission. She and her baby are both currently doing well.

H&E stain on second biopsy after treatment with ATRA 45 mg/m2 total daily (g BID) and ATO 0.15 mg/kg/day. This post-treatment bone marrow biopsy shows normalization of hematopoiesis with only scattered residual promyelocytes. H&E, hematoxylin and eosin; ATRA, all-trans retinoic acid; ATO, arsenic trioxide.

CD34 on second biopsy after treatment with ATRA 45 mg/m2 total daily (g BID) and ATO 0.15 mg/kg/day. This stain highlights rare blasts, consistent with morphologic remission. ATRA, all-trans retinoic acid; ATO, arsenic trioxide.
Discussion
This case describes a 31-year-old female who developed APML during pregnancy. The literature demonstrates that the unique physiology of this cancer, which is characterized by the PML-RARA fusion gene, leads to risks of coagulopathies and potentially severe maternal-fetal complications.12,13 The risk of coagulopathy, especially disseminated intravascular coagulation (DIC), is further increased during pregnancy. Because of this, there are recommendations to immediately implement supportive measures to manage coagulopathy, which can otherwise lead to early mortality in untreated patients. 14 The prognosis of APML has significantly improved with the use of ATRA and ATO, which have brought remission rates above 85% in non-pregnant patients and 89% in pregnant women. 13 Guidelines from the European LeukemiaNet recommend beginning ATRA as soon as APML is suspected, given its rapid action in stabilizing coagulopathies and reducing leukemic burden. 14 The decision to deliver the fetus at 31 weeks 3 days to initiate ATO therapy allowed for rapid treatment of the mother without exposing the fetus to the teratogenic effects of the drug. According to the literature and several case studies, the third trimester is the ideal time for delivery for this same reason. 15 This management strategy is consistent with the literature that recommends a tailored approach based on gestational age and maternal health. 16 Overall, the management of the patient aligns with the best practices observed in the literature, demonstrating the benefit of immediate treatment to stabilize maternal health without compromising fetal safety.
The incidence of APML is considered a rare event, as it occurs in roughly 1 in 75 000 to 100 000 pregnancies.13,17 The existing literature stresses the importance of rapidly diagnosing this condition to mitigate the risk of coagulopathy, which is highly characteristic of APML due to the PML-RARA fusion gene. This genetic mutation from a chromosomal translation disrupts myeloid differentiation and promotes the formation of Auer rods, which predisposes the patients to significant bleeding disorders and DIC, a major contributor to maternal morbidity and mortality.12,14 A systematic review by Santolaria et al analyzed 92 cases of APML during pregnancy, reporting a high complete remission rate of 89% when treated with the prompt initiation of ATRA in pregnancy along with chemotherapy post-delivery. This review also noted that outcomes were particularly favorable when treatment was timed to avoid fetal exposure, as seen in cases diagnosed in the third trimester. 13 Another review by Fei et al reported their analysis of 10 years of hematologic malignancies in pregnancy seen at their institution. Their findings reinforced the idea that rapid induction of APML-specific treatment was essential for remission, while delayed treatment was associated with a higher rate of adverse outcomes like intrauterine fetal demise and maternal mortality. 5
Several case reports within the literature underscore the importance of early diagnosis and treatment of pregnancy-associated APML. A study from Tianjin Medical University by Huiyang Li et al presented 2 cases of APML during pregnancy in which the significant difference in outcomes could be attributed to the highly variable differences in the timing of diagnosis. One patient presented with an intracranial hemorrhage secondary to delayed diagnosis and did not survive despite supportive measures. By contrast, the second patient, diagnosed promptly, achieved remission with ATRA therapy, resulting in a successful delivery and maternal recovery. 11
Our case also demonstrates the importance of an interdisciplinary approach to the management of APML during pregnancy. Guidelines given by European LeukemiaNet, as well as studies like the review by Brenner et al, highlight that hematology/oncology, obstetrics, and neonatology teams must collaborate closely to adequately address all the diseases associated with maternal and fetal risks. In addition, agents such as ATRA and ATO have complex potential side effects, including differentiation syndrome and cardiotoxicity, particularly QT interval prolongation that can precipitate significant ventricular arrhythmias such as torsades de pointes.14,17 Continuous cardiac monitoring and frequent electrolyte assessment are therefore essential, and multidisciplinary teams, including hematology, cardiology, and critical care, are required to manage these risks and ensure treatment safety. Overall, patients need routine prenatal screening, collaborative, interdisciplinary care, timing and initiation of treatment, and standardized guidelines.
This case provides several valuable clinical insights and implications for the future management of APML in pregnancy. The first takeaway is the importance of routine prenatal screening with CBCs and other hematological tests that allow for the early detection of hematological malignancies. The current literature, including the study by Barzilai et al, 16 emphasizes the need for proactive testing in pregnant patients with non-specific symptoms like fatigue, as the symptoms of APML may significantly overlap with normal pregnancy symptoms. Timely detection is crucial to improve outcomes, as early intervention significantly reduces maternal and fetal complications.
Another key implication from this case is the role of collaborative, interdisciplinary care. As discussed above, the management of APML in pregnancy requires coordinated input from multiple specialties, given the risks to the mother and child. The review by Dragana Milojkovic and Jane F. Apperley underscores the necessity of interdisciplinary collaboration in managing leukemia in pregnancy. They advocate for a well-integrated approach to address the complexities of using ATRA and ATO during pregnancy while monitoring APML-specific complications such as differentiation syndrome and QT prolongation. 15 This case exemplifies how interdisciplinary coordination leads to the most effective maternal and fetal care and favorable outcomes.
The timing of therapy initiation and delivery is a significant area of focus within this case. Studies, including the review by Brenner et al, show that achieving optimal outcomes in APML requires a delicate balance between maternal treatment and fetal safety. In this case, delivery at 31 weeks 3 days allowed for the administration of APML-specific therapies postpartum, protecting the fetus from teratogenic exposure while enabling timely maternal treatment. 5 Late second or third trimester deliveries provide higher maternal survival rates when compared to those of non-pregnant patients, as illustrated by studies in this review.13,17
Finally, this case contributes to the limited literature describing the treatment of APML during pregnancy. The literature review shows that there are no standardized guidelines for the treatment of this condition. The current recommendations rely on expert consensus and case reports of previous successful treatments, which demonstrate the need for larger prospective studies to establish evidence-based protocols. The systematic review by Santolaria et al 13 suggests that collaborative, multi-institutional research is essential to improving our understanding and management of APML in pregnancy.
Conclusion
This case underscores the critical role of close prenatal care as it allows for the identification of serious health conditions like APML early, which can significantly enhance maternal and fetal outcomes. The successful management of this patient case illustrates the value of a multidisciplinary, collaborative approach among the different healthcare teams, ensuring that both maternal treatment and fetal safety are prioritized. Furthermore, this case contributes to the growing body of literature advocating for the development of research-based, standardized guidelines to establish the best management of hematologic malignancies during pregnancy, ultimately improving care for future patients.
Footnotes
Author Note
Prior Presentation of Abstract Statement: Not applicable
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series
Consent to Participate
Verbal informed consent was obtained from the patient for their anonymized information to be included and published in this article.
Consent for Publication
The patient provided verbal consent for publication of the case.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed for this case.
