Abstract
Lung adenocarcinoma (LUAD) is the most common subtype of non-small-cell lung cancer and a leading cause of cancer-related mortality worldwide. Although it typically presents as a solitary pulmonary nodule or mass, it can rarely mimic interstitial lung disease, complicating diagnosis and delaying appropriate treatment. We report a 55-year-old male firefighter who presented with a 3-month history of persistent nonproductive cough, predominantly nocturnal, and recent onset of hoarseness. He denied hemoptysis, weight loss, or fever, and his past medical history included ischemic heart disease, hypertension, and a prior brief episode of cough. He was a heavy smoker. Initial laboratory investigations and bronchoalveolar lavage excluded eosinophilic pneumonia and drug-induced pneumonitis. Chest computed tomography revealed mediastinal lymphadenopathy and multiple left-sided pulmonary nodules. The patient underwent right video-assisted thoracoscopic surgery with wedge resection of the right upper lobe, mediastinal lymph node biopsy, and pleural biopsy. Histopathology confirmed invasive moderately differentiated LUAD, with mediastinal and pleural involvement. Immunohistochemistry showed positivity for CK7, CK20, and TTF-1 and negativity for CDX2. This case underscores the diagnostic challenge of LUAD presenting with atypical radiological features resembling pulmonary fibrosis and highlights the importance of integrating clinical, radiological, and pathological findings, with early tissue sampling critical when conventional therapy fails. Molecular testing and personalized therapy, including targeted agents and immunotherapy, are essential in guiding management and improving outcomes. Clinicians should maintain a high index of suspicion for malignancy in patients with unusual respiratory presentations to ensure timely diagnosis, accurate staging, and optimal therapeutic intervention.
Introduction
Lung cancer is the second most common malignancy in humans and the leading cause of cancer-related mortality globally. Adenocarcinoma has become the predominant histological subtype, representing nearly half of all lung cancer cases. 1 This type of cancer may be asymptomatic or present with a variety of respiratory and systemic symptoms, such as persistent cough, hemoptysis, dyspnea, unintentional weight loss, fatigue, anorexia, chest pain, hoarseness, and, in advanced stages, superior vena cava syndrome. 2 Typically, lung adenocarcinoma (LUAD) appears as a solitary pulmonary nodule (SPN) or a mass lesion, but it can occasionally present as diffuse parenchymal infiltrates resembling bacterial pneumonia. Treatment options are based on the stage and extent of the disease. Surgical resection is preferred for localized cases, while radiation therapy, with or without chemotherapy, may be considered for cases where surgery is not possible. 3
We present a case of LUAD that mimicked pulmonary fibrosis, based on clinical symptoms and radiographic findings. The diagnosis of invasive moderately differentiated LUAD was confirmed through histological examination.
This case has been reported in line with the CARE criteria. 4
Case Presentation
A 55-year-old male firefighter presented for follow-up evaluation of persistent dry cough and mediastinal lymphadenopathy. His cough had been present for 3 months, was nonproductive, occurred daily and predominantly at night, and frequently disturbed his sleep. He also reported hoarseness of voice for the preceding 15 days. He denied hemoptysis, night sweats, weight loss, dyspnea, orthopnea, fever, or any gastrointestinal or urinary symptoms. Multiple prior medical consultations provided only partial symptomatic relief.
The patient had a history of ischemic heart disease, status post-cardiac catheterization and stenting 2 years earlier, and hypertension. He also recalled a similar episode of persistent cough 5 years previously, which resolved after intravenous antibiotics. He was a heavy smoker, had no known allergies, and his medications included aspirin, clopidogrel, atorvastatin, bisoprolol, and amlodipine.
At the time of hospital admission, the initial differential diagnosis included cryptogenic organizing pneumonia, chronic eosinophilic pneumonia, drug-induced pneumonitis, and usual interstitial pneumonia. Laboratory investigations at that time demonstrated the following results (Table 1).
Laboratory Findings at Hospital Admission.
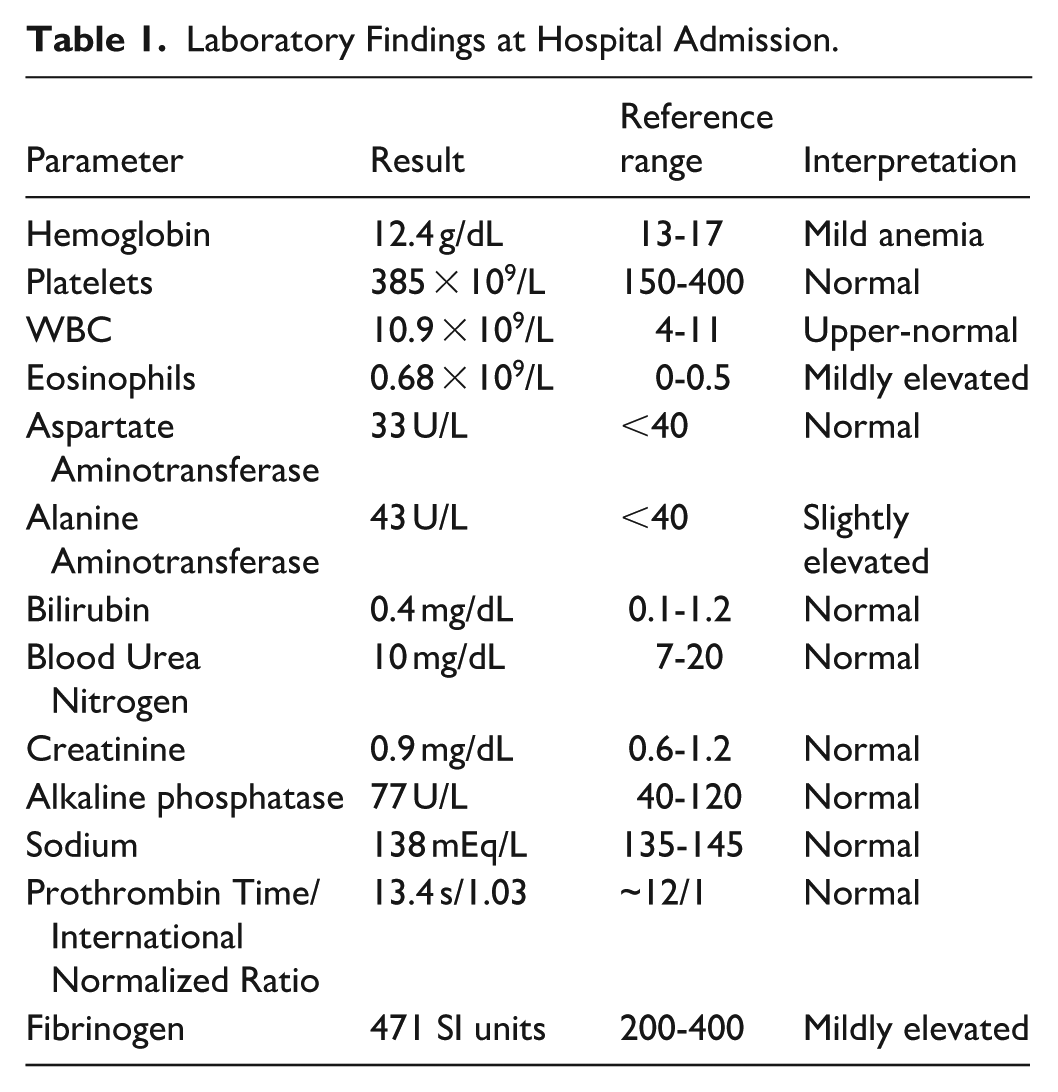
The absence of hypereosinophilia in both peripheral blood and bronchoalveolar lavage (BAL) fluid, along with the lack of response to corticosteroids and antibiotics, excluded chronic eosinophilic pneumonia and cryptogenic organizing pneumonia. The absence of a temporal relationship with medication use argued against drug-induced pneumonitis, while the lack of radiologic evidence of basal fibrosis made usual interstitial pneumonia less likely.
High-resolution chest computed tomography (CT) performed during evaluation revealed multiple bilateral pulmonary nodules, most prominent in the upper lobes, including a cavitating nodule in the posterior segment of the left lower lobe (17 × 15 mm) and a subpleural lesion with peripheral ground-glass changes in the apicoposterior segment of the left upper lobe (17 × 25 mm). Two additional partially solid nodules were identified in the anterior segment of the left upper lobe, the largest measuring 8 × 11 mm. Pathological lymphadenopathy was present, including a necrotic right paratracheal node (station 4R, 12 mm short axis), suspicious left lower cervical nodes (largest 11 × 15 mm), and bilateral axillary nodes (largest 10 mm on the left). Additional findings included mild right pleural effusion, coarse interlobular septal thickening (most evident in the right upper lobe), and a right adrenal hypodense lesion (25 × 19 mm, pre-contrast density 20 HU) with diffuse thickening of the left adrenal gland (Figure 1). Echocardiography prior to surgery showed preserved cardiac function (EF 63%) without valvular or pericardial abnormalities. At this stage, metastatic multifocal LUAD was the leading diagnosis, although granulomatosis with polyangiitis, sarcoidosis, and cavitating lung infections (tuberculosis, fungal disease) were considered, but the absence of systemic features and lack of exposure history made these etiologies unlikely.

High-resolution chest CT shows findings of left upper lobe subpleural lesion with peripheral ground glass changes, left upper lobe 2 partially solid nodules, and multiple enlarged mediastinal lymph nodes. CT, computed tomography.
The patient subsequently underwent right video-assisted thoracoscopic surgery (VATS), which included wedge resection of the right upper lobe measuring 7 × 1.5 cm, mediastinal lymph node biopsy measuring 0.8 × 0.8 cm, and pleural biopsy measuring 0.6 × 0.6 cm. The procedure was completed without complications. Postoperatively, he was admitted to the intensive care unit for close observation, maintained nil per os until recovery, and chest drainage was adequate without evidence of air leaks.
He improved clinically and was transferred to the ward the following day. He remained stable, awake, afebrile, tolerating oral intake, ambulating, and continued chest physiotherapy with incentive spirometry. The chest tube was removed after radiographic confirmation of satisfactory lung expansion (Figure 2), and he was discharged in good condition with follow-up arranged with oncology and pulmonology services.

X-ray shows right lung interstitial infiltrate with a blunted costophrenic angle.
Histopathological examination of the wedge resection specimen from the right upper lobe revealed invasive moderately differentiated LUAD (Figure 3). The mediastinal lymph node biopsy confirmed metastatic adenocarcinoma, and the pleural biopsy also demonstrated malignant involvement. Immunohistochemistry showed tumor cells positive for CK7, CK20, and TTF-1, and negative for CDX2, confirming pulmonary origin (Figure 4). According to the AJCC ninth edition TNM classification, the clinical stage was Stage IIB (T1aN2aM0) LUAD.

The tumor is composed of malignant glands and micropapillary structures (Hematoxylin and Eosin stain, 20×).

The tumor cells show nuclear positivity for TTF-1 immunostain.
At early postoperative follow-up, the patient reported occasional shortness of breath and easy fatigue, although he denied cough, fever, or hemoptysis. On physical examination, he was hemodynamically stable with oxygen saturation of 96% on room air. Chest auscultation revealed good bilateral air entry without wheezes or crackles, and the surgical wound appeared healthy with no signs of infection or dehiscence. The chest X-ray at this time was unremarkable (Figure 5). He was referred to the oncology service for definitive management and continued follow-up with pulmonology.

X-ray shows bilateral interstitial infiltrate with hilar lymphadenopathy.
Discussion
Lung cancer is a significant global health concern, accounting for a substantial percentage of new cancer cases (12.4%) and cancer-related deaths (19.4%).5,6 According to the World Health Organization, it remains the leading cause of cancer-related mortality worldwide, largely due to late-stage diagnosis and limited access to curative treatment in many regions. Lung cancers are classified into 2 main histological types: small-cell lung cancer (SCLC) and non-SCLC (NSCLC). 7 SCLCs are aggressive tumors primarily caused by smoking, while NSCLC comprises ~85% of primary lung cancers and includes subtypes such as lung squamous cell carcinoma, LUAD, bronchial carcinoid tumor, and large-cell carcinoma. Among the NSCLC subtypes, LUAD is the most common primary lung tumor and frequently occurs in non-smoking females, accounting for 14% of cases.6,8 It is important to note that LUAD is a heterogeneous group of malignancies with diverse pathological and radiological presentations. Studies have shown that the predominant growth pattern (solid, acinar, lepidic, or papillary) observed in LUAD differs in radiological presentation and is associated with variations in treatment outcomes and prognosis. 9 The radiological appearance of peripheral LUADs can range from ground glass nodules to solid mass lesions on CT, reflecting the histological heterogeneity of the tumors. 10 This heterogeneity explains, in part, why LUAD can mimic interstitial lung disease (ILD), adding significant diagnostic challenges.
The clinical manifestation of adenocarcinoma exhibits a spectrum of presentations, spanning from asymptomatic cases that remain undetected until disease advancement to symptomatic cases presenting with a constellation of respiratory and systemic symptoms. These can include persistent cough, hemoptysis, dyspnea, wheezing, unintentional weight loss, fatigue, anorexia, and chest pain. 2 In advanced stages, patients may experience hoarseness and superior vena cava syndrome, which is characterized by swelling in the face and neck due to obstruction of venous return. 11 In rare cases, patients may initially present with diffuse infiltrates on imaging, misleading clinicians toward infectious or inflammatory etiologies, as happened in this case.
The pathophysiology of LUAD involves genetic alterations induced by exposure to carcinogens, including tobacco smoke, environmental pollutants, and occupational hazards. 2 These exposures can lead to DNA damage and chromosomal rearrangements, involving genes such as KRAS, epidermal growth factor receptor (EGFR), ROS1, and ALK, which contribute to the malignant transformation of lung epithelial cells.12,13 Typically, LUAD presents as a SPN or a mass lesion. It is worth noting that neoplastic processes like lymphoma and leukemia are more likely to present as diffuse pulmonary infiltrates, causing acute respiratory distress syndrome, rather than primary lung carcinoma. 14 Therefore, the diffuse interstitial presentation of adenocarcinoma in our patient is an exceptional diagnostic pitfall.
On first impression, the imaging findings in this case suggest several potential diagnoses, such as cryptogenic organizing pneumonia, chronic eosinophilic pneumonia, drug-induced pneumonitis, and usual interstitial pneumonia. However, these conditions can be ruled out based on specific criteria. The absence of hypereosinophilia in the blood and BAL fluid, as well as the lack of response to corticosteroids and antibiotics, helps exclude chronic eosinophilic pneumonia and drug-induced pneumonitis. Furthermore, the subacute progression without clear infectious markers reduced the likelihood of bacterial pneumonia or atypical infections.
Typically, this particular cancer presents as peripheral nodules, although in rare cases, it can manifest as an interstitial pattern, as observed in our case. The differential diagnosis includes lymphangitic carcinomatosis, which requires the presence of neoplastic cells in perivascular, peribronchial, and lymphatic spaces. In addition, there have been reported cases of adenocarcinoma masquerading as pneumonia, lung fibrosis with honeycombing, and diffuse inflammatory ILD.15-18 Metastatic tumors, such as melanoma, can also exhibit a diffuse interstitial pneumonia-like pattern. In situations where the diagnosis remains unclear despite optimal medical therapy for the presumed diagnosis, immunohistochemical studies following biopsy are usually recommended. 19 This underscores the importance of pursuing tissue diagnosis early when clinical and radiological findings are discordant.
While surgical lung biopsy is considered the gold standard for diagnosing ILD, it is an invasive procedure and not typically preferred. However, with the advancement of VATS, surgical lung biopsy can now be performed safely. A study by Kayatta et al showed that VATS biopsy provided an accurate, specific diagnosis of ILD in 88% of cases compared to only 15% without a surgical lung biopsy. 20 VATS biopsy carries a low mortality rate of 0% to 2% in low-risk patients and 0% to 11% in patients with unstratified risk. 20 In our patient, the decision to proceed with a biopsy was crucial for establishing the diagnosis, ultimately guiding treatment decisions.
The selection of treatment is determined by the stage and extent of the disease. For localized forms, surgical resection may be recommended, while in cases where surgical resection is not feasible, radiation therapy with or without chemotherapy can be considered. 3 It is worth noting that NSCLC cases with an EGFR gene mutation may benefit from treatment with tyrosine kinase inhibitors. 21 This targeted therapy approach can specifically target the mutated EGFR and potentially improve outcomes for these patients.
Conclusion
This case highlights the importance of considering LUAD in patients with atypical clinical and radiological features, particularly when these mimic pulmonary fibrosis. The patient’s persistent dry cough and mediastinal lymphadenopathy demonstrate the need for a systematic and vigilant diagnostic approach, with histological and immunohistochemical analysis proving essential for definitive diagnosis and resolving uncertainty. It underscores the value of integrating clinical, radiological, and pathological findings when conventional management of presumed benign or inflammatory conditions fails, supporting early biopsy to avoid delays. Molecular testing in NSCLC is increasingly critical to guide targeted therapy and immunotherapy, which have improved outcomes in advanced disease. Clinicians should maintain a high index of suspicion for LUAD in patients with unusual respiratory presentations and pursue comprehensive evaluation when the clinical course deviates from expectations. Awareness of atypical presentations, timely tissue diagnosis, and incorporation of personalized molecular-guided therapy can optimize management and improve patient outcomes.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
