Abstract
Large granular lymphocytic (LGL) leukemia is a rare lymphoproliferative disorder characterized by cytopenia and autoimmune manifestations. While cytopenia and rheumatoid arthritis (RA) are common, transfusion-dependent anemia (TDA) without RA or other autoimmune disease is less frequently observed. We present a 79-year-old male with T-cell LGL leukemia, exhibiting isolated TDA without an autoimmune disease. Anemia remained refractory to erythropoietin and methotrexate, requiring transfusions, but showed a response to cyclophosphamide after 3 months of therapy. This case highlights an uncommon presentation of LGL leukemia, emphasizing the importance of considering LGL leukemia in the differential diagnosis of TDA.
Keywords
Introduction
Large granular lymphocytic (LGL) leukemia represents an uncommon, chronic lymphoproliferative condition characterized by the expansion of cytotoxic lymphocytes, which may arise from either T-cell or natural killer (NK)-cell lineages. In the fifth edition of the World Health Organization classification, 1 mature T-cell and NK-cell leukemias are categorized into 6 distinct molecular entities: T-prolymphocytic leukemia, T-cell LGL (T-LGL) leukemia, NK-cell LGL leukemia, adult T-cell leukemia/lymphoma, Sézary syndrome, and aggressive NK-cell leukemia.
The clinical presentation of T-LGL leukemia most often involves cytopenia, particularly neutropenia, which is reported in about 70% to 85% of patients and occurs more frequently in T-LGL than in its NK-cell counterpart. 2 Autoimmune manifestations are seen in roughly one-third of cases, with rheumatoid arthritis (RA) representing the most frequent association. 3 Anemia is another common finding, usually mild to moderate in severity, while only 10% to 20% of patients require regular transfusion support. 4 Data from the Epic Cosmos database indicate that fewer than 1% of individuals with LGL develop the combination of transfusion-dependent anemia (TDA) and autoimmune disease. We describe here a patient with T-LGL leukemia who presented with TDA in the absence of any autoimmune manifestations.
Case Presentation
A 79-year-old male was referred to the hematology and oncology clinic for evaluation of anemia with chronic constipation for the past 6 months. His past surgical history includes benign thyroid surgery 20 years ago and a left knee replacement 3 to 4 months ago.
The patient recently was hospitalized due to constipation and was found to have a hemoglobin level of 7 g/dL. He reports a poor appetite over the past year but denies any active bleeding or unintentional weight loss. He has a 15-year history of heavy alcohol consumption but has never smoked. His family history is notable for pancreatic cancer in his brother, diagnosed at age 65.
He underwent a comprehensive gastrointestinal evaluation, including a colonoscopy which was negative. Multiple complete blood counts, iron studies, immunoglobulin assessments, and tumor marker evaluations were performed at regular intervals. The results indicate a progressive decline in hemoglobin levels, reflecting persistent, and worsening anemia that eventually led to transfusion dependence. White blood cell counts remained within normal limits throughout. Blood smear showed an increased number of LGLs and normochromic, normocytic red blood cells with marked reduced density (Figure 1).

Peripheral blood smear and flow cytometry findings at initial presentation and 4-month follow-up. (A) Peripheral blood smear showed normocytic red blood cells with decreased density accompanied by thrombocytosis and lymphocytosis. (B) Flow cytometric analysis showed no evidence of an abnormal B-cell or myeloid population but demonstrated an increased presence of T-cell large granular lymphocytes, suggesting an underlying clonal expansion. (C) A repeat peripheral blood smear at 4-month follow-up showed a further increase in large granular lymphocytes with persistent normochromic, normocytic anemia. (D) Follow-up flow cytometry revealed a progressive increase in atypical large granular lymphocytes, consistent with ongoing clonal expansion.
Peripheral blood testing was initially performed to assess for a circulating clonal T-LGL population, and the findings supported the possibility of T-LGL leukemia. T cell population suggests CD8+ predominance. CD8+ T cells showed slightly decreased expression of CD7, were CD57+, and partially CD16+. Additional T-cell receptor (TCR) panel demonstrated a TRBC1−/TRBC1+ ratio of 202:1, which is indicative of a clonal T-cell population. A PCR-based assay for TCR gamma gene rearrangement also confirmed T-cell clonality with detected a clonal T-cell population. Bone marrow aspiration was performed for further evaluation which revealed LGL infiltrated bone marrow and decreased erythroid activity, a clonal T-LGL population, comprising 25% of nucleated cells, with an immunophenotype consistent with T-LGL leukemia (Figure 2). Flow cytometry confirmed a CD4/CD8 ratio of 0.1, with the aberrant T- cell population expressing CD2, CD3, CD5 (dim), CD7 (dim), CD8, CD57, and dim CD16, along with restricted negative TRBC1 expression. Genetic testing detected TCR gamma and beta gene rearrangements, further supporting the presence of a clonal T-cell population (Figure 1). However, no pathogenic alterations were identified in genes frequently associated with myeloid malignancies, including CALR, FLT3, IDH1, IDH2, JAK2, MPL, NPM1, and TP53. The variant male karyotype with loss of Y chromosome, observed in 35% of cells, is likely an age-related phenomenon with no known clinical significance.

LGL infiltration in bone marrow aspirate. (A) The bone marrow aspirate reveals increased infiltration of small lymphocytes (arrows) alongside maturing trilineage hematopoiesis (40×). (B) The LGL lymphocytes (arrows) exhibit multiple cytoplasmic granules (100×). LGL, large granular lymphocyte.
There was no evidence of myelodysplasia, as granulocytes and monocytes showed normal maturation without dysplastic features. CD34+ progenitor cells were present at a low level (1.3%), and plasma cells were not increased. The autoimmune workup was negative, including antinuclear antibody, direct antiglobulin test, anti-RNP (anti-ribonucleoprotein) antibody, rheumatoid factor, antimitochondrial antibody, anticentromere antibody, thyroid peroxidase antibody, and thyroglobulin antibody. Immunoglobulin levels were within normal limits as IgG 913 mg/dL, IgA 84 mg/dL, and IgM 132 mg/dL. Additionally, the infectious disease panel including hepatitis B surface antigen, hepatitis C antibody, HIV, and HTLV I/II were all negative. Overall, the findings are consistent with T-LGL leukemia without evidence of associated autoimmune disorders. PET/CT (Positron Emission Tomography/Com) showed no evidence of nodal or abnormal osseous activity, with no definite signs of active disease identified. The patient was started on epoetin alfa (5 times/month for 12 cycles) for anemia, but there was no significant improvement. Consequently, methotrexate 50 mg twice weekly without effects then followed by cyclophosphamide 100 mg orally once daily was prescribed as the next line of treatment. He subsequently responded with Hb increased to 11 g/dL and achieved transfusion independence after 3 months of cyclophosphamide treatment. Figure 3 shows the correlation between the hemoglobin and treatment.
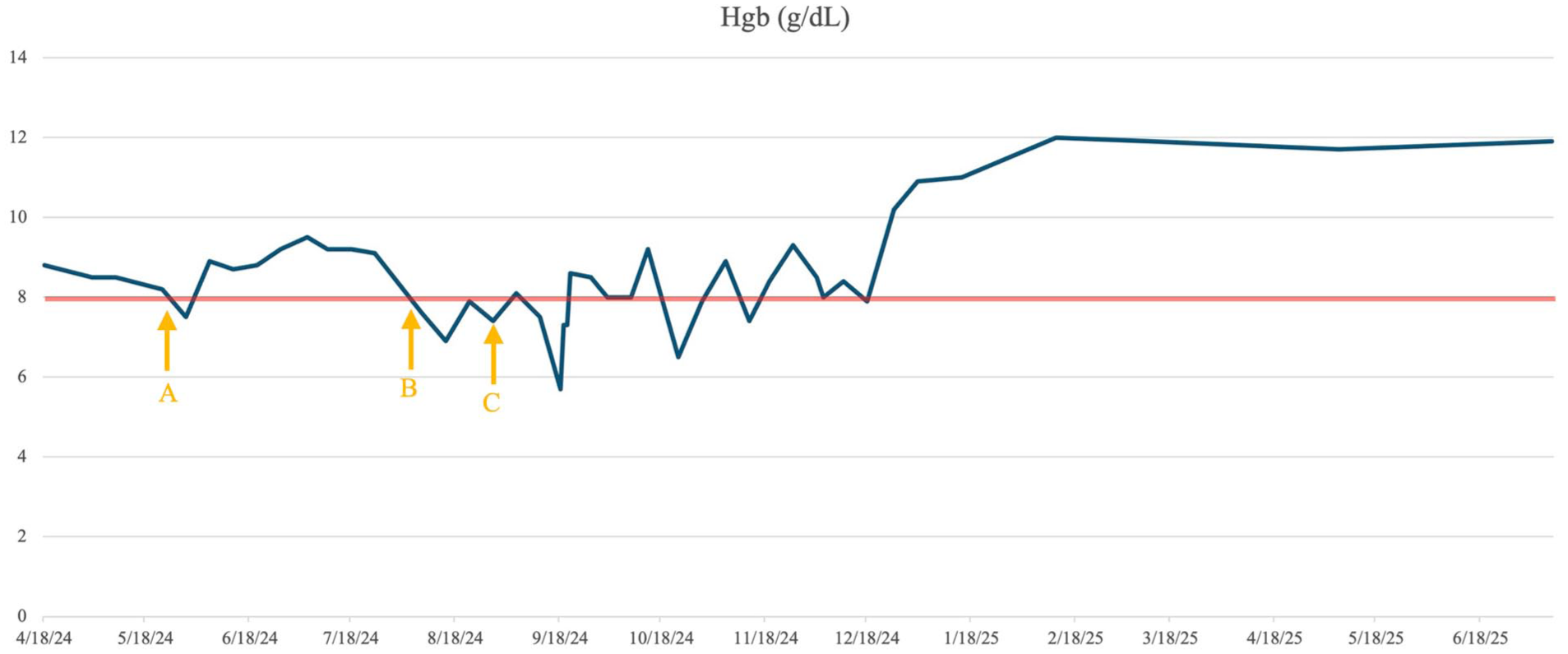
Hemoglobin trends in relation to treatment. The red line indicates the transfusion threshold. (A) Weekly Procrit initiated on May 23, 2024. (B) Methotrexate 50 mg administered twice weekly, starting August 8, 2024. (C) Cyclophosphamide 100 mg administered daily, starting September 5, 2024.
Discussion
LGL leukemia (LGLL) is a rare chronic lymphoproliferative disorder characterized by the clonal expansion of cytotoxic T cells (T-LGLL) or NK cells (NK-LGLL), accounting for 85% and 15% of cases, respectively. 2 The disease is frequently associated with hematologic malignancies, including B-cell lymphoma, multiple myeloma, and myeloid neoplasms, as well as autoimmune disorders, particularly RA. 5
Roughly 30% of individuals with LGL leukemia are diagnosed incidentally without symptoms. In symptomatic patients presented with neutropenia, anemia, or autoimmune disorders are the most common findings. Severe neutropenia (absolute neutrophil count [ANC] <0.5 × 109/L) is reported in 16% to 48% of cases, while moderate neutropenia (ANC >0.5 × 109/L) occurs in 48% to 80%. 6 RA described in 25% to 33% of patients.2,3 Anemia is also frequent, with transfusion dependence documented in about 10% to 30% of cases. 7 In a French study of 229 patients, 2 24% were noted to have anemia, which only 7% required transfusion. Other studies8-11 indicate that ~44% of T-LGL leukemia patients experience anemia, with nearly one-quarter showing hemoglobin levels below 8 g/dL. A study 12 demonstrated geographic variation, with anemia more often observed in Asian patients, whereas Western patients more commonly presented with neutropenia, splenomegaly, RA, or recurrent infections.
The coexistence of TDA and autoimmune disorders in patients with LGL leukemia is rare. In the United States, we used data from the Epic Cosmos database (2020-2025), 13 a dataset developed in collaboration with a network of health systems that use the Epic electronic health record platform. Cosmos integrates both inpatient and outpatient medical records into a unified longitudinal health record, even as patients transition between health systems. Using ICD-10 codes documented at the time of diagnosis, we identified patients diagnosed with LGL leukemia. It showed that 16% of patients with LGL leukemia had a hematocrit below 21%, meeting the threshold for transfusion. Additionally, 24% were diagnosed with RA or another specified autoimmune disorder. And only <0.4% (10 cases or fewer) of LGL leukemia patients had both TDA and an autoimmune disorder or RA, while another study reported just 1% and 0.4% of patients with both conditions.5,12
Isolated TDA, defined as marked anemia occurring in the absence of neutropenia or thrombocytopenia, represents an uncommon and underreported manifestation of LGL leukemia. In a cohort of 244 patients, Salama et al 5 noted isolated anemia in 14% of cases, with only 8% requiring transfusion. Affected individuals were predominantly male, tended to have lower hemoglobin levels and higher mean corpuscular volumes, and were less frequently associated with RA which closely align with the presentation of our patient. In our case, the patient demonstrated isolated TDA without coexisting autoimmune disease. A review of prior studies suggests that this pattern occurs in ~7% of individuals with LGL leukemia. It emphasizes the need to include LGL leukemia in the diagnostic evaluation of patients presenting with unexplained severe anemia. Clinical details of our case are provided in Table 1, together with comparative data from more than 50 previously reported cases.2,5,10-15
Clinicopathologic Features of Transfusion-Dependent Anemia and Autoimmune Diseases in LGL Leukemia Patients From Literatures Review and the Epic Cosmos Database.

Abbreviations: AID, autoimmune diseases; EHR, electronic health record; LGL, large granular lymphocyte; PRCA, pure red cell aplasia; RA, rheumatoid arthritis; TDA, transfusion-dependent anemia.
Previous studies identified a total of 88 Asian patients across 20 articles.
Epic Cosmos is the largest database of EHR patient information, integrating data from multiple health systems. ICD-10 codes are used at admission to identify and filter target patients.
These patients have isolated TDA, without associated neutropenia or thrombocytosis.
We include a hematocrit level of <21% as a TDA patient. And only showed 7% of LGL patients with isolated TDA.
Standard therapy for LGL leukemia is immunosuppression, though recommendations are largely based on small retrospective studies. Most clinical data are derived from the use of low-dose methotrexate, cyclophosphamide, and cyclosporine A as monotherapy. 6 Treatment should be considered for patients with severe neutropenia (absolute neutrophil count [ANC] < 0.5 × 10⁹/L), moderate neutropenia (ANC > 0.5 × 10⁹/L) accompanied by recurrent infections, anemia that is symptomatic or requires transfusion support, or autoimmune diseases that warrant medical intervention. 16 For patients with anemia, erythropoietin may be offered as supportive therapy. Reports of erythropoietin use in LGL leukemia are limited and outcomes have generally been unsatisfactory. 2 Cyclophosphamide is frequently preferred for patients with prominent anemia, particularly those with pure red cell aplasia. 7 Retrospective data have shown that relapsed T-LGL leukemia patients treated with cyclophosphamide can achieve durable remissions, with sustained responses observed during long-term follow-up. 17 Additional studies from the same group reported improved clinical outcomes when cyclophosphamide was used as a second-line agent following methotrexate failure. 18
Conclusion
T-LGL leukemia typically presents with neutropenia and autoimmune disorders, while isolated TDA without associated auto-immune disorders is rare. Our case highlights this uncommon presentation, emphasizing the need for considering LGL leukemia in unexplained TDA cases. Treatment includes immunosuppressive agents, with cyclophosphamide showing efficacy in transfusion-dependent cases. Early recognition and intervention are crucial for improving outcomes, particularly in patients without classic LGL leukemia features such as neutropenia or autoimmune conditions.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Brookdale Hospital Research Fund.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jen C. wang received research grant from Kartos incorporation, all others have no conflict interests with the pharmaceutical companies.
