Abstract
Bronchus-associated lymphoid tissue (BALT) lymphoma is a rare subtype of extra-nodal marginal zone B-cell lymphomas, accounting for less than 5% of cases. It is typically indolent and often discovered incidentally through imaging studies performed for unrelated reasons. We present the case of a 72-year-old male with a known history of thoracic aortic dilatation, who was found to have a spiculated right upper lobe (RUL) pulmonary nodule on surveillance imaging. He remained asymptomatic, with no constitutional or respiratory symptoms. Positron emission tomography imaging demonstrated mildly increased uptake (standardized uptake value 2.4) of the RUL nodule, and no mediastinal or hilar lymphadenopathy. Bronchoscopic biopsy with histopathology and immunohistochemistry revealed a low-grade B-cell non-Hodgkin’s lymphoma, with CD20 and BCL2 positivity. Flow cytometry confirmed monoclonal B-cell proliferation, and fluorescence in situ hybridization analysis identified the BIRC3-MALT1 translocation, consistent with BALT lymphoma. The patient was staged as IE, and a decision was made for conservative management with active surveillance, given the localized and asymptomatic nature of his disease. While treatment options may include surgery, radiotherapy, immunotherapy, or chemotherapy, there is no consensus on the optimal approach due to the rarity and heterogeneous presentation of this lymphoma. In many early-stage cases without symptoms, a “watch and wait” strategy is reasonable. This case underscores the importance of including BALT lymphoma in the differential diagnosis of incidental pulmonary nodules and highlights the need for greater awareness and research to establish standardized treatment protocols, with each case adding valuable insight to guide future management strategies.
Keywords
Introduction
Marginal zone lymphomas (MZL) are a heterogeneous group of indolent B-cell non-Hodgkin lymphomas classified into extra-nodal MZL (EMZL) of mucosa-associated lymphoid tissue (MALT), splenic MZL, and nodal MZL. Among these, EMZL of MALT lymphoma is further subclassified by site of origin, with gastric involvement being most common. Bronchus-associated lymphoid tissue (BALT) lymphoma represents a rare form, accounting for less than 5% of MALT lymphomas. Despite its rarity, BALT lymphoma is the most common histologic subtype (77%-87%) of primary pulmonary lymphomas (PPL), which themselves are uncommon, constituting only 0.5% to 1% of all pulmonary tumors. 1
The etiology of BALT lymphoma remains poorly understood but is thought to involve chronic antigenic stimulation due to infections, autoimmune diseases, or environmental exposures. Clinically, it often follows an indolent course and is typically identified incidentally on imaging studies. Histopathologic evaluation with immunohistochemistry and molecular testing, including fluorescence in situ hybridization (FISH), is essential for diagnosis. Management strategies vary based on disease burden and patient symptoms, ranging from active surveillance in asymptomatic cases to radiotherapy or systemic therapy in symptomatic or advanced disease. We present a rare case of an asymptomatic, nonsmoking patient with an incidentally discovered pulmonary nodule, subsequently diagnosed as BALT lymphoma on tissue biopsy and managed with a conservative surveillance approach.
Case Presentation
A 72-year-old male with a past medical history notable for thoracic aortic dilatation was referred to the pulmonology clinic for further evaluation of an incidental pulmonary nodule. The nodule was identified on a follow-up computed tomography angiography (CTA) performed to monitor the progression of his aortic pathology. Imaging revealed a 1.7 cm spiculated nodule located in the right upper lobe (RUL) of the lung (Figure 1). The patient was entirely asymptomatic at the time of discovery, denying any complaints of dyspnea, cough, hemoptysis, chest pain, fever, night sweats, weight loss, or recent respiratory illness. He also reported no history of smoking or significant environmental exposures.
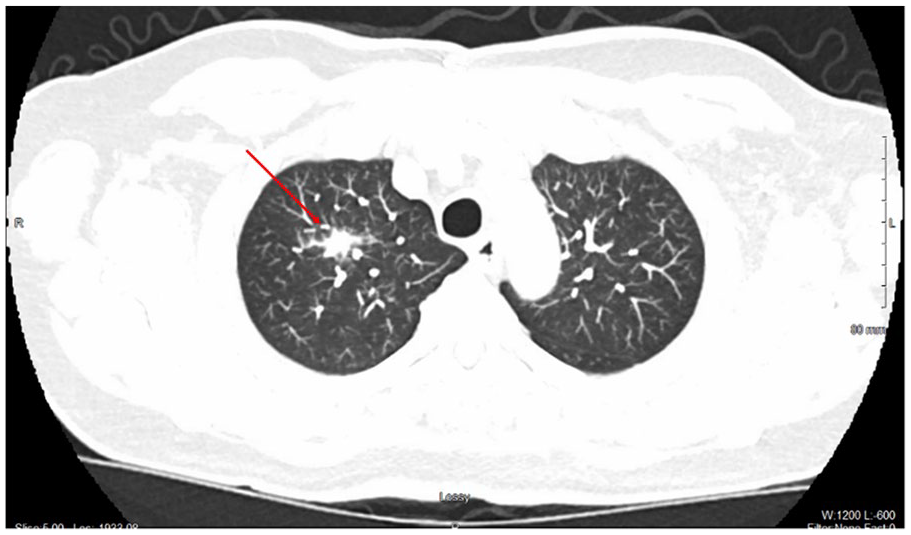
CTA identifying a 1.7 cm right upper lobe nodule with spiculated margin.
Based on the Mayo Clinic malignancy risk calculator, 2 the pre-test probability of malignancy for the RUL nodule was estimated at 49.5%, placing it in the intermediate-risk category. Given this risk stratification, a positron emission tomography (PET) scan was pursued to further characterize the lesion. The PET scan demonstrated mild fluorodeoxyglucose uptake in the RUL nodule with a maximum standardized uptake value (SUV) of 2.4, without evidence of hypermetabolic hilar or mediastinal lymphadenopathy (Figure 2). To obtain tissue for diagnosis, the patient underwent a robotic-assisted bronchoscopy with bronchoalveolar lavage and transbronchial biopsy of the RUL nodule. An endobronchial ultrasound was performed concurrently, which did not reveal any enlarged lymph nodes warranting transbronchial needle aspiration.

PET scan revealing a right upper lobe nodule with an SUV of 2.4.
Comprehensive infectious workup, including testing for acid-fast bacilli, fungal, viral, and respiratory pathogens, as well as specific antigens such as histoplasma and legionella, was negative. Histopathological analysis of the transbronchial biopsy demonstrated atypical lymphoid proliferation. Immunohistochemical staining revealed a predominance of CD20 and BCL2-positive B-cells, consistent with low-grade marginal zone B-cell non-Hodgkin lymphoma (Figure 3). Flow cytometry confirmed monoclonal B-cell proliferation with positive markers for CD19, CD20, CD38, CD45, and lambda light chain, while CD5 and CD10 were negative. FISH analysis identified a BIRC3-MALT1 (11;18) gene fusion, confirming the diagnosis of BALT lymphoma. Given the localized nature of the disease (stage IE) and the patient’s asymptomatic status, a conservative management strategy with active surveillance was initiated.

Histopathologic examination. (A) Nodular lymphoid aggregate centered on bronchiole (arrow) with extension into alveoli (*). (B and C) Lymphoid cells are aberrant with overwhelming CD20-positive B-cells (B) over CD3-positive T-cells (C) phenotype. (D) Aberrant BCL-2 positive protein expression.
Discussion
The pathogenesis of BALT lymphoma is not well established, but similar to other MALT lymphomas, it is linked to chronic antigenic stimulation. In healthy adults, BALT is generally absent or minimally present in the bronchial mucosa. However, under chronic inflammatory conditions such as smoking, autoimmune diseases, or recurrent pulmonary infections, BALT can be induced. 3 This acquired lymphoid tissue becomes a reservoir for immune cell activation, setting the stage for lymphoproliferative disorders. Chronic antigenic stimulation, particularly from autoimmune conditions like Sjögren’s syndrome and rheumatoid arthritis, has been strongly associated with pulmonary MALT lymphomas, suggesting an antigen-driven lymphomagenesis pathway.4,5 Persistent antigenic drive promotes polyclonal B-cell proliferation, which can evolve into a monoclonal neoplastic process due to genetic hits.
MALT lymphomas are associated with several genetic abnormalities, with the most common 1 being t(11;18) (q21;q21). It causes fusion of the BIRC3 (also called API2) gene and the MALT1 gene, leading to constant activation of the NF-κB pathway and autonomous tumor growth. 5 No obvious etiology is identified in many cases. 6
The culmination of chronic immune stimulation, B-cell clonal expansion, and oncogenic mutations results in the formation of malignant lymphoid aggregates within the bronchial mucosa. These can form lymphoepithelial lesions, infiltrate alveolar structures, and present radiographically as pulmonary nodules or infiltrates. In our patient, who had no history of smoking or respiratory symptoms, an autoimmune workup including antinuclear antibodies, anti-Ro/SSA, anti-La/SSB, rheumatoid factor, and anti-cyclic citrullinated peptide antibodies was negative, ruling out an autoimmune etiology. Genetic testing for chromosomal translocations was not performed.
BALT lymphomas typically affect adults, with a median age of diagnosis around 50 to 60 years, with no significant gender predilection. 7 Most cases are asymptomatic and discovered incidentally on chest radiography. Occasional patients can present with nonspecific symptomatology, including dyspnea, cough, chest pain, or recurrent pulmonary infections. Less than a quarter of the patients have constitutional symptoms such as fever, night sweats, and weight loss. 8 Radiographically, BALT lymphoma may show solitary nodules (55%), solitary, or multiple localized areas of consolidation (55%), masses (50%), reticulation (10%), and bilateral ground-glass opacities (25%), especially in the lower lobes. 9 Significant mediastinal lymphadenopathy may be seen in about 15% of cases. 9 BALT lymphomas are typically located along the broncho-vascular bundle. 10
Definitive diagnosis is established by histopathological examination of tissue biopsy obtained by transbronchial, CT-guided, or open thoracotomy approach. Immunohistochemical studies showed expression of CD20 B-cell phenotype with the absence of CD5, CD10, CD23, cyclin D1, which helps in differentiating from other B-cell lymphomas such as mantle cell or follicular lymphoma. 11 Molecular studies with FISH testing can help identify the gene rearrangements. Additional investigations, like gastroscopy, should be taken into consideration for the possible involvement of other organ systems, to rule out lymph node lymphoma or MALT lymphoma dissemination. 12
Various treatment options are available, including surgical excision, surgery followed by radiotherapy, and chemotherapy. However, the optimal treatment strategy remains unclear due to the lack of prospective data. Treatment is individualized based on the patient’s symptoms and disease burden. Staging is based on the Lugano classification system (Table 1). 13
Lugano Classification System.

Asymptomatic patients with BALT lymphoma can be safely monitored without immediate treatment, following a “watch and wait” approach. 12 This is especially appropriate in cases with indolent behavior and low tumor burden.
Limited-stage disease is often managed with radiotherapy, which has shown excellent local control rates exceeding 90%. While surgery was previously used, it is now rarely performed unless it has already been done for diagnostic purposes. 14 Radiation therapy remains highly effective, with typical doses around 25 to 35 Gy. 12 For patients who are not candidates for radiation, due to pulmonary function limitations or multifocal disease, monotherapy with rituximab, an anti-CD20 monoclonal antibody, is a well-tolerated alternative.
Symptomatic or advanced cases warrant systemic therapy with rituximab alone or in combination with cyclophosphamide, vincristine, and prednisolone. 15 Various combinations of systemic treatments such as bendamustine with rituximab, lenalidomide with rituximab, with better disease control and safety profiles, are in various phases of clinical trials.16,17 In select cases, antibiotics may be considered if there is a suspected infectious trigger, although this is more established in gastric MALT lymphoma. 18 BALT lymphomas have an excellent prognosis with an overall survival rate of 80% to 90%, with a median survival of over 10 years. 1
Conclusion
BALT lymphoma is a rare and often under-recognized subtype of marginal zone B-cell lymphomas, typically identified incidentally due to its indolent course and nonspecific clinical presentation. Its slow progression poses challenges to conducting long-term prospective studies, resulting in significant gaps in the evidence guiding its optimal management. Although novel therapies, including targeted agents and immunomodulatory treatments being investigated to improve disease control and reduce treatment-related toxicity, standardized guidelines remain elusive. As such, each reported case plays a pivotal role in expanding our understanding of this uncommon malignancy and contributes meaningfully to the development of future diagnostic and therapeutic strategies.
Footnotes
Acknowledgements
None.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
