Abstract
Influenza infection is known to induce significant immunosuppression, particularly in vulnerable populations, but its impact can be exacerbated by underlying immunodeficiencies. This case report describes the rapid deterioration of a previously healthy 30-year-old male who developed acute respiratory distress syndrome and septic shock due to methicillin-resistant Staphylococcus aureus pneumonia and bacteremia following Influenza A infection. The patient had a history of frequent plasma donations, leading to a significant reduction of immunoglobulins contributing to an increased susceptibility to bacterial coinfection. The patient’s condition declined rapidly necessitating veno-venous extracorporeal membrane oxygenation (ECMO) and further veno-arterial-venous ECMO. However, despite all treatments and interventions, he continued to deteriorate, sustaining cardiac arrest and death. This case highlights the potential risks associated with frequent plasma donations, particularly their role in causing hypogammaglobulinemia and predisposing individuals to severe bacterial infections. Further studies are necessary to explore the effects of immunoglobulin depletion from plasma donation and its role in the development of severe infections and increased mortality.
Introduction
Influenza is widely recognized for its potential to induce extensive immunosuppression, particularly in vulnerable populations. However, in an otherwise healthy, immunocompetent individual, the repercussions of infection are typically less severe. In this case, a unique combination of factors contributed to the patient’s rapid deterioration. Specifically, frequent plasma donations resulted in a depletion of the patient’s immunoglobulin levels, thereby heightening their susceptibility to secondary infections, such as post-viral bacterial pneumonia caused by Staphylococcus aureus. Bacterial coinfection during influenza is a common occurrence, with S. aureus, Haemophilus influenzae, and Streptococcus pneumoniae being the most frequently implicated pathogens. Studies have consistently demonstrated that bacterial coinfections during influenza are associated with significantly increased mortality rates, as evidenced in a study examining fatal H1N1 cases between May and August 2009. 1 Despite this, there is limited literature on the specific effects of hypogammaglobulinemia secondary to plasma donation.
This case underscores the susceptibility of a previously healthy young male, whose immunosuppressed state—resulting from both plasma donation and influenza infection—led to rapid clinical deterioration. The patient developed acute respiratory distress syndrome (ARDS) and septic shock due to methicillin-resistant S. aureus (MRSA) pneumonia and bacteremia, necessitating veno-venous (VV), and later veno-arterial-venous extracorporeal membrane oxygenation (VAV ECMO), ultimately resulting in the patient’s demise.
Case Presentation
A 30-year-old male with a medical history of major depressive disorder (currently untreated) and childhood asthma presented to the emergency department with subjective fever, dry cough, progressive shortness of breath, and myalgias for 5 days. On initial assessment, the patient was hemodynamically stable; however, laboratory results revealed leukopenia white blood cell 1.2 × 103 (4.5-11.0 × 103), thrombocytopenia platelets 96 K/mm3 (140-440 K/mm3), absolute neutrophil count of 0.17 (1.3-7.8), accompanied by bandemia of 8%, and a procalcitonin level of 15.1 ng/mL (0.05-0.16 ng/mL). The chemistry panel indicated an elevated creatinine of 2.27 mg/dL (0.60-1.30 mg/dL) and lactic acid of 6.6 mmol/L (0.5-2.2 mmol/L; Table 1). A respiratory viral panel was positive for Influenza A. The patient also had markedly elevated creatine kinase (CK) levels exceeding 100 000 U/L, likely due to influenza. Chest radiography revealed bilateral patchy opacities. The patient was initiated on Tamiflu, azithromycin, vancomycin, and cefepime empirically, along with fluid resuscitation. Despite therapy, the patient became increasingly hypoxic, requiring high-flow nasal cannula and subsequently was intubated.
Laboratory values.

Further discussion with the family revealed that the patient had been donating plasma regularly over the preceding weeks at multiple donation centers but could not specify the frequency of these donations. Following the discussion with the family, immunoglobulin levels were measured and found to be low, with IgG at 389 mg/dL, IgM at 29 mg/dL, and IgA at 76 mg/dL.
On the first day of ICU admission, the patient’s pneumonia panel revealed S. aureus with the mecA gene detected, indicative of MRSA pneumonia. Sputum and blood cultures later grew MRSA. CT angiography revealed bilateral patchy opacities bilaterally, more on the left than the right (Figures 1 and 2). Bronchoscopy showed diffusely erythematous trachea and major bronchi with blood pooling in the superior segment of the left lower lobe. Arterial blood gas analysis revealed increasing hypoxia despite ventilator adjustments, with a PaO2/FiO2 (P/F) ratio of 140, indicative of severe ARDS. The patient was subsequently placed on VV ECMO. The following day, a chest tube was inserted for a large left-sided pleural effusion (Figure 3). Pleural fluid analysis revealed empyema, with MRSA cultured. Bedside echocardiography revealed a left ventricular ejection fraction of 35% to 50% with global hypokinesis. Given the patient’s continued deterioration, the decision was made to transition to VAV ECMO. Efforts including continuous VV hemofiltration and VAV ECMO failed to halt the patient’s decline, and he ultimately expired within 3 days of admission.

CT angiography: bilateral patchy opacities, more on the left than the right, with a consolidation on the left.

Computerized tomography angiography: bilateral patchy opacities, more on the left than the right, with a consolidation on the left.
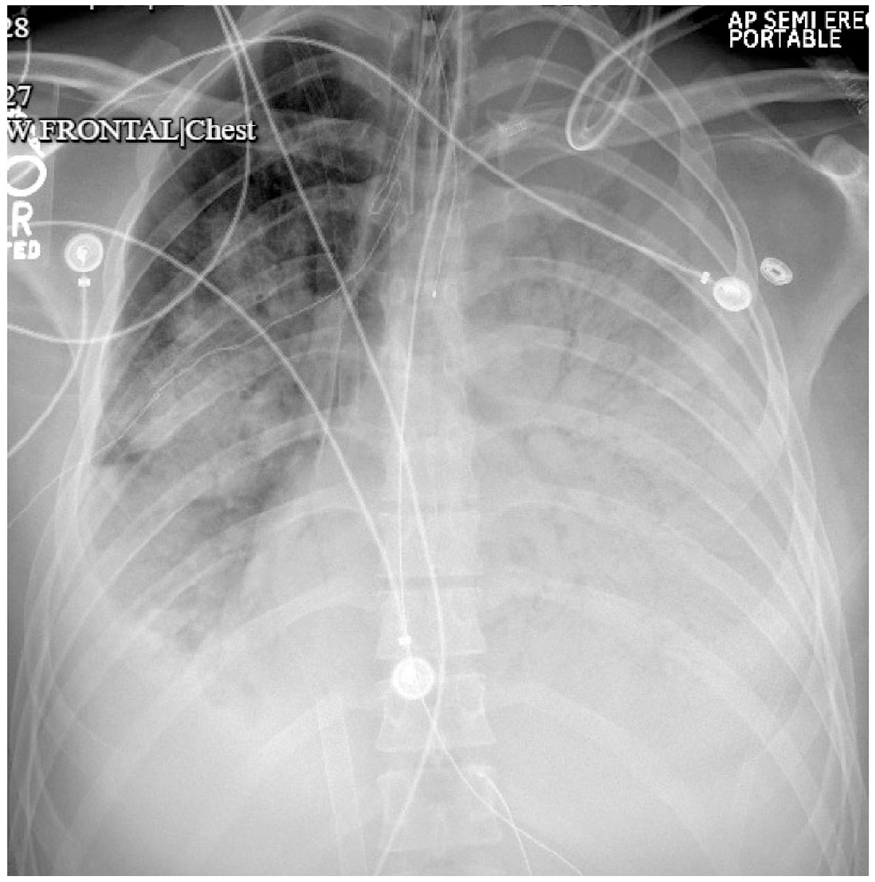
X-ray frontal chest: left-sided pneumothorax, with surrounding effusion.
Discussion
Influenza, a member of the Orthomyxoviridae family, is an RNA virus historically responsible for numerous pandemics. Influenza A and B are the primary strains involved in such outbreaks, due to their frequent mutations and resulting changes in antigenicity.2,3 The H1N1 strain, first identified in 1918 during the Spanish flu pandemic, resurfaced in 2009 as the swine flu. Pneumonia was the most common complication during the 2009 H1N1 pandemic, with many cases progressing to ARDS. 2 Influenza has the potential to impact the immune system in various ways, including the induction of severe bicytopenia. In a study of hospitalized patients with H1N1, leukopenia and thrombocytopenia were observed in 20% and 14% of cases, respectively. 4 However, neutropenia secondary to influenza is less commonly reported. 4 The cytokine storm triggered by influenza and sepsis can alter bone marrow production, impairing both lymphoid and myeloid cell lines. 5 Neutrophils, derived from myeloid progenitor cells, are equipped to respond to tissue injury or infection, and play many roles in the body’s defense system. 6 They are capable of phagocytosing pathogens and presenting their antigens, producing antimicrobial enzymes such as NADPH oxidase system in the creation of reactive oxygen species for elimination of microbes, and releasing chemokines that assist in the body’s response to infection. 7 Neutrophils play a major role in the body’s immune defense against bacteria such as Staphylococci, Streptococci, and Escherichia coli, and fungi like Candida. 7 Severe neutropenia is defined as <0.5 × 109/L, and once the neutrophil count is below 1.5 × 109/L, one becomes more susceptible to the aforementioned organisms. 8 Neutrophils and immunoglobulins work collectively against a pathogen by the process of opsonization. 6 Immunoglobulins, made by plasma cells, produce 5 types: IgG, IgM, IgA, IgE, and IgD. Their major function is opsonization of foreign pathogens and activation of the complement system; therefore, hypogammaglobulinemia predisposes individuals to recurrent infections. 6 Primary immunodeficiencies are caused by genetic mutations, leading to decreased immunoglobulins and therefore impaired immune response. 9 They have a bimodal distribution noted at the ages of 6 to 10, and 20 to 40, and lead to an increased risk of recurrent infections, allergies, and neoplasm.10,11 Common variable immunodeficiency (CVID) is the most common in adults, and X-linked agammaglobulinemia (XLA) in children. 12 CVID is an immune dysfunction of B, T, and dendritic cells due to the inability of B-cell differentiation into plasma cells, resulting in a decrease in all immunoglobulins. 13 XLA is caused by a mutation in Bruton tyrosine kinase, which assists in the maturation of B-cells, thereby leading to low plasma cells and ultimately immunoglobulins. 14 Secondary immunodeficiencies are caused by external elements affecting immunoglobulin production, such as corticosteroids, immunosuppressant drugs, nutritional deficiencies, chemotherapy, malignancy, and infection. 9
ARDS, as defined by the Berlin criteria, is characterized by acute onset (within 7 days), significant hypoxemia (P/F ratio <300 with positive end-expiratory pressure (PEEP) or contious postive airway pressure (CPAP) ≥5, and bilateral chest radiographic findings consistent with pulmonary edema with no clinical evidence of left atrial hypertension. 15 The mortality rate for ARDS can reach up to 30%. 15 Common risk factors include pneumonia, sepsis, and pancreatitis, with e-cigarette and vaping-associated lung injury also contributing to its incidence.15-17 The diagnosis of ARDS is primarily clinical, based on the exclusion of other causes of pulmonary edema and hypoxemia. The pathogenesis involves endothelial cell damage, leading to increased capillary permeability, osmotic dysregulation, and the influx of inflammatory mediators that result in diffuse alveolar damage. Ventilator management strategies for ARDS emphasize low tidal volume (6 mL/kg) and plateau pressures <30 cm H2O, with adjustments to PEEP and FiO2 according to ARDSnet protocols.15,18,19 Prone positioning has been shown to improve outcomes in patients with severe ARDS (P/F ratio <150), and neuromuscular blockade with cisatracurium has demonstrated mortality benefits in some randomized trials; however, neuromuscular blockade is not universally accepted to improve mortality.15,20,21 ECMO is considered in severe ARDS with potentially reversible respiratory failure.
Plasma is the liquid base of blood and contains 91% to 92% water and 8% to 9% solids such as coagulants, mostly fibrinogen, proteins such as albumin and globulin, electrolytes, and immunoglobulins.22,23 Plasma donation by plasmapheresis is regulated by the Food and Drug Administration and outlined by the Code of Federal Regulations as the following: the amount of whole blood, not including anticoagulant, removed from a donor during a manual plasmapheresis procedure or in any 2-day period shall not exceed 1000 mL unless the donor’s weight is 175 pounds or greater, in which case the amount of whole blood, not including anticoagulant, removed from the donor during a manual plasmapheresis procedure or in any 2-day period shall not exceed 1200 mL. No more than 500 mL of whole blood shall be removed from a donor at one time, unless the donor’s weight is 175 pounds or greater, in which case no more than 600 mL of whole blood shall be removed from the donor at one time. Collection shall not occur <2 days apart or more frequently than twice in a 7-day period. 24 Regulation of laboratory levels are also enforced and described as the following: If the protein composition is not within normal limits established by the testing laboratory, or if the total protein level is <6.0 or more than 9.0 g/dL in a plasma sample or serum sample, the donor must be deferred from donation until the protein composition returns to acceptable levels. Reinstatement of the donor into the plasmapheresis program when the donor’s protein composition values have returned to an acceptable level must first be approved by the responsible physician. 24 Such regulations are put in place to ensure safe donation by the patient. There is 1 report previously documented of immunoglobulin depletion following frequent plasma donation, as seen in a 43-year-old female who donated plasma biweekly and subsequently developed recurrent infections due to low IgG levels. 25
In this case, the patient’s immunocompromised state, compounded by influenza infection and septic shock, created a “perfect storm” for the development of severe bacterial coinfection, leading to ARDS and ultimately fatal outcomes. In this case, the patient’s frequent plasma donations depleted his immunoglobulin stores, resulting in low IgG and IgM levels and hence an immunocompromised state. His initial Influenza A infection was further compounded by a superimposed bacterial pneumonia leading to septic shock, further complicated by ARDS, multiorgan failure, and ultimately death. Our aim in this paper is to shed light on the association between infections and immunocompromised states and to focus on the importance of recognizing these conditions to target early and effective treatment.
Conclusion
This case highlights the critical role that plasma donation can play in predisposing individuals to hypogammaglobulinemia and its potential contribution to severe infectious complications. The interplay between influenza, septic shock, and immunoglobulin depletion underscores the importance of considering all underlying factors in the management of patients with complex clinical presentations. Further research is needed to better understand the risks associated with frequent plasma donation and its impact on immune function.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval/waiver for case reports.
Informed Consent
Verbal informed consent was obtained from the patient’s family for anonymized patient information to be published in this article.
