Abstract
Hereditary diffuse gastric cancer (HDGC) is a rare and aggressive cancer that accounts for 1% to 3% of gastric cancer cases, often diagnosed late with poor outcomes, and it is due to a mutation in the Cadherin 1 (CDH1) gene. We present 2 sisters, ages 62 and 70, both carrying the CDH1 mutation, which is associated with significantly increased risks of HDGC and breast cancer. Both were diagnosed with invasive lobular carcinoma of the breast, a known manifestation of CDH1 mutations, emphasizing the need for genetic screening in patients with strong family cancer histories. Despite their elevated HDGC risk, the sisters declined prophylactic surgery. Instead, they pursued annual endoscopic gastro-duodenoscopies with ultrasound surveillance tailored to their needs. This case highlights the importance of recognizing rare genetic mutations and integrating family history into personalized clinical management.
Keywords
Introduction
Gastric cancer is the fourth most common cancer worldwide and the second cause of cancer mortality. It is most prevalent in Eastern Asia, with an incidence rate of ~900 000 yearly. 1 Histologically, 10% of tumors are classified as diffuse type, which are infiltrative and poorly differentiated. 2 While many gastric cancers are sporadic, about 1% to 3% are inherited. 3 Hereditary diffuse gastric cancer (HDGC) is an autosomal dominant condition associated with mutations in the Cadherin 1 (CDH1) gene, and it has an estimated incidence of 5 cases/100 000 individuals. 4 HDGC is part of a broader hereditary cancer syndrome, which is strongly associated with hereditary lobular breast cancer. Studies indicate that ~1.5% of women with lobular breast cancer carry pathogenic CDH1 variants, which are linked to an earlier age at diagnosis and a family history of breast cancer. Notably, lobular breast cancer may be the first manifestation of hereditary cancer syndrome, even in the absence of diffuse gastric cancer. 5
The prognosis for HDGC remains poor due to its aggressive behavior, late-stage diagnosis, and worse response to treatment. 6 The uncertainty surrounding the safety and efficacy of endoscopic surveillance options has resulted in recommendations for prophylactic total gastrectomy (PTG) in individuals with the CDH1 pathogenic variant. However, this approach needs to be reconsidered as recent studies suggest the risk of developing advanced HDGC is lower than initially thought and depends on the penetration in the family. 7 Annual endoscopic surveillance may be a suitable alternative for those who choose to delay surgery due to potential complications, such as dumping syndrome and malabsorption. 3 In such cases, endoscopic surveillance provides a viable option for closely monitoring disease progression. 3
This case series highlights the critical importance of considering hereditary factors, such as the CDH1 gene mutation, in patients with a family history of invasive lobular carcinoma or gastric cancer. Recognizing these rare genetic associations is essential for screening family members for associated malignancies and screening affected individuals for HDGC, enabling early detection and proactive management. 8 It also highlights alternative methods for early detection of HDGC in patients with CDH1 gene mutations.
Case Presentation
We present a case series of 2 sisters, aged 62 and 70, with invasive lobular carcinoma of the breast and a shared pathogenic CDH1 mutation. They have a strong family history of multiple malignancies, as shown in the pedigree (Figure 1), which prompted us to consider hereditary cancer syndromes.

Family pedigree with hereditary cancer syndrome. Patients that are filled in carry the CDH1 genetic mutation.
Case 1: The Younger Sister
The 62-year-old female was initially found to have a suspicious focal asymmetry with a 4.1 cm mass-like appearance on the right breast and indistinct borders on routine mammography, classified as Breast Imaging Reporting and Data System category 4. The patient underwent a biopsy (Figure 2A) and was diagnosed with estrogen receptor (ER) positive and human epidermal growth factor receptor 2/neu (HER2/neu) negative invasive lobular carcinoma of breast (T2N0M0, Grade 2, Stage IIA). For this, she underwent lumpectomy, radiation, and Tamoxifen therapy.

(A, B) H&E, original magnification 100×. (A, case 1) Right breast tissue: invasive lobular carcinoma, Grade 2, Nottingham histologic score 6 (tubules 3, nuclei 2, mitoses 1), measuring 41 mm in greatest dimension. A tumor is present at the anterior margin, but no lymphovascular invasion is identified. Lobular carcinoma in situ present. (B, case 2) Right breast tissue: invasive lobular carcinoma with pleomorphic features, Grade 2, Nottingham histologic score 6 (tubules 3, nuclei 2, mitoses 1), measuring 24 mm in greatest dimension. Tumor invades the dermis without skin ulceration. Margins are negative for malignancy. No lymphovascular invasion is identified.
Genetic testing with the breast cancer gene (BRCA) and panel identified a pathological heterozygous CDH1 mutation (p.G239R). Recognizing her elevated cancer risks, she underwent esophagogastroduodenoscopy (EGD) as part of gastric cancer surveillance. Initial findings included benign antral mucosa with superficial hyperplastic changes (Figure 3). Follow-up EGD (Figure 4) with endoscopic ultrasound (EUS; Figure 5) and biopsies per the Cambridge protocol revealed reactive gastropathy without malignancy. The patient declined PTG against current recommendations and instead chose annual EGD and mammographic surveillance. Since then, she has remained asymptomatic with no signs of HDGC.

(A, B) H&E, original magnification 100×. (A) Antral polyp low-power polypoid fragment of antral mucosa with superficial erosion and marked thermal artifact precluding further evaluation and (B) high-power, fragment of benign antral mucosa with superficial hyperplastic change.
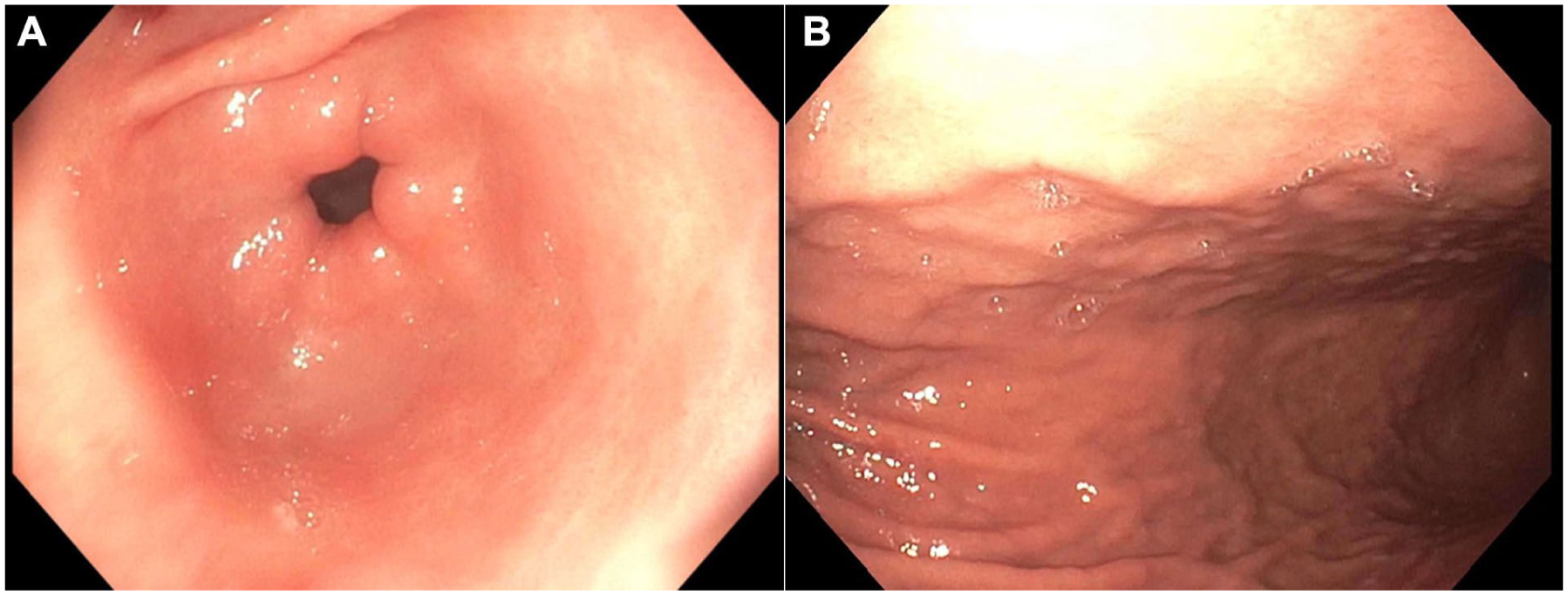
(A, B) Esophagogastoduodenoscopy. (A) Prepyloric stomach and (B) gastric body.

(A, B) Endosonographic images of the stomach were unremarkable.
Case 2: The Older Sister
A 70-year-old woman was diagnosed with 2.7 cm left breast invasive lobular carcinoma (T2N0M0, Grade 2, Stage IA), ER/progesterone receptor (PR) positive, HER2/neu negative. She underwent lumpectomy and radiotherapy. She did not require chemo or hormonal therapy. Three years later, she developed right breast invasive lobular carcinoma (T2N0M0, Grade 2, Stage IA; Figure 2B), which is ER/PR positive, HER2/neu negative. She required a bilateral mastectomy and is undergoing Anastrozole therapy. Genetic testing revealed a CDH1 mutation (p.G239R) and a variant of unknown significance (PTCH1 p.A1141V). She also opted against a PTG and has chosen to undergo annual EGD surveillance instead and has shown no evidence of developing HDGC to date. Notably, this patient transmitted the CDH1 mutation to her daughter, who was diagnosed with breast cancer and underwent prophylactic gastrectomy, featuring the penetrance of this mutation within the family.
Discussion
We present a case series involving 2 sisters who carry the CDH1 gene mutation. This mutation is linked to a high risk of HDGC and invasive lobular carcinoma of the breast. The 70-year-old patient had bilateral lobular breast cancer and passed the CDH1 gene mutation to her daughter, who was subsequently diagnosed with breast cancer and underwent prophylactic gastrectomy. Despite the heightened risk associated with their mutation, both sisters declined PTG and opted for regular surveillance. This case stresses the critical importance of family history and genetic screening in managing rare genetic cancer disorders. Furthermore, this case report prompts further research into the penetrance and expressivity of the CDH1 mutation, contributing to the understanding of hereditary cancer syndromes.
HDGC is notorious for being a more clinically aggressive form of gastric cancer, often associated with peritoneal metastases and poor survival rates. 9 Most individuals with CDH1 mutations develop related cancers at ages 30 to 40. This also provides insight into variability in disease presentation and progression and valuable clinical insights into the manifestation and management of HDGC in older adults. This case report raises awareness about HDGC and the need for vigilance in screening and diagnosis, regardless of age. By age 80, carriers of this mutation have been reported to have a cumulative incidence of gastric cancer of 70% in males and 56% in females. 10 Additionally, the risk of breast cancer in females reaches 42%. 10 However, a recent study showed the risk of developing advanced HDGC is lower than previously believed and is influenced by the degree of penetration within the family. 10
To contextualize our findings further, we reviewed the literature to summarize the reported cases of CDH1 mutation carriers with associated hereditary cancer syndromes. To the best of our knowledge, we found 15 patients with HDGC. Table 1 highlights key characteristics, including clinical presentations, genetic findings, management strategies, and outcomes.
Previous Reports Detailing the Presentation, Mutation, Biopsy Findings, Treatment, and Outcomes in Managing Patients With CDH1 Mutations.

Abbreviations: CA-19.9, carbohydrate antigen 19-9; CDH1, Cadherin; CEA, carcinoembryonic antigen; CT, computerized tomography; EGD, esophagogastroduodenoscopy; GERD, gastro esophageal reflux disease; PET, positron emission tomography; pT3 pN1 G4, tumor Stage 3, Nodal 1, and Grade 4.
In this case series, a review of similar cases of CDH1 mutations reveals several key trends and outcomes. The patients, predominantly young adults, ranged from 22 to 53 years old, with a slight female predominance (60%). About 70% of the patients presented with a strong family history of diffuse gastric cancer or invasive lobular carcinoma of breast, reflecting the autosomal dominant inheritance of the CDH1 mutation. The CDH1 mutation, which includes various pathogenic variants such as frameshift mutations, deletions, and splicing defects, emerged as a critical genetic factor linked to HDGC—mutations identified range from monoallelic deletions to deleterious germline mutations. About 80% of them underwent PTG, despite some cases showing negative gastric biopsies initially. A significant proportion of these individuals (~65%) showed signet-ring cell carcinoma in the histopathology results.11 -17
Treatment approaches varied depending on the stage of cancer. Those with early-stage cancers, including 3 cases with T1aN0M0 stage, underwent PTG, and no recurrence was observed in the following year (about 60% of these patients). Conversely, those diagnosed with advanced gastric cancer (pT3, pN1) received chemotherapy, with some requiring additional interventions like splenectomy and colic resection. Unfortunately, the recurrence rates were notable, with several cases showing signs of cancer progression 1 to 2 years postsurgery, especially for those with advanced disease at diagnosis. Approximately, 20% of the cases in the table had recurrence after gastrectomy.11 -17
In terms of survival outcomes, some patients experienced favorable results with no recurrence for over 3 years postsurgery, while others had a more aggressive course, with the majority of patients who died having advanced stages of disease at the time of diagnosis.11 -17
Regarding genetics, CDH1 gene mutation follows an autosomal-dominant inheritance pattern. 18 HDGC is defined by the presence of pathological germline mutations accounting for almost 40% of the cases 18 in either the CDH1 tumor suppressor gene, known to have 155 different germline mutations, 10 or the Catenin Alpha-1 (CTNNA1) gene plays a vital role in cell adhesion to actin filaments. CDH and CTNNA1 are tumor suppressor genes implicated in pathogenesis and responsible for cell-to-cell adhesion.4,19 HDGC has been closely linked with CDH1 mutations, which lead to CDH-1 loss of function. Of note, CDH-1 is responsible for cell proliferation, maintenance of cell adhesion, cell polarity, and epithelial-mesenchymal transition. 20
First-degree relatives of individuals with a CDH1 gene mutation have a 50% chance of inheriting the same mutation. Genetic counseling should be offered to patients and their first-degree family members, as it helps assess risk, explores genetic testing options, and provides guidance on surveillance and preventive strategies. 21 Additionally, it aids families in understanding the implications of hereditary cancer risks. Genetic testing for CDH-1 is recommended for patients with a personal history of bilateral lobular breast cancer diagnosed at age 70 or younger or multiple close relatives with lobular breast cancer. This test may also be offered to biological relatives spanning 3 generations.18,22 Given the significant impact of pathogenic CDH1 mutations in hereditary lobular breast cancer, comprehensive genetic screening for pathogenic or likely pathogenic germline CDH1 variants in patients with lobular breast cancer who do not meet the classic HDGC criteria but have an uncertain risk of developing gastric cancer may even provide further valuable insights for genetic counseling. 5
The primary management strategy for CDH1 carriers remains PTG, which can potentially be lifesaving but may lead to lifelong digestive complications. 4 Also, the best timing for PTG is not well established and tends to vary greatly based on individual circumstances. 22 On the other side, recent studies indicate that CDH1 carriers have a lower incidence of HDGC than previously thought, suggesting that endoscopic surveillance may prove more beneficial in the future. 23
The Cambridge protocol offers guidance for annual endoscopic surveillance in CDH1 carriers. 21 Since HDGC spreads in the stomach lining, multiple biopsies are necessary (4-5 biopsy specimens in the following anatomical sections: cardia, fundus, corpus, transitional zone, antrum, and prepyloric area). 24 This approach aims to increase the likelihood of detecting early lesions that may indicate the presence of cancer. Ideally, surveillance should occur in multidisciplinary, high-volume centers staffed by experts in HDGC. Endoscopic surveillance should include thorough high-definition white light examination with repeated insufflation and deflation to enhance visualization of the gastric mucosa and assess distensibility. A lack of distensibility should raise suspicion for an infiltrative process like linitis plastica. If poor distensibility is observed, further evaluation with a computed tomography scan and EUS is recommended. 24
Despite following surveillance protocols, the endoscopic detection of cancer foci in HDGC remains suboptimal and insufficient for guiding decision-making. Alternative endoscopic techniques, such as chromoendoscopy, EUS, and other nonwhite light methods, have been employed. However, their effectiveness in enhancing cancer detection and risk stratification in HDGC is limited. 25
Some experts prefer the term “risk-reduction gastrectomy” over “prophylactic gastrectomy,” as many mutation carriers may already have microscopic signet ring cell carcinomas at the time of surgery. These cancers typically have a dormant period due to their low proliferative index, and PTG is generally performed at a younger age than when overt cancer usually develops. Nonetheless, total gastrectomy effectively eliminates the risk of gastric cancer and prevents death from invasive gastric cancer. However, if HDGC develops the prognosis is poor, with only about 10% having a chance for potentially curable disease. Even among those who may be cured, the 5-year survival rate does not exceed 30%. 22
Conclusion
This case highlights the need for clinicians, particularly breast cancer oncologists, gastroenterologists and gastrointestinal cancer specialists to remain vigilant about the potential for hereditary cancer syndromes, especially in cases where family history suggests a possible genetic link. For individuals and families affected by CDH1 mutations associated with breast cancer, proactive genetic counseling is crucial to inform at-risk individuals about their options. All CDH1 carriers should undergo an initial upper endoscopy, and those choosing to defer PTG should commit to rigorous annual endoscopic surveillance. This approach facilitates early detection and timely intervention for HDGC. By presenting this case series, we hope to highlight the significance of early genetic screening and the importance of individualized surveillance strategies, which could ultimately impact outcomes in hereditary cancer syndromes like HDGC.
Footnotes
Acknowledgements
We sincerely thank the Research Committee of the Internal Medicine Department, Marshall University, WV for their invaluable support and guidance throughout the development of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
