Abstract
Here, we present a 33-years-old pregnant female who at 27-weeks gestation developed an unusual rash on her left knee after a spider bite. The rash rapidly became systemic requiring admission and treatment with high-dose steroids. Patient rapidly developed complications of steroid therapy and saw no benefits in reducing the activity of the rash of symptoms of itching. A repeat biopsy showed linear fluorescence along the dermal epidermal junction with C3 (strong) and IgG (weak) suggestive of pemphigus gestationis (PG). After balancing concerns for the health of the mother and risk for disease transmission to the child, we decided to treat the patient with high-dose intravenous immunoglobulin (IVIg). This resulted in rapid remission of disease and delivery of a healthy child without evidence of PG at 36-weeks gestation. A discussion of the mechanism(s) of action of IVIg that were salutary in this case is presented. Importantly, we discuss the likely benefits of IVIg in saturation of the Fc-receptor neonatal IgG recycling and preservation system in accelerating the degradation of pathogenic IgGs and the inhibitory of effects of IVIg on C3 activation which was the predominant immunoreactant in skin biopsy. The patient recovered completely after the fourth monthly IVIg infusion and continues to do well.
Introduction
Autoimmune bullous diseases (AIBDs) represent a group of mucocutaneous disorders usually associated with specific autoantibodies aimed at adhesion molecules located in the dermal/epidermal junction. 1 The appearance of AIBDs in women of childbearing age is usually associated with a diagnosis of pemphigus vulgaris (PV) or pemphigus gestationis (PG). 2 However, PG is more commonly related to pregnancy.
PG is a rare AIBD seen in ~1/50 000 pregnancies. Presentation usually occurs in the second or third trimester and has a proclivity for recurrence in subsequent pregnancies, usually with earlier onset and more severe features.3,4 Rapid diagnosis is critical for formulation of therapeutic interventions and requires a proper skin biopsy with immunofluorescent assessments. Once diagnosis is established, it is also critical to determine therapeutic options that are efficacious and pose little or no safety concerns for the unborn child and do not have potential for transmission to the newborn through breast milk.
These considerations often require a multidisciplinary approach to management of the mother with PG and the unborn child which could be potential affected by pathogenic IgG autoantibodies being transmitted across the placenta by the Fc-receptor neonatal (FcRn) transport system. 5 This could also happen with breastfeeding as FcRn receptors have increased expression on neonatal enterocytes. 6 In this regard, IVIg use would be an important consideration as it can inhibit FcRn recycling of pathogenic antibodies and treat antibody-mediated disorders. 7 Thus, this would allow physicians to avoid cytotoxic therapies that can cross the placenta and potentially damage the fetus. With these considerations, PG poses major therapeutic challenges to the mother and unborn child. Neonates born with PG can exhibit similar features as seen in the mother, but these usually resolve within 4-weeks commensurate with the reduction in half-life of IgG transmitted from mother to child. 8
Here, we present an unusual case of PG that was unresponsive to topical and high-dose oral steroids with toxicity, but rapidly responded to high-dose IVIg therapy.
Case Presentation
The patient is a 33-years-old Caucasian female who presented to an outside facility complaining of a spider bite to her left knee area. According to the patient, she was in her usual state of good health and 27-weeks’ gestation with her first child until the erythematous lesion on her left knee developed into a macular rash with raised edges and central pustule that spread over her entire body (Figure 1A and B). An initial biopsy performed at an outside facility reported findings consistent with hypersensitivity reaction. The patient had no personal or family history of autoimmunity. She was admitted to hospital and treated with high-dose (80 mg/daily) prednisone without significant benefit over a course of 1 month. At this point, the patient was experiencing extensive steroid-related side-effects including weight gain, considerable Cushingoid features, and glucose intolerance. Eventually, immunology consult was obtained. Importantly, there was no significant response to the steroids. A repeat skin biopsy was done with immunofluorescence. Results are shown in (Figure 2A–C). Briefly, the biopsy showed mixed dermal inflammation with eosinophils (Figure 2A) with immunofluorescence showing intense linear basement membrane zone deposition with C3 (3+) and IgG (1+) (Figure 2B and C) consistent with a subepidermal autoimmune blistering disorder. Given the clinical history, these findings were most consistent with the clinical impression of pemphigoid gestationis (PG).

(A) This figure shows the initial lesion noted by the patient on her left knee at initial presentation to the emergency room she indicated this arose after a spider bite. Note the maculopapular nature of the lesion with beginning of satellite lesions. (B, C) After admission, the rash rapidly spread to the entire body and was diffuse maculopapular in nature with erythema with extreme pruritus (B) Arrow indicates maculopapular lesion. (C) The evolution of the rash to a bullous, erythematous, and weeping condition with severe pruritus.
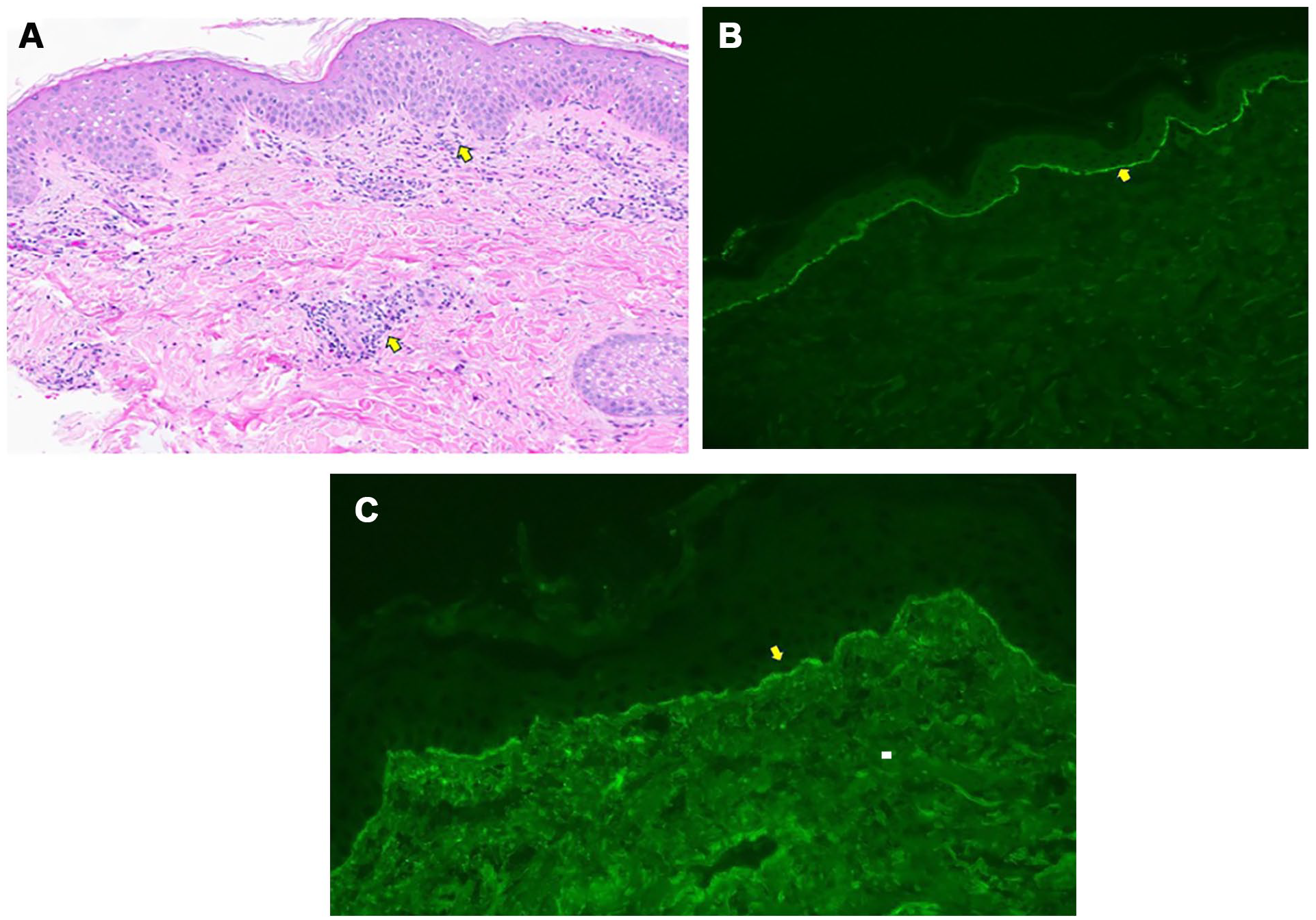
(A) Histologic examination shows mild acanthosis without an obvious subepidermal separation; however, eosinophils are easily found at the dermo-epidermal junction and within the perivascular inflammatory cell infiltrate (original magnification: ×20; see arrows). (B) Direct immunofluorescence shows 3+ continuous linear deposition of C3 along the basement membrane zone (original magnification: ×20). No immunoreactivity was identified for IgA, IgM, and fibrinogen. (C) Direct immunofluorescence shows continuous 1+ linear deposition of IgG along the basement membrane zone (original magnification: ×20).
After a discussion with the multidisciplinary care team, a consensus was obtained on altering therapeutics. Given the considerations mentioned above regarding providing a therapeutic approach that would potentially be effective and would lack toxicities to the mother and child, we decided to treat the patient with IVIg (2 g/kg, max dose 140 g) monthly with tapering steroids. The patient showed almost immediate reductions in new lesions after the first dose and showed steady improvements with subsequent doses. The patient subsequently developed thrombocytopenia and slight increases in blood pressure felt to be consistent with early pre-eclampsia. She underwent elective C-section delivery at 36-weeks 3 days gestation and delivered a normal and healthy baby boy. The child showed no manifestations of PG. The patient continued IVIg monthly and now has shown complete resolution at 6 months postdelivery (Figure 3A and B). The mother has breast fed her child without any noted adverse events.

(A) Rapid improvement in the rash at 2 weeks after the first IVIg infusion. (B) The complete resolution of the rash at 4 months postdelivery and after her fifth IVIg dose.
Discussion
Autoimmunity and autoimmune diseases result primarily from an inability of the immune system to distinguish antigenic anatomy of self from nonself. Here, initiation of immune activation pathways against self-antigens, results in tissue injury which can be tissue-specific or systemic. Importantly, disease initiation likely occurs after an insult to the target organ (ie, spider bite) in this case that results in immune responses to foreign and self-antigens that cannot be terminated and are perpetuated. For several autoimmune disorders, including PV and PG, autoreactive IgG antibodies play a central role in disease pathology, and decreasing autoantibody levels, effector pathways (ie, complement activation), and possibly autoantibody producing cells is paramount for achieving disease remission.
PG is a rare dermatologic complication of pregnancy which is often recalcitrant to therapeutic interventions. Standard of care usually consist of high-dose oral and topical steroids. However, these can cause significant toxicities and, as seen in this case, can be ineffective. Other potential treatments include agents such as Imuran, cyclosporine, and anti-CD20 monoclonals which could pose toxicities to the neonate, especially if continued postdelivery in breastfeeding mothers. Thus, finding a therapeutically effective agent that poses little or no toxicities to mother and fetus is desirable. IVIg has been successfully used to treat AIBDs, with reduction of blisters and allowing tapering of high-dose steroid; however, there are very few reports of use of IVIg in treatment of PG.9-11 In our case, there was rapid resolution of skin lesions after initiation of IVIg and the child was born without any evidence of PG.
The mechanism(s) of action of IVIg which are ameliorative to autoimmune diseases including AIBDs, have recently become clearer.7,9-11 Previous reports suggests that IVIg acts through multiple mechanisms, these include anti-idiotypic antibodies that block the actions of pathogenic IgGs, inhibition of T- and B-cell activation, absorption of complement activation fragments and inhibition of cytokines. All could have benefits in reducing the pathologic features of PG; however, the primary pathway for efficacy of IVIg has become clearer.12,13
A novel and emerging concept for treatment of autoantibody mediated disorders is inhibition of the FcRn IgG recycling system. FcRn, is a nonclassical Fc gamma receptor that is expressed on most cells, including epithelial cells, endothelial cells, most immune cells, enterocytes in the gastrointestinal system, and in syncytiotrophoblast of the placenta. Initially, the FcRn was felt to be isolated to the placenta and responsible for transferring maternal IgGs to the unborn child, which is incapable of generating humoral immunity. However, we now know that the FcRn plays a central role in preserving IgGs and albumin by shunting them to recycling pathways in all cell types mentioned above.5-7 This occurs through a pH-dependent process where circulating IgGs are pinocytosed into FcRn-expressing cells. Here, the IgG Fc bind to FcRn in acidified endosomes at pH ≤6.5 with high affinity. The FcRn/IgG complexes are transcytosis to the cell surface and IgG released from the complex at the pH 7.4. IgG molecules not bound to FcRn are digested in the endosomes. This pH-dependent process allows for recycling and preservation of the precious IgG molecules, extending half-life of IgG to ~24 days. This salvage pathway is responsible for the relatively high concentration of IgG in the circulation and for the long half-life of IgG compared to IgA and IgM, which are not recycled. Indeed, the half-life of IgG in FcRn (−/−) animals is reduced to 3 days. Therefore, therapeutic agents that block the interaction of FcRn with IgGs would likely increase degradation and rapid clearance, especially IgGs with pathogenic potential.
It is now known that IVIg’s beneficial effects in human autoimmune diseases is likely through saturation of the FcRn system, preventing recycling of pathogenic IgGs and thus reducing disease manifestations. Agents that mimic this saturation effect, exerting very high affinity for FcRn have been developed and now are used to treat numerous autoimmune diseases mediated by pathogenic antibodies. 14 These are important considerations for our case since the placenta has high expression of FcRn needed for transmission of IgG from mother to child. Thus, inhibiting this system with IVIg likely prevented the transplacental transmission of pathogenic IgGs across the placenta to the newborn, preventing disease. It is also possible that the use of new agents that specifically block FcRn recycling of IgG such as efgartigomod might also be useful in treating AIBDs like PG. However, a recent report on a phase 3 clinical trial of (argenx Inc. Ghent, Belgium), evaluating efgartigimod (FcRn inhibitor) in adults with PV and pemphigus foliaceus showed the proportion of PV patients achieving the primary endpoint of complete remission on was not significantly different between efgartigimod and placebo. This suggest that the beneficial effects of IVIg may also relate to inhibition of complement activation and injury which has also been previously described. 15 Here, IVIg given in high-doses (2 g/kg) has shown ability to inhibit C3 activation by pathogenic IgG molecules and inhibit complement-mediated cytotoxicity, preventing injury to tissues and cells. These are important considerations in treatment of complement-mediated diseases.
In conclusion, we present a case of PG occurring at 27-weeks gestation that was unresponsive to steroid therapy. The use of IVIg administration rapidly improved the rash and allowed for delivery of a healthy male child at 36-weeks gestation without evidence of PG. Postdelivery use of IVIg has allowed for complete resolution of the rash. The mechanism(s) of action of IVIg in ameliorating PG likely relate to saturation of the FcRn allowing increased degradation of pathogenic IgGs and inhibition of complement activation and injury as C3 was the dominant reactant detected in the dermal/epidermal junction.
Footnotes
Acknowledgements
We would like to thank the patient for allowing us to provide the details of her clinical course and the medical staff of Cedars-Sinai Medical Center who provided care for her.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
This information has not been presented before.
