Abstract
Right atrial myxomas (RAMs) are exceedingly rare cardiac tumors that can incur devastating complications. These tumors are capable of producing right-sided heart failure (rHF) through obstruction of venous return and tricuspid inflow. Herein, we describe a 52-year-old woman presenting with classic symptomatology of rHF as clinical sequelae of a giant RAM. The clinician should be cognizant of RAMs as an atypical etiology of rHF, and the complementary imaging modalities in the diagnostic work-up and early surgical resection as key management strategies.
Introduction
Primary cardiac tumors are rare, with an incidence of less than 0.1% in autopsy findings. 1 Among these, myxomas are the most frequently benign and predominantly located in the left atrium (75%), while right atrial involvement is less common, accounting for only 15% to 20% of cases.2,3 Right atrial myxomas (RAMs) can cause profound hemodynamic derangements, primarily through mechanical obstruction of venous return and tricuspid inflow, resulting in venous congestion, tricuspid regurgitation (TR), and right heart failure (rHF).1,4 Clinical presentation can often be nonspecific, mimicking more common conditions such as pulmonary hypertension or chronic obstructive pulmonary disease. 5 This report presents an exceedingly rare case of a giant RAM causing severe rHF, highlighting the challenges of diagnosis and the imperative need for urgent surgical extirpation.
Case Presentation
A 52-year-old Caribbean South Asian woman with a history of controlled hypertension presented to the Cardiology outpatient clinic with a 2-month history of progressively worsening dyspnea and bilateral pedal edema. She denied angina, palpitations, or syncope. Her solitary medication for which she was adherent was lisinopril 20 mg. She did not have any pertinent family history of premature coronary artery disease or sudden cardiac death. She did not travel recently, nor did she have any pets. Her social history was positive for occasional alcohol use on special occasions, without any tobacco or illicit recreational drug use. Her review of symptoms revealed worsening fatigue and malaise with 5-pound weight loss without any recent viral illness or melenic bowel movements.
Physical examination revealed relatively normal and stable vital signs apart from resting tachycardia. The patient was alert, coherent, and responsive without any neurodeficit. The cardiac examination revealed a mildly tachycardic, regular pulse with an elevated jugular venous pressure (10 cm of water) with prominent v waves (Supplemental Video 1). There was a 3/6 pan systolic murmur auscultated at the left lower sternal border, which radiated throughout the precordium with a prominent parasternal heave. Her point of maximal apical impulse was palpated at the fifth intercostal space, mid-axillary line, with a normal tapping character. Her respiratory examination was relatively unremarkable, with bilateral vesicular breath sounds without wheezing or crackles. With respect to her abdominal exam, there was evidence of borderline hepatomegaly with a positive hepatojugular reflux and bilateral 2+ pitting edema up to the level of the tibial tuberosities. There was no sacral edema or anasarca. Normal bowel sounds were auscultated.
Routine outpatient laboratory investigations, including a complete blood count and comprehensive metabolic panel, were relatively unremarkable. However, 2-dimensional transthoracic echocardiography (2D-TTE) revealed a giant, mobile, heterogeneous mass measuring 7.8 cm × 3.9 cm attached to the interatrial septum, prolapsing into the tricuspid valve during diastole (Figure 1A and B). An eccentric jet of TR was noted but could not be accurately estimated due to the lesion (Figure 1B). Right atrial dilation and mild right ventricular systolic dysfunction were also observed. Further evaluation with a transesophageal echocardiogram (TEE) confirmed that the mass originated from the interatrial septum with near-complete prolapse through the tricuspid valve during diastole (Figure 1C). These preliminary findings were highly suggestive of a RAM with rHF, as evidenced by the clinical features and reduced tricuspid annular plane systolic excursion. A non-contrast-enhanced computed tomography (CT) scan of the chest, abdomen, and pelvis did not reveal any metastatic disease or generalized lymphadenopathy and reported that the lesion was localized within the right heart, consistent with the echocardiographic findings (Figure 2).

The patient’s echocardiographic series. (A) This is a parasternal short-axis view at the aortic valve level, which demonstrates a large, mobile, heterogeneous mass measuring 7.8 cm × 3.9 cm attached to the interatrial septum, prolapsing into the tricuspid valve during diastole outlined by the stippled line. (B) This is a zoomed-in apical 4-chamber view demonstrating the large, mobile, heterogeneous mass traversing the tricuspid valve with evidence of severe—torrential TR. (C) This is a mid-esophageal transesophageal image at 100°, also demarcating the large, mobile, heterogeneous mass traversing the tricuspid valve. TR, tricuspid regurgitation.
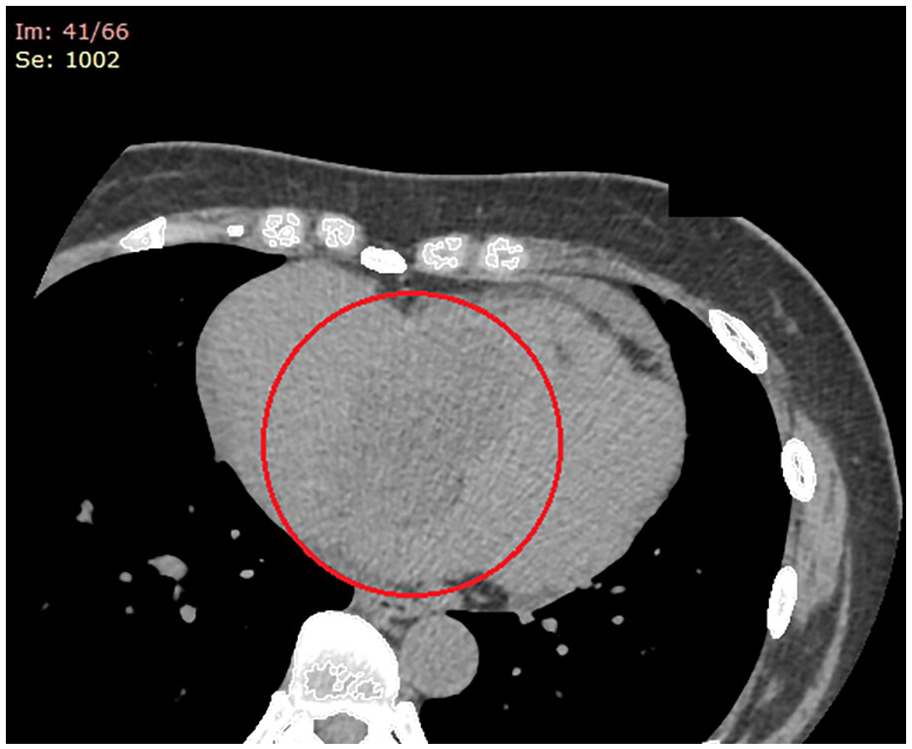
The patient’s computed tomography scan displayed the large, amorphous hypodensity within the right atrium and ventricle, encircled in red.
The patient underwent urgent surgical resection of the tumor. The mass was excised en bloc, including its pedunculated attachment to the interatrial septum (Figure 3A and B). Histopathological analysis confirmed the diagnosis of a myxoma, characterized by myxoid stroma with bland stellate cells and Gandy-Gamna bodies (Figure 4A–C).6,7 The patient experienced an almost immediate resolution of symptoms, including dyspnea and peripheral edema. She was discharged and advised to undergo routine interval echocardiographic surveillance.

The patient’s gross pathological resected specimen exhibits its giant horizontal (Panel A) and longitudinal dimensions (Panel B).

The patient’s histopathology. (A) This is a low-power (100×) hematoxylin and eosin stain of the cardiac myxoma, consisting of bland stellate (“lepidic”) cells with hemorrhage and Gandy-Gamna bodies. (B) This is an intermediate-power (200×) hematoxylin and eosin stain of the cardiac myxoma, consisting of bland stellate (“lepidic”) cells with hemorrhage and Gandy-Gamna bodies. (C) This is a high-power (400×) hematoxylin and eosin stain of the cardiac myxoma, highlighting the bland stellate cells in the myxoid.
Discussion
RAMs are unique among cardiac tumors in exerting profound hemodynamic effects, chiefly attributed to their size and location.8,9 Unlike left atrial myxomas, which typically exhibit embolic events or mitral valve dysfunction, RAMs often induce mechanical effects via obstructing venous return from the superior and inferior vena cava, as well as impeding tricuspid inflow.4,10 This results in elevated right atrial pressures, venous congestion, and a cascade of hemodynamic alterations that culminate in rHF. 8
In our case, the patient’s signs of elevated jugular venous distension with prominent v waves and bilateral pedal edema were clinical manifestations of venous congestion and impaired right ventricular filling. Diastolic prolapse of the RAM into the tricuspid valve orifice further accentuated the obstruction, creating a pseudo-functional stenosis. This dynamic obstruction is particularly concerning in large, mobile tumors, as it can lead to positional symptoms such as trepopnea and syncope. 10 The presence of TR, a common finding in RAM, exacerbates the pathophysiology. Chronic volume overload from regurgitation results in progressive dilation of the right atrium and ventricle, eventually resulting in right ventricular dysfunction. 11 These structural changes attenuate the heart’s ability to compensate, accelerating the progression of heart failure. 12
The nonspecific symptoms of RAMs can often obfuscate the clinical picture, with delays or misdiagnoses such as pulmonary hypertension, chronic lung disease, or rHF due to incipient left-sided dysfunction.5,13 In our patient, her initial symptoms were thought to reflect heart failure with reduced ejection fraction until imaging revealed the giant right atrial mass. This diagnostic delay highlights the need for clinicians to maintain a high index of suspicion for cardiac tumors in cases of unexplained rHF. While the classical triad of myxomas (obstruction, constitutional symptoms, and embolization) is well documented, the presentation of RAM is predominantly obstructive.9,14 The clinical scenario of jugular venous and abdominal distension with lower limb edema is characteristic but not specific. Constitutional symptoms, such as fever and fatigue, are less common but may result from cytokine release by the tumor. 6
Echocardiography remains the pivotal diagnostic imaging modality for cardiac myxomas. 2D-TTE serves as an initial screen, offering a reliable, swift, and noninvasive approach to detecting intracardiac masses. 15 Its comprehensive assessment includes salient features such as tumor size, mobility, heterogeneity, attachment to the interatrial septum, and hemodynamic impact. 6 In this case, it identified a large, mobile mass prolapsing into the tricuspid valve, with associated TR and right atrial dilation. The TEE provided superior resolution, affirming the tumor’s attachment to the interatrial septum and its partial prolapse through the tricuspid valve.15,16 Cardiac magnetic resonance imaging and CT have become complementary adjuncts for further characterizing cardiac masses. 1 MRI offers enhanced soft tissue contrast, enabling differentiation between benign myxomas, vascularity, and potential invasion into adjacent structures.16,17
Complete surgical excision is the definitive treatment for cardiac myxomas, addressing both mechanical obstruction and the risk of embolization or sudden cardiac death.10,18 In our patient, the tumor was successfully resected without complications. The immediate and complete resolution of symptoms observed postoperatively underscores the effectiveness of early surgical intervention.10,19 Histopathological confirmation remains the gold standard for diagnosing myxomas, with characteristic features including myxoid stroma, stellate cells, and vascular channels. 2 Gamna-Gandy bodies, seen in this case, are rare and may indicate chronic microhemorrhages within the tumor. 20 The prognosis following complete resection is excellent, with recurrence rates of 2% to 6% typically associated with incomplete resection or familial syndromes like the Carney complex.6,19 Regular follow-up with surveillance echocardiography is essential for the detection of recurrence. 8
Conclusion
This case underscores the clinical sequelae of RAMs in precipitating rHF. Through mechanical obstruction of venous return and impeded tricuspid inflow, these tumors can result in incipient rHF, warranting early diagnosis and surgical intervention. Complementary imaging modalities and a multidisciplinary approach are instrumental in optimizing outcomes. Long-term surveillance is also imperative to monitor for interval recurrence.
Footnotes
Acknowledgements
None.
Author Contributions
All authors contributed equally to writing the manuscript, and all authors read and approved the final manuscript.
Data Sharing Statement
All available data can be obtained by contacting the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance With Ethics Guidelines and Standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
