Abstract
Methimazole is commonly prescribed for patients with hyperthyroidism. It typically exhibits a well-tolerated profile, with common side effects including gastrointestinal disorders and rash. However, more serious rare yet adverse reactions, notably agranulocytosis and hepatotoxicity have been documented in literature. Here we present a case of a 27-year-old female, recently diagnosed with Graves’ disease, who was prescribed methimazole and developed severe pruritus with cholestatic jaundice 13 days later. Concomitant causes of liver disease were ruled out. The treatment was discontinued, and a switch to corticosteroid therapy with a regimen of radioactive iodine sessions was initiated. The patient’s condition showed a resolution of pruritus and jaundice, a disappearance of cytolysis with an aggravation of cholestasis followed by a gradual decrease, leading to the liver function normalization after 2 years. Methimazole-induced cholestatic jaundice is a rare yet severe adverse effect. Patients should be aware of this complication and advised to immediately stop taking the treatment when suggestive symptoms (pruritus, jaundice, dark urine, light-colored stool) occur.
Introduction
Methimazole is frequently prescribed as a pharmaceutical agent for the treatment of hyperthyroidism. It is generally well-tolerated. Commonly observed side effects include gastrointestinal disturbances such as indigestion, vomiting, and rash. 1 Nonetheless, literature documents more severe adverse reactions, prominently featuring agranulocytosis and hepatic involvement. The accurate mechanisms underpinning these adverse effects remain elusive. 2 Herein, we present a clinical case detailing Methimazole-induced cholestatic jaundice in a 27-year-old female patient, and we compare this case with current literature data.
Case Presentation
A 27-year-old female patient, without notable medical history, was diagnosed with Graves’ disease (GD) in December 2020. The diagnosis was confirmed based on suggestive clinical signs including chronic fatigue accompanied by paroxysmal palpitations persisting for approximately 2 months. Physical examination revealed sinus tachycardia with a heart rate of 102 beats per minute, associated with exophthalmos. Biological investigations demonstrated markedly reduced Thyroid Stimulating Hormone (TSH) levels and elevated Free Thyroxine 4 (FT4) levels in laboratory tests (TSH = 0.002 mU/L, FT4 = 30.08 pmol/L). She was subsequently admitted to the endocrinology department of La Rabta Hospital in Tunis. The pre-therapeutic assessment, including liver function tests, revealed no abnormalities. Thus, she was put on propranolol and methimazole at a daily dose of 120 and 20 mg, respectively. Thirteen days post-initiation of treatment, she manifested severe pruritus along with cutaneous-mucosal jaundice. Hepatic assessment revealed cytolysis (Aspartate Transaminase (AST) = 95 U/L, Alanine Transaminase (ALT) = 117 U/L), cholestasis (Gamma-glutamyl Transpeptidase (GGT) = 103 U/L, Alkaline Phosphatase (ALP) = 759 U/L), and predominantly conjugated hyperbilirubinemia (total bilirubin/direct bilirubin = 412/400 μmol/L). Methimazole was consequently discontinued, and the patient was initiated on ursodeoxycholic acid (UDCA) at a dosage of 13 mg/kg/day, along with oral corticosteroid therapy using prednisone at a dosage of 1 mg/kg/day. She was, then, referred to the Gastroenterology Department “B” at La Rabta Hospital in Tunis for further care. The clinical course was characterized by a marked reduction in pruritus and jaundice, accompanied by a regression of cytolysis in laboratory parameters and a gradual exacerbation of cholestasis (Table 1). A follow-up thyroid assessment revealed a slight increase in TSH levels alongside a decrease in FT4 levels, which remain above the laboratory’s biological reference ranges (TSH = 0.01 mU/L, FT4 = 25 pmol/L). A comprehensive etiological assessment, including viral serologies, immunological profiling, and biliary magnetic resonance imaging (Bili-MRI), yielded negative results. An associated seronegative primary biliary cholangitis was suspected at this juncture due to the persistence of cholestasis, but it was subsequently ruled out by liver biopsy. However, liver biopsy specimen demonstrated portal and periportal fibrosis, characterized by infrequent septa with portal spaces containing a lymphocytic and plasmocytic infiltrate. This infiltrate induces hepatocyte necrosis with the limiting plate forming an interface hepatitis. This histological presentation may correspond to autoimmune hepatitis or drug-induced liver injury (Figure 1). In collaboration with endocrinologists, the corticosteroid therapy was gradually discontinued. Hydrocortisone treatment was initiated at a dosage of 30 mg/day, and a regimen of radioactive iodine sessions commenced in February 2021. Biochemically, the progression was characterized by a transition to hypothyroidism (TSH = 3.01 mU/L, FT4 <0.42 pmol/L), requiring the initiation of hormonal substitutive treatment (Levothyroxine) at a daily dose of 75 µg. Cholestatic parameters remained stable over a 12-month period, followed by a gradual regression, ultimately leading to the liver tests complete normalization by January 2023.
Evolution of Biological Parameters.

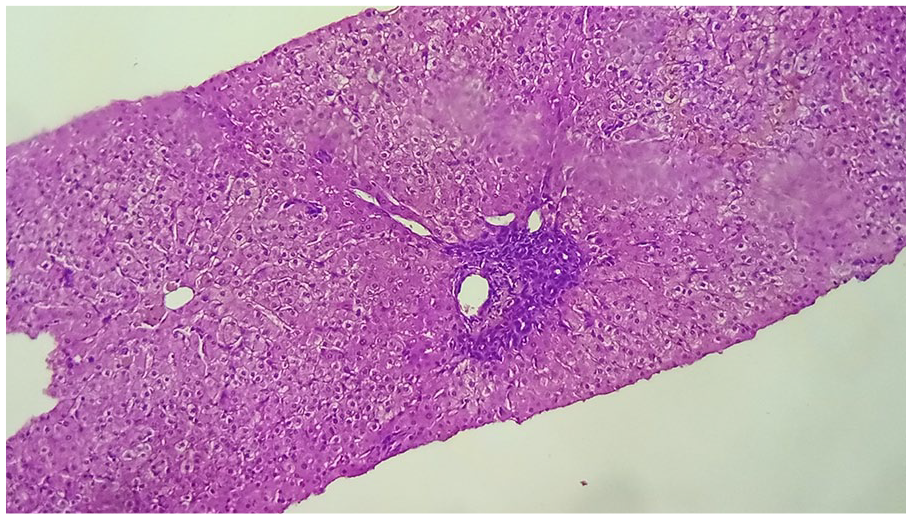
Liver biopsy specimen demonstrating portal and periportal fibrosis, characterized by infrequent septa with portal spaces containing a lymphocytic and plasmocytic infiltrate. This infiltrate induces hepatocyte necrosis with the limiting plate forming an interface hepatitis.
Discussion
The approach to managing hyperthyroidism revolves around 3 therapeutic options: medical treatment represented by antithyroid drugs (propylthiouracil, methimazole, and carbimazole), radioiodine therapy, and surgical treatment. Outside the United States, where radioactive iodine remains the treatment of choice, antithyroid drugs are the preferred first-line treatment. 3 Antithyroid drugs are usually well-tolerated. While rare, severe adverse effects have been documented in the literature, including agranulocytosis, hepatotoxicity, and the occurrence of vasculitis, typically associated with antineutrophil cytoplasmic antibodies. 3 Hepatotoxicity is extremely rare, occurring in approximately 0.1% to 0.2% of patients using antithyroid drugs. 4 A review of various cases reported in the literature has suggested a predominantly female prevalence and a selective risk among Asian patients. 5
The mechanisms underlying the occurrence of cholestasis during methimazole treatment remain poorly elucidated. Indeed, several assumptions have been proposed to explain this manifestation, including relative hypoxia leading to hepatocellular injury, the emergence under treatment of a rare condition such as Gilbert’s or Dubin-Johnson syndrome, the release of a hepatotoxic metabolite during methimazole hepatic metabolism, or a potential allergic reaction to the medication. 2 Another hypothesis is that the hepatotoxicity encountered during methimazole treatment may be induced by an imbalance between the oxidant and antioxidant systems caused by this molecule. 6 Indeed, methimazole-induced hypothyroidism appears to cause cellular damage by increasing the release of free radicals, enhancing lipid peroxidation, and inhibiting the protective enzymatic system, particularly the enzyme catalase. 6 Currently, the hypothesis of a hypersensitivity allergic reaction is the most widely acknowledged. Furthermore, recent studies have identified a susceptibility in patients with specific Human Leukocyte Antigens (HLA) subtypes to develop cholestasis under methimazole or hypersensitivity skin reactions to the drug. 7
In our case, hepatotoxicity manifested 13 days after the initiation of treatment, consistent with reported cases in the literature where this phenomenon tends to occur early, typically within 30 days of treatment initiation. 8 The management of such complications remains insufficiently standardized. In our case, we chose to discontinue the treatment, initiate corticosteroid therapy, and implement background hyperthyroidism management through radioiodine therapy. Various approaches have been documented in the literature. In some instances, adjusting the dosage in the presence of mild alterations in liver function tests has effectively controlled both the disease and the hepatotoxicity. However, discontinuation becomes imperative if jaundice occurs. 9 In other cases, the switch to another antithyroid drug, particularly propylthiouracil (PTU) proved to be successful. 2 Still, this approach has been questioned by a meta-analysis indicating a higher incidence of hepatotoxicity with PTU compared with methimazole. 10 Interestingly, another case, exhibiting similar patterns as our patient (progressive exacerbation of cholestasis post-treatment cessation) responded well to a corticosteroid regimen combined with UDCA, thereby reinforcing the hypothesis of a potential allergic reaction. 11
Hepatotoxicity typically resolves spontaneously following the discontinuation of treatment, with variable timelines ranging from 16 to 145 days in cases documented with propylthiouracil. 12 In some instances, hepatotoxicity proves irreversible. 13 In our patient, the progression was characterized by an initial exacerbation of cholestasis followed by a delayed normalization over a period of approximately 2 years. In this context, it was entirely justified to explore the possibility of another etiology for the persistent cholestasis, particularly an associated primary biliary cholangitis, especially given the backdrop of autoimmunity in our patient.
The question lingers as to whether a systematic follow-up assessment should be undertaken to identify this complication. European and American guidelines on hyperthyroidism management recommend an initial liver function test but suggest against routine monitoring of liver function.9,14 They propose conducting follow-up liver function tests only if the patient presents suggestive symptoms, given the rare and unpredictable nature of this complication. Nevertheless, some authors have advised for closer initial monitoring of liver function, particularly in light of the severity of this complication. 2
Conclusions
In conclusion, the occurrence of cholestatic jaundice is a rare but severe complication during methimazole treatment for hyperthyroidism. While current recommendations do not endorse routine monitoring of liver function, it is crucial to educate patients about the necessity of discontinuing the treatment in the presence of symptoms suggestive of hepatotoxicity. Managing this complication should ideally involve collaboration between endocrinologists and hepatologists, and thorough investigations should be pursued to rule out alternative causes that might explain the clinical and biochemical presentation.
Footnotes
Acknowledgements
I would like to express my gratitude to all those who have supported me throughout this endeavor. Your encouragement and assistance have been invaluable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
The abstract was not presented elsewhere.
