Abstract
Pulmonary nodules caused by mucosa-associated lymphomas (MALT lymphomas) are uncommon, comprising for less than 0.5% of all primary lung tumors. While biopsy is the main method for diagnosing the disease, the significance of differential pulmonary diagnosis based on imaging is often overlooked. In this report, we present a case of a patient with multiple disseminated nodules in the lungs, as well as severe anemia and incidental involvement of multiple lymph nodes. We discuss the differential diagnosis of pulmonary MALT lymphomas on computed tomography (CT) images and emphasize the significance of considering this uncommon entity in the evaluation of pulmonary nodules.
Keywords
Introduction
Primary pulmonary mucosa-associated lymphoma (MALT) lymphoma is a relatively fairly low-grade malignant tumor, accounting for less than 1% of all lung malignancies. 1 It originates in bronchus-associated lymphoid tissue (BLAT). 2 It is more commonly seen in middle-aged females and can be asymptomatic but may also present with symptoms such as cough, sputum, dyspnea, and systemic symptoms including bone marrow infiltration, fever, night sweats, and weight loss, patients often present with localized stage I or II disease, but in this stage IV report, the lymphoma was found to be disseminated at the time of diagnosis.
Case Report
A 57-year-old woman presented to local hospital with progressive dyspnea and low-grade fever. Her routine blood work showed abnormal levels of red blood (RB) cells at 1.46 × 1012/L and hemoglobin (HB) at 45 g/L. Despite treatment with piperacillin sulbactam and levofloxacin, her symptoms did not improve and she was subsequently admitted to our hospital. Physical examination was normal. A chest computed tomography (CT) scan (Figure 1) showed bilaterally multiple pulmonary nodules with air bronchograms. Laboratory tests were recommended upon admission to investigate the lung lesions (Table 1). The patient was treated with meropenem and voriconazole for 13 days but showed no improvement. Anemia was ruled out as her levels of VitB12 and FOLATE were norma, so a bone marrow aspiration and biopsy were performed which showed mainly lymphocyte proliferation (Figure 2). Flow cytometric immunophenotyping revealed a proliferation of B lymphocytes, consisting of 57.8% of all nuclear cells with, as well as CD19-positive B-cell making up 35.8% of all lymphocytes. All B Lymphocytes were presented positive for CD19 and lambda, and partial B-cells were revealed positive for FMC-7, CD79a, and CD20. The analysis showed that all lymphocytes were negative for CD5, CD7, CD117, CD13, CD34, CD33, CD14, CD103, CD23, CD25, CD15, CD41a, CD235a, CD3356, CD11b, CD64, CD138, CD11c, kappa, MPO, and cCD3. An ultrasound of the superficial lymph node revealed abnormal lymph nodes on both sides of the neck, supraclavicular area, left armpit, and bilateral groin, rang in size from 0.9 cm × 0.7 cm to 2.8 cm × 1.8 cm. A [ 18 F] Fluorodexyglucose-positron emission tomography ( 18 F-FDG PET/CT) scan showed highly metabolic lymph nodes on the left submandibular, neck, bilateral supraclavicular area, left axilla, mediastinum, hepatogastric space, retroperitoneum area, pelvic cavity, left gluteus maximus, right groin, as well as lesions and multiple high-metabolic nodes in both lungs, the left pleura, and bones. Further immunofixation revealed a negative result for myeloid differentiation factor (MYD88) and electrophoresis. After considering all of these examinations, the patient was diagnosed with marginal zone B-cell lymphoma (MZL).

Thoracic computerized tomography showed bilateral, multiple pulmonary nodules with air bronchogram in peribronchovascular and subpleural distribution (a, b, c, d), and identified feature of a well-circumscribed nodule or mass (a, d).
Laboratory Finding on Admission.

Abbreviations: ANA, antinuclear antibody; ACA, anticardiolipin.
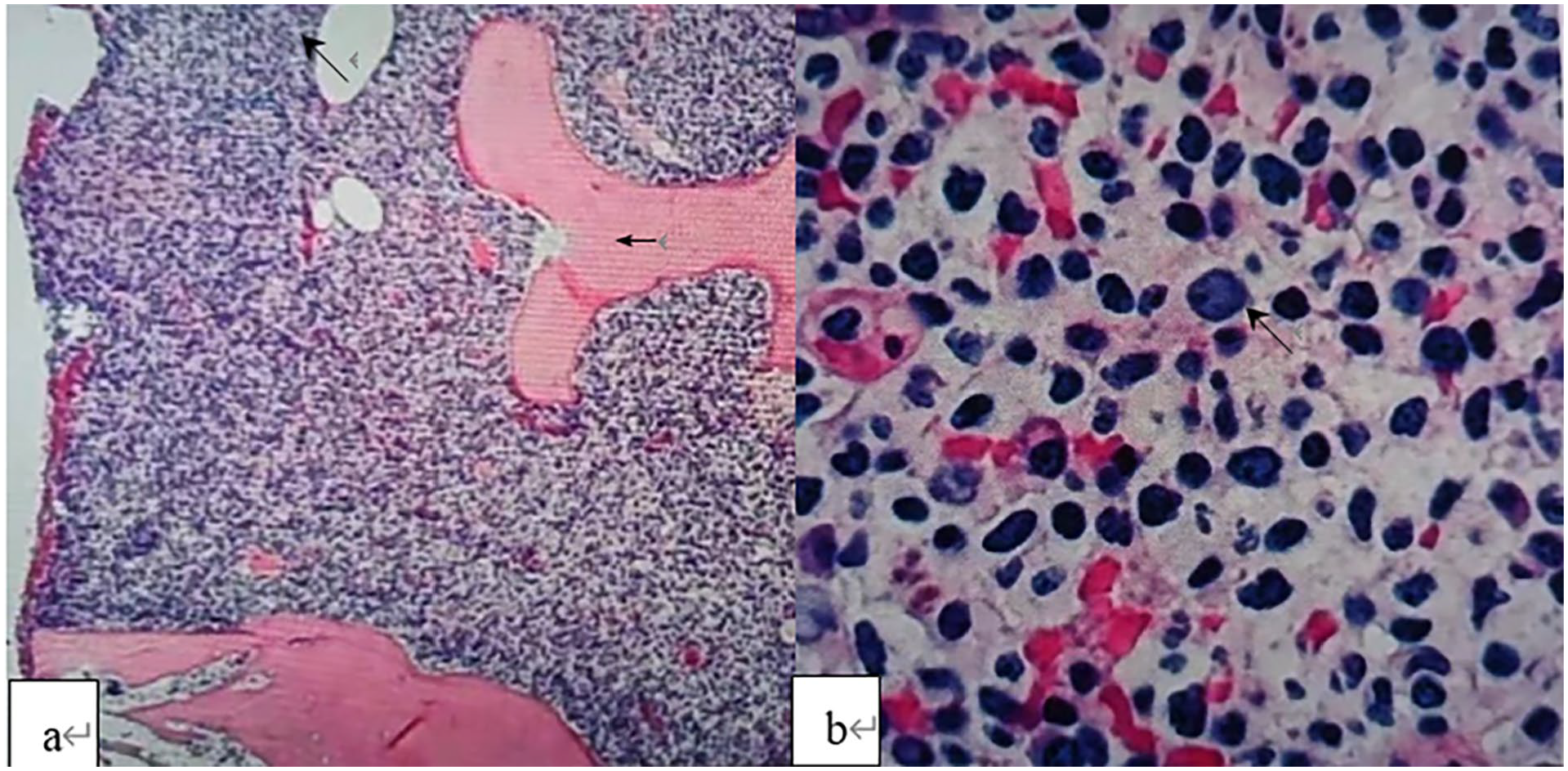
H&E and PAS staining features of bone marrow (transverse arrow) aspiration showed extremely active bone marrow hyperplasia (Oblique arrow), accounting for more than 90% (a). Bone marrow hyperplasia was dominated by oval or irregular nuclei lymphocytes which showed low cytoplasm and thick nuclear staining (b).
Discussion
Pulmonary MALT lymphoma is a type of the group of MZLs, which also includes nodal and splenic marginal-zone lymphomas. The development of MZL is frequently associated with long-term inflammation, particularly the Epstein-Barr (EB) virus, Mycobacterium tuberculosis, 3 and Chlamydia psittaci. 4 Immune stimulation—such as Sjogren’s syndrome, Hashimoto’s thyroiditis, and newly discovered IgG4-related diseases—has also been associated with MZL. Unique chromosomal rearrangement including (11;18) (q21;q21)/BIRC3-MALT1, t(14;18)/IGH-MALT1, t(11;14)(p22;q32)/IGH-BCL10, and t(3;14)(p14;q32)/IGHFOXP1, which affect the nuclear factor kappa B (NF-κB) signal, also play a significant role in pulmonary MALT lymphomas.5,6 In this patient, a number of abnormal immune indicators suggested that lymphoma may be a secondary lesion associated with connective tissue disease.
The diagnosis of MALT lymphoma is primarily determined through immunohistochemistry of the appropriate biopsies. The mandatory markers include CD20+, CD5− (with rare exceptions), CD10−, and Cyclin D1− (in cases positive for CD5). CD23±, IgD−, MYD88− (rare exceptions can occur) are suggested markers. In addition to routine immunohistochemistry, fluorescence in situ hybridization (FISH) analysis is used to the detection of chromosomal translocations involving IGH, MALT1, FOXP1, and BCL10, as well as numerical chromosomal abnormalities including deletions and trisomy of chromosome 3, 12, and 18. 7
Although a bone marrow biopsy is the main method for diagnosing pulmonary MALT lymphoma, it is clinically significant for clinicians to be able to diagnose it through imaging as well. The predominant abnormality of pulmonary MALT lymphoma on chest CT includes bilaterally (60%-70%) multiple (70%-77%) nodules, masses, and local consolidation. The persistently indolent lesions are ofen typically found in a peribronchovascular distribution. Indicative images of this type of lymphoma include air bronchograms within consolidations and well-defined masses.8,9 The lesions show increased filling and destruction of alveolar parenchyma toward their centers, tracking along bronchovascular bundles and interlobular septa at the periphery of the masses that making airways intact. 2 However, nonspecific images make it difficult to diagnose primary pulmonary MALT lymphoma.
The differential diagnostic considerations for pulmonary MALT are extensive, including noninfectious and infectious disease. Other conditions nodular lymphoid hyperplasia (NLH), lymphomatoid granulomatosis, and posttransplant lymphoproliferative disorder (PTLD) can share similar chest CT images and are associated with EB virus-infected B-cell proliferations. However, unlike MLAT lymphoma with multiple or bilateral lesions, the lesions of NLH are typically present with a small single area of lung, a well-circumscribed nodule or mass with an average diameter of 2 cm, or a focal mass-like consolidation. Nodular lymphoid hyperplasia may regress or remain stable for years. Typically, mediastinal and hilar lymphadenopathy are absent in NLH due to the noninvasion of the adjacent pleura or bronchus, as well as pleural effusion. While occasional focal lymphangitic extension (long glitch sign) may be seen in NLH; however, up to 30% of MALT lymphoma is involved in mediastinal and hilar lymphadenopathy. In contrast, necrosis is more common in lymphomatoid granulomatosis. The extent of necrosis can arrange from minimal to extensive necrosis due to the nature of angioinvasive and angiodestructive. 10 Furthermore, compared to MALT lymphomas, poorly marginated nodules in lymphomatoid granulomatosis can coalesce to form patchy consolidations which is another characteristic type. Multiple pulmonary nodules are also common findings in PTLD,10,11 but they are more often single rather than multiple, and the “Halo sign” around nodules is frequently seen in PTLD. Variable grafts within the first 2 years after transplantation can also be indicative of the condition. The differential diagnosis for nodular or mass-type in pulmonary MALT lymphoma should include IgG4-RLD. Although enlargement of the hilar or mediastinal lymph nodes of IgG4-RLD accounts 46% that may be seen in pulmonary MALT lymphoma, these nodules or mass of IgG4-RLD often have a focal spiculated appearance. 12
Pulmonary MALT lymphoma should be differentiated from a variety of infectious diseases including lobar pneumonia and invasive aspergillosis. The diameter and texture of the bronchi and bronchioles in consolidation of lobar pneumonia are normal, rather than showing lack of tapering or cystic bronchiectasis as seen in pulmonary MLAT lymphoma.13,14 Pulmonary MALT lymphoma can manifest as “angiogram” sign and a halo of ground-glass shadowing around the lesion margin, 14 but the nodules of invasive aspergillosis often present ground-glass “halo” sign in the first week, followed by the continuous damage-related “air crescent” sign at the second week. 15 In our case, the CT images did not show any signs of foliation or glitch associated with lung malignancy, and there was no patchy or lobar consolidation with a uniform density and well-margined distribution of lobar pneumonia.
Pulmonary MALT lymphoma shares the potential of spontaneous regression. For limited-stage nongastric MALT lymphoma, close observation and surgical resection as well as radiotherapy are options. For advanced-stage patients with a low tumor burden are asymptomatic, close observation or rituximab-based treatment should be considered. Rituximab had significantly regressed of pulmonary lesions, and median progression-free survival was 66.0 months after finishing single 4 to 8 cycles of rituximab. 16 In our case, the patient with a high tumor burden and organ impairment were symptomatic, and we administered 4 cycles of cyclophosphamide, doxorubicin, vincristine, and prednisone(CHOP) and 2 cycles of R-CHOP which led to the disappearance of most lesions over a period of 5 months. She has experienced no recurrence for more than 5 years since the diagnosis of the disease. Moreover, R-CVP regimen (rituximab, cyclophosphamide, vincristine, and prednisolone) and the BR regimen (bendamustineand rituximab) are the suggested therapies. 17 Although the pulmonary MALT lymphomas can slowly grow over a prolonged period of time, however, the recrudescence accounting for 32.8% is frequently occurred and it can progress to diffuse larger B-cell lymphoma.17,18
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their gratitude to the patient and their family for their great contribution. This article was funded by the Weifang Science and Technology Bureau (grant no. 2021YX080).
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
