Abstract
Bisphosphonate-related osteonecrosis of the jaw (BRONJ) occurs in the jawbone and interfacing oral mucosa of patients treated with bisphosphonates. Herein, we report novel histopathological findings in the oral mucosa of a surgical specimen obtained from a 61-year-old man with BRONJ. The resected jawbone and adjacent oral mucosa were separated for histological examination. The mucosal tissue was examined using Von Kossa staining and immunohistochemical (CK5/6, p63) staining of non-decalcified paraffin sections. Pseudoepitheliomatous hyperplasia (PEH), a microscopic feature of the mucosal epithelium in BRONJ, was observed in soft tissue specimens, concomitant with inflammatory cell infiltration. Von Kossa staining revealed small fragments of necrotic bone, tens to hundreds of micrometers in size, scattered within the connective tissues; the PEH forefront contacted some of the bone fragments. Immunohistochemical staining demonstrated that occasionally, the PEH not only contacted but also encompassed the bone fragments. To our knowledge, this is the first report of presence of micro bone fragments and their association with PEH in the oral mucosa in BRONJ.
Keywords
Introduction
Bisphosphonate-related osteonecrosis of the jaw (BRONJ) is a serious complication of nitrogen-containing bisphosphonate (BP) use. Although many studies have been conducted on BRONJ since its first report, 1 the exact pathophysiology of the disease remains unclear. As BP is an osteoclastic inhibitor that suppresses bone turnover, bone is considered the primary lesion site in this disease.2,3 Extensive histological studies of bone samples from patients with BRONJ have revealed osteomyelitis, necrotic bone with empty osteocyte lacunae, and bacterial colonies as histological features.4-6
Despite growing awareness regarding bone lesions in BRONJ, knowledge regarding its histopathological findings in the gingival or alveolar mucosa is lacking. This may be because most pathological specimens studied have consisted only of bone tissues, and mucosal tissues have not been examined as extensively. To date, inflammatory cell infiltration, epithelial proliferation—called pseudoepitheliomatous hyperplasia (PEH)—and obliterated vessels with numerous spindle-shaped cells have been identified as microscopic features of the mucosa in BRONJ.6-9 However, no further distinct histopathological findings have been reported in mucosal tissues. This study describes a novel histopathological finding in the soft tissue of a surgical specimen obtained from a patient with BRONJ.
Case Report
A 54-year-old man with complaints of bone exposure and pain in the left lower molar region was referred to our hospital. He had been diagnosed with BRONJ and received conservative treatment at a referral hospital for approximately 1 year. His medical history included osteoporosis secondary to chronic kidney disease. He had previously received a kidney transplant but experienced post-transplant renal failure. He had bilateral femoral head fractures and undergone arthroplasty when he was 51 years old. For osteoporosis treatment, zoledronate was administered for 6 years from the age of 48 years. Blood tests showed that his serum calcium and phosphorus levels were within normal limits. Biochemical markers related to calcium metabolism, such as parathyroid hormone (PTH), vitamin D, and markers for bone formation and resorption, were not examined. He was diagnosed with stage 2 BRONJ of the left mandible, and conservative treatment with antibiotics and lesion rinsing was initiated. The necrotic lesions gradually grew, and surgery was suggested. However, the patient was reluctant because he had undergone several major surgeries. Conservative treatment was continued upon the patient’s refusal to undergo surgery. Consequently, a pathological fracture of the left mandible occurred at the age of 60 years. Owing to mastication disturbances and pain, the patient consented to surgery at the age of 61 years.
Bisphosphonate-related osteonecrosis of the jaw was diagnosed to be at stage 3 at the time of surgery. Externally, the mandible deviated slightly to the left, and a fistula was observed on the skin of the left mental region. Intraorally, necrotic bone exposure was observed, extending from the left lower canine to the second molar region (Figure 1A). A bone exposure of approximately 5 mm was observed in the right lower premolar region. Orthopanoramic radiography revealed progressive osteolysis extending to the inferior border and an associated pathological fracture of the left mandible. In the right mandible, the osteolytic lesion was confined to the alveolar region of the molars (Figure 1B). Bisphosphonate-related osteonecrosis of the jaw was diagnosed to be at stages 3 and 2 in the left and right mandibles, respectively. The patient underwent segmental resection of the left mandible and marginal resection of the right mandible. The affected oral mucosa adjacent to the exposed bone on the left side was resected because it interfered with postsurgical wound healing (Figure 1A). The mucosal tissues were stripped and removed from the bone lesions in the layer corresponding to the periosteum prior to bone resection. The postoperative course was uneventful, and no BRONJ recurrence was observed during the 1-year follow-up (Figure 1C, D).

(A) Intraoral findings showing exposed bone (arrowheads) and the interfacing mucosa. Area of the resected oral mucosa (arrows). (B) Preoperative orthopanoramic radiograph. (C) Intraoral findings 1 year postsurgery. (D) Orthopanoramic radiograph 1 year postsurgery.
For histopathological examination, decalcified and non-decalcified paraffin sections were prepared from hard and soft tissue specimens, respectively. The bone specimens presented typical histological findings, including osteomyelitis, sequestrum with empty osteocyte lacunae, and colonies of Actinomyces-like bacteria (Figure 2A). Soft tissue specimens comprised the mucosal epithelium and subepithelial connective tissue (lamina propria). Infiltration of inflammatory cells, including lymphocytes, plasma cells, and macrophages, into connective tissues was observed. Pseudoepitheliomatous hyperplasia of the mucosal epithelium and elongation of the epithelial apices that deeply descended into the lamina propria were frequently observed in inflamed areas (Figure 2B). Moreover, small fragments of osseous tissues were scattered and buried within connective tissues. Those fragments were considered to be necrotic bone because they were positive for Von Kossa staining and contained empty osteocyte lacunae (Figure 3). The bone fragments were observed in 18/30 histological sections examined, with sizes ranging from 20 to 700 µm. Most of these micro bone fragments (MBFs) were associated with inflammatory cell infiltration, whereas others were found in areas with few inflammatory cells. Interestingly, the forefront of the PEH occasionally contacted MBFs in submucosal connective tissues (Figure 4). Contact between the PEH and MBFs was observed in 3 of the 30 histological sections examined; in addition, complete encompassment of MBFs by the PEH was observed (Figure 4). Immunohistochemical staining of epithelial markers (p63, CK5/6) not only clearly indicated that the PEH was in contact with MBFs but also that the epithelial cells in the apices of the PEH extended downward along MBFs, seemingly beginning to encompass them (Figure 5).

(A) Photomicrograph of the bone specimen showing osteomyelitis, empty osteocyte lacunae, and bacterial colonies (arrows) (hematoxylin and eosin staining, 100×, Bar: 100 µm). (B) Photomicrographs of the soft tissue specimens showing pseudoepitheliomatous hyperplasia in the epithelium and inflammatory cell infiltration in the subepithelial connective tissue (hematoxylin and eosin staining, 40×, Bar: 500 µm).

(A-C and D-E) Representative photomicrographs showing micro bone fragments buried within the connective tissue, located away from the epithelium. (A, D) Hematoxylin-eosin and (B, E) Von Kossa staining of the serial sections of (A) and (D) (40×, Bar: 500 µm). (A, B) Single bone fragment is seen (arrowheads). (D, E) Various sizes of bone fragments are seen (arrowheads). Hard tissues are partly detached due to non-decalcification (*). (C, F) High-magnification view of the bone fragments in A and D. Empty osteocyte lacunae are seen within the fragments (arrows) (100×, Bar: 100 µm).
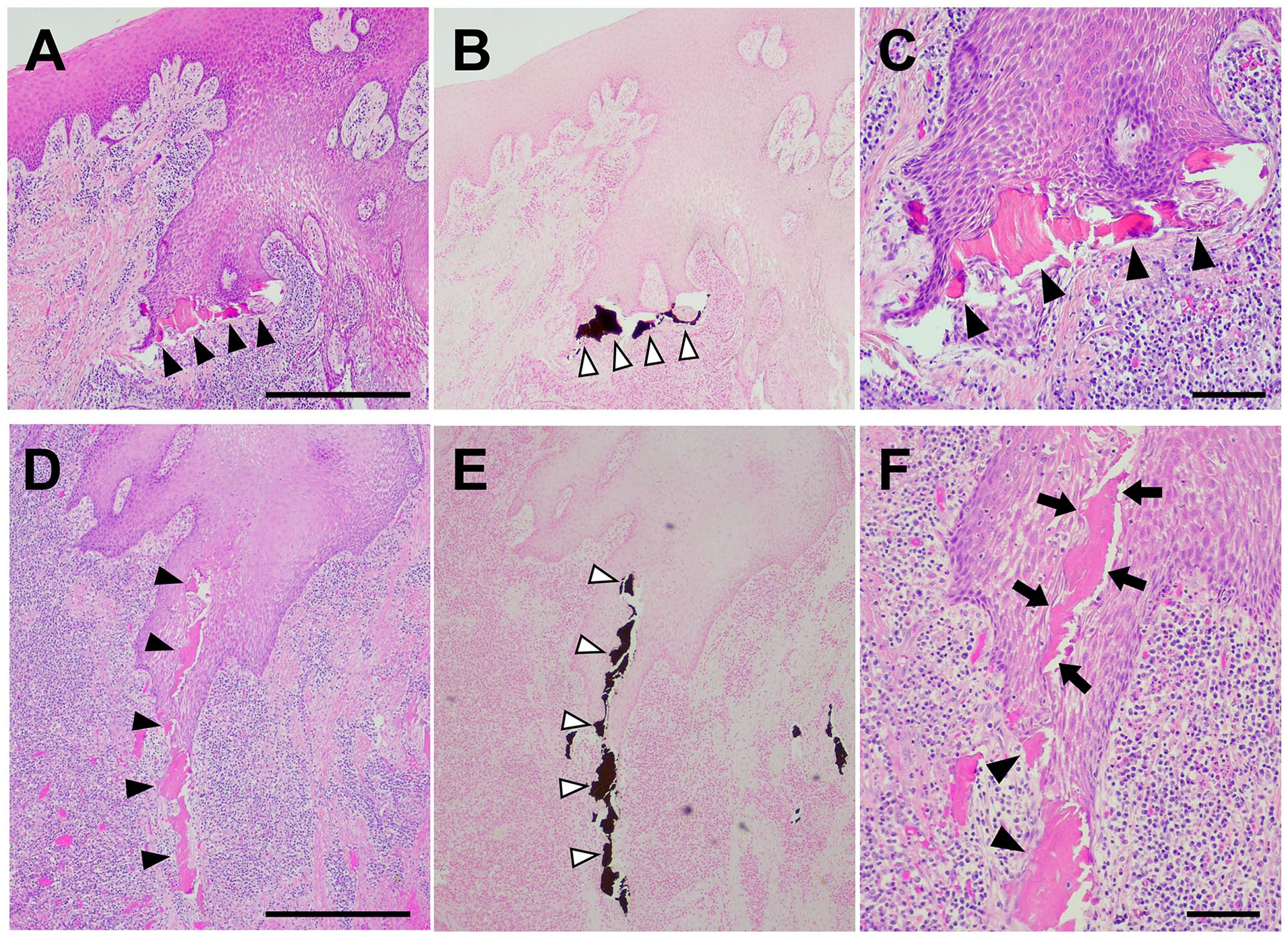
(A-C and D-F) Photomicrographs showing PEH contacting MBFs. (A, D) Forefront of the PEH contacting MBFs of various sizes (arrowheads) (hematoxylin and eosin staining, 40×, Bar: 500 µm). (B, E) Von Kossa staining of the serial sections of (A) and (B) (40×). (C, F) High-magnification images of A and D (100×, Bar: 100 µm). MBFs contacted (arrowheads) or encompassed (arrow) by PEH.

(A, B) Hematoxylin and eosin staining showing MBFs in contact with the PEH (40×, Bar: 500 µm). (C, D) High-magnification view of the square area in (A) and (B) (200×, Bar: 50 µm). (E, F and G, H) Immunostaining of the serial sections of (C) and (D) demonstrating epithelial cells extending downward along the MBF (arrowheads in E, G) or encompassing the MBF (arrowheads in F, H) (200×). (E, F) p63 and (G, H) CK5/6.
Discussion
Two decades have passed since Marx first described a series of patients with exposed and non-healing jawbones after BP treatment. 1 Microscopically, advanced BRONJ lesions are characterized by purulent inflammation, inflamed granulation tissue, presence of bacteria, and necrotic bone sequestra.4-7,9 Specific histopathological findings of BRONJ compared with those of common infectious osteomyelitis and radiogenic osteonecrosis of the jaw have been extensively studied. However, histopathological findings are not sufficient to distinguish bone lesions in BRONJ from those in the aforementioned diseases.10,11 Although knowledge regarding bone lesion has been accumulated, soft tissues have not been examined in detail as extensively.5,8
Herein, MBFs were scattered in the subepithelial area of the oral mucosa. This finding was observed in 18/30 histological sections examined. The presence of such bone fragments in the oral mucosa has not been previously reported because most studies have investigated bone-based decalcified tissues taken from BRONJ lesions4,5,7,12,13 and, therefore, had few opportunities to observe soft tissues, especially those with sufficiently preserved morphology for analysis. This enabled us to identify MBFs; the hard and soft tissues were separated beforehand and an undecalcified method of processing was chosen for the soft tissue specimens. The presence of hard tissues during sectioning was first noticed because chattering, an artifact that occurs during paraffin block thinning of hard specimens, was experienced frequently. To date, there are few reports on intramucosal calcified substance or calcification in BRONJ. Meiller et al 14 previously focused on soft tissues and examined surgical specimens that contained not only bone but also adjacent oral mucosa from 10 cases of BRONJ using zoledronic acid. They performed Von Kossa staining on paraffin sections of these specimens and observed calcium deposition in submucosal connective tissues in 8/10 cases. They suggested that calcium deposition in the submucosal tissues was similar to that observed in non-traditional calciphylaxis, which occurs in certain hypercalcemic states and leads to soft tissue ischemia, necrosis, and non-healing lesions. 15 Whether the calcium deposition observed by Meiller et al on decalcified sections is related to MBFs seen in the non-decalcified sections from our patient remains unclear. In either calcium deposition or scattering of MBF, such ectopic calcified substances in connective tissues may contribute to the initiation and persistence of BRONJ.
It remains unclear when and how these bone fragments originate from the jawbone and migrate to the submucosal connective tissue. These bone fragments were surrounded by connective tissues, indicating that they had not formed during surgical resection but was already present within submucosal connective tissues. As a phenomenon related to bone collapse, microcrack accumulation has been reported in bone samples from patients with BRONJ16,17 and animal models18-21 using scanning electron microscopy. The jawbone experiences a high degree of mechanical stress owing to masticatory forces, which continuously leads to microcrack formation in the jawbone. Normally, these microcracks are continuously repaired. However, long-term BP administration leads to microcrack accumulation due to reduced bone remodeling caused by suppressed osteoclast function.18-21 Previously, microcracks were observed in 54% of bone samples obtained from patients who underwent intravenous BP therapy, in contrast to samples obtained from patients with osteoradionecrosis and osteomyelitis in which no microcrack accumulation was detected. 17 Unrepaired microcrack accumulation may cause necrotic bone surface collapse due to mechanical stress and scattering of the bone fragments within the mucosal tissues.
Pseudoepitheliomatous hyperplasia is a benign reactivated epithelial lesion microscopically characterized by irregular downward epithelial projections. This relatively common feature found in the affected oral mucosa of patients with BRONJ7,8 was also observed herein. Pseudoepitheliomatous hyperplasia is a well-known phenomenon in dermatology as a benign proliferation of the epidermis extending down into the dermis.22,23 It mimics squamous cell carcinoma but presents no cytological atypia or mitotic figures. This reactive proliferation of the epithelium occurs secondary to persistent inflammation or chronic infection resulting from chronic trauma, microbial infection, degenerative changes, or the retention of foreign material.23-25 Many studies have associated this condition with various skin diseases. Pseudoepitheliomatous hyperplasia is also associated with various inflammatory conditions in the oral mucosa.7,8,26,27 Pseudoepitheliomatous hyperplasia in patients with BRONJ was first described by Hansen et al, 4 who reported that PEH was observed in 5/8 patients with BRONJ. Zustin et al 8 reported that PEH was observed in 11/17 patients with BRONJ. However, the function of PEH in the oral mucosa remains unclear.
Dermatologically, PEH is thought to act as a defense mechanism facilitating the elimination of foreign bodies. 28 Cutaneous PEH arises due to the release of various cytokines produced by inflammatory cells and migrates foreign material from the dermis to the exterior via the stratum corneum.24,29 This phenomenon is known as transepidermal elimination (TE). The biological concept of TE was formulated by Pinkus in 1954 30 and established by Mehregan as a common response to a number of skin diseases. 31 The TE of calcium-containing materials has been previously reported in several instances. In nodular-type calcinosis cutis, the epidermis showed areas of PEH engulfing masses of calcified material, and occasionally formed channels through which calcified particles moved to the surface. 32 It has also been reported that calcium deposition in the skin following the use of calcium-containing materials results in the TE of the materials.33,34 These reports suggest that calcium-containing foreign bodies are susceptible to TE. Herein, we showed that PEH was in contact with—in some cases even encompassed—MBF, mimicking TE in the skin. To our knowledge, this is the first report demonstrating the presence of MBF and its association with PEH in the oral mucosa of patients with BRONJ lesions.
Regarding our findings, because the bone fragments were necrotic, they could have been deemed calcium-containing foreign bodies and activate inflammation. Moreover, as MBF contains BP (zoledronate in this case) and is not absorbed by osteoclasts, it may be retained for a long period within the tissues. Such residual bone fragments may prolong chronic inflammation and trigger elimination via PEH. The histological finding of PEH being in contact with MBFs was occasionally observed in 3 of the 30 histological sections examined. Therefore, it remains unclear whether the TE-like phenomenon of MBF is common among patients with BRONJ or depends on the severity or duration of the disease. It is also important to examine whether the TE-like phenomenon of MBF is observed in denosumab-induced osteonecrosis of the jaw. This study has limitations as the results are based on a single patient. Further research on gingival and alveolar mucosa of patients with BRONJ is required to address these questions. As there are few opportunities to examine oral mucosa with morphologies sufficiently preserved for analysis in a clinical setting, animal studies may be helpful in explaining the possible relationship between MBF and PEH.
Footnotes
Author Contributions
GK contributed to the investigation and wrote the original draft. AH contributed to the methodology. AT contributed to imaging. RY acquired resources. TM contributed to the writing, reviewing, and editing of the manuscript. KS validated the findings. MW supervised the study. NA conceptualized the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases.
Informed Consent
Informed consent for the publication of this case report was obtained through an opt-out methodology defined in our institution.
