Abstract
Primary cardiac lymphoma is an exceedingly rare malignant tumor, with diffuse large B-cell lymphoma (DLBCL) being the most prevalent histological subtype. This disease has non-specific clinical manifestations, making early diagnosis crucial. However, DLBCL diagnosis is commonly delayed, and its prognosis is typically poor. Herein, we report the case of a 51-year-old male patient with DLBCL who presented with recurrent chest tightness for 4 months as the primary clinical symptom. The patient was admitted to the hospital and diagnosed with acute myocardial infarction and left ventricular hypertrophy with heart failure. Echocardiography revealed a progression from left ventricular thickening to local pericardial thickening and adhesion in the inferior and lateral walls of the left ventricle. Finally, pathological analysis of myocardial biopsy confirmed the diagnosis of DLBCL. After treatment with the R-CHOP chemotherapy regimen, the patient’s chest tightness improved, and he was discharged. After 2 months, the patient succumbed to death owing to sudden ventricular tachycardia, ventricular fibrillation, and decreased blood pressure despite rescue efforts. Transthoracic echocardiography is inevitable for the early diagnosis of DLBCL, as it can narrow the differential and guide further investigations and interventions, thereby improving the survival of these patients.
Keywords
Introduction
Primary cardiac lymphoma (PCL) is a rare malignant tumor with various clinical manifestations, and diffuse large B-cell lymphoma (DLBCL) is the most common subtype. The median survival of patients with DLBCL is <1 month without chemotherapy. 1 Owing to the location and vague signs and symptoms of PCL lesions that invade the heart, PCL diagnosis can be challenging, often leading to delayed diagnosis and poor prognosis.2,3 There are various symptoms and clinical features of PCL with cardiac involvement, such as dyspnoea, arrhythmia, pericardial effusion, chest pain, and chest tightness. Prompt diagnosis and treatment are crucial in preventing fatal outcomes. 4 In this report, we present the case of a male patient who was successively diagnosed with acute myocardial infarction and hypertrophic cardiomyopathy with heart failure based on his clinical manifestations and echocardiography findings, which revealed progression from left ventricular thickening to local pericardial thickening and adhesion in the inferior and lateral walls of the left ventricle. Finally, myocardial biopsy and pathological examination confirmed the diagnosis of DLBCL. The patient showed symptom improvement and was discharged after receiving R-CHOP chemotherapy. After 2 months, the patient succumbed to death owing to sudden ventricular tachycardia, ventricular fibrillation, and a drop in blood pressure despite rescue efforts.
Case Presentation
A 51-year-old male patient was admitted to the hospital because of recurrent chest tightness for 4 months and further exacerbation for 2 weeks. The patient experienced chest tightness and discomfort 4 months ago without notable predisposing factors, and no significantly abnormal signs were noted during the physical examination. The electrocardiogram (ECG) analysis indicated acute high lateral myocardial infarction and T wave changes (Figure 1). The levels of troponin T (254.9 pg/mL), creatine kinase (CK; 440 U/L), creatine kinase isoenzyme MB (CK-MB; 30.3 U/L), myoglobin (103.1 ng/mL) and measured using Philips IU22 color ultrasonic diagnostic instrument demonstrated 70% left ventricular ejection fraction, left heart enlargement, left ventricular wall thickening, left regional ventricular wall motion abnormality, moderate mitral regurgitation, a small amount of pericardial effusion, and normal left ventricular systolic function (Figure 2A). The medical team diagnosed acute high lateral myocardial infarction and selective coronary angiography after coronary showed no clear stenosis of the left and right coronary arteries, with thrombolysis in myocardial infarction (TIMI) grade 3 flow. The possibility of coronary artery spasm was considered because no abnormal findings were found during coronary angiography.
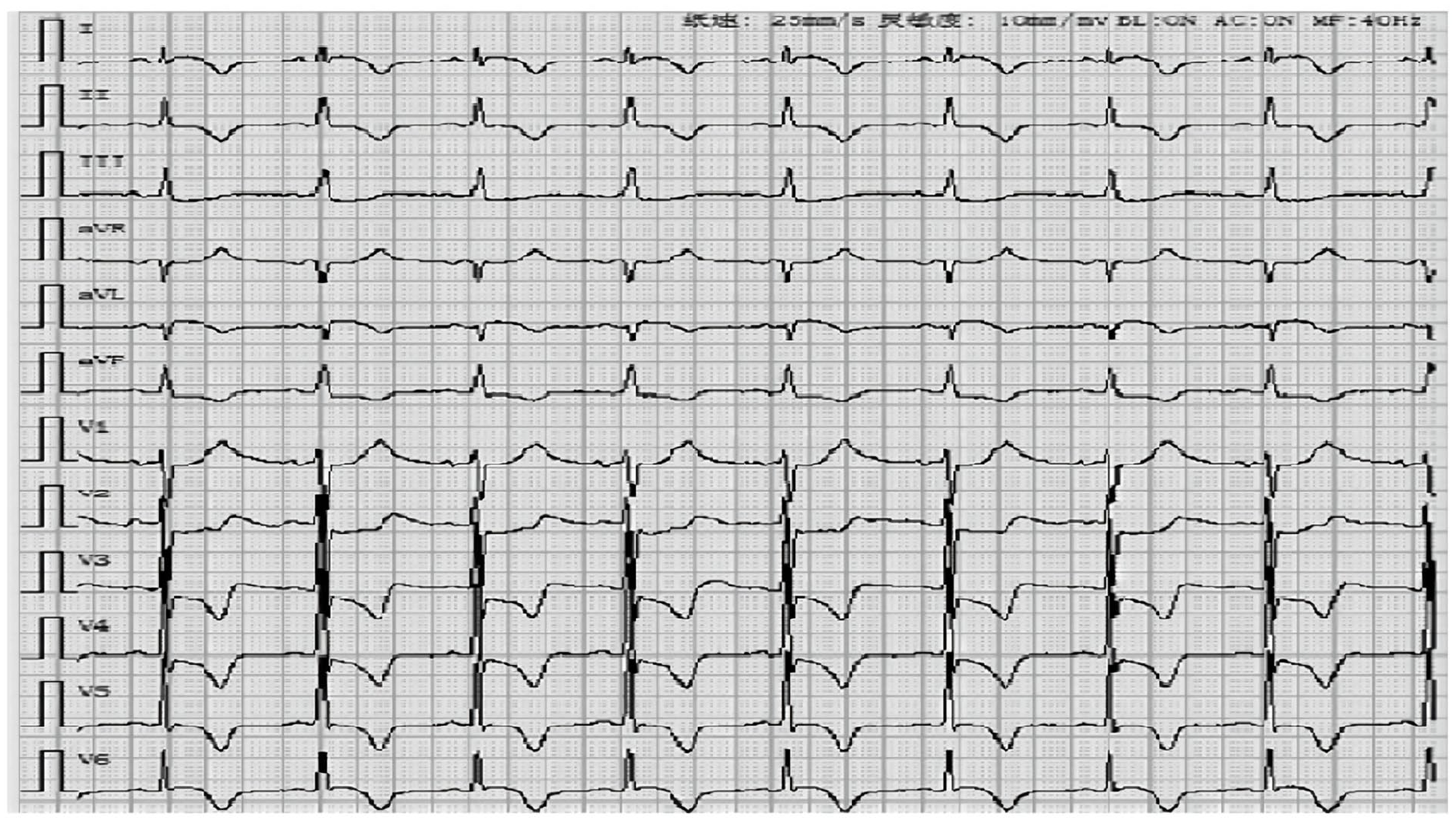
Electrocardiogram at admission. ECG at the initial admission showed (I) an elevated ST segment in AVL lead and (II and III) changes in AVF and V2–V6 ST-T waves, indicating a high lateral myocardial infarction.

Changes in transthoracic echocardiography. (A) Echocardiography at admission showed left ventricular thickening. (B) Echocardiography 1 month after discharge showed local pericardial thickening and adhesion in the inferior and lateral walls of the left ventricle during outpatient follow-up. (C) Echocardiography at the second admission showed extensive involvement in the aorta, peripulmonary arteries, left ventricle, and left atrium. (D) Echocardiography 4 months after the initial admission showed extensive involvement in the aorta, peripulmonary arteries, left ventricle, and left atrium.
The patient received secondary postoperative prophylaxis for coronary heart disease, including antiplatelet aggregation and plaque stabilization. The outpatient follow-up examination was performed 1 month after discharge, and the patient was found to have chest tightness and difficulty in breathing. Wet rales were heard in both lungs. The ECG results indicated sinus rhythm, ST-T changes, local pericardial thickening, and adhesion in the inferior and lateral walls of the left ventricle (Figure 2B). The levels of myocardial enzymes and troponin were normal. The level of N-terminal B-type natriuretic peptide (NT-ProBNP) was 5407.0 pg/mL. The possibility of left ventricular hypertrophy with chronic heart failure was considered, and the medical team initiated systemic treatment, such as positive inotropic diuretic therapy, to control heart failure. After 2 months, the patient was re-hospitalized because of recurrent chest tightness and discomfort. Follow-up echocardiography revealed the aorta, peripulmonary artery, left ventricle, and left atrium to be extensively affected. Rheumatic immune diseases could not be ruled out, and the medical team suspected IgG4-related diseases. The patient’s left ventricular systolic and diastolic function decreased (Figure 2C), and the levels of the tumor markers CA125 and neuron-specific enolase were abnormal. The patient underwent positron emission tomography-computed tomography (PET-CT), which revealed space-occupying lesions in the aorta, peripulmonary arteries, aortic root, and interventricular septum and around the left atrium and ventricle. Multiple lymph nodes were observed in the mediastinum, bilateral internal mammary area, and cardiac diaphragm angle, with increased glucose metabolism. Myocardial biopsy and immunohistochemistry revealed non-Hodgkin’s lymphoma, B-cell tumor, and DLBCL with germinal center B-cell (GCB)-like subtype (Han’s classification). The patient was diagnosed with cardiac DLBCL with myocardial damage and chronic congestive heart failure. After managing heart failure, the patient received the first course of chemotherapy: cyclophosphamide (0.45 g on days 1–3), epirubicin (40 mg on day 1, 45 mg on days 2–3), vindesine (2 mg on days 1–2), rituximab (600 mg on day 4), and dexamethasone (10 mg on days 1–5). The patient’s chest tightness improved after administering this therapy, and he was discharged. At 1 month follow-up, he reported that his chest tightness was better than before. However, echocardiography still showed extensive involvement in the aorta, peripulmonary arteries, left ventricle, and left atrium (Figure 2D), suggesting a poor prognosis. 5 One month after this follow-up, the patient succumbed to death owing to a sudden episode of ventricular tachycardia, ventricular fibrillation, and decreased blood pressure despite resuscitation efforts (Figure 3).

Timeline showing the clinical course in this patient.
Discussion
Extra medullary manifestations, such as leukemia, lymphoma infiltration and multiple myeloma, are less commonly observed in cardiac hematological malignancies than in other malignant cardiac tumors. 6 Cardiac lymphoma is categorized as primary and secondary cardiac lymphoma, which mainly affects the heart and/or pericardium. Primary cardiac lymphoma is an extremely rare and unusual malignant tumor, accounting for <1.2% of all cardiac tumors and <1.0% of extranodal lymphoma. Almost all cardiac lymphomas are B-cell diseases, with DLBCL being the most frequent histopathology, accounting for approximately 80% of PCL. 7 Primary cardiac lymphoma is highly prevalent among elderly men. Patients with PCL exhibit different symptoms depending on the location, size, growth rate, invasion, and fragility of the tumor. The right atrium, right ventricle, left ventricle, left atrium, atrial septum, and ventricular septum are the most common sites of cardiac involvement. In one third of patients, the pericardium is affected, and in <10% of cases, the left ventricular cavity is affected. Valve involvement is uncommon, with right ventricular system involvement being the most common type.2,8-10
In this report, transthoracic echocardiography reflected the progression of DLBCL in the patient. Initially, the patient presented with chest tightness, ST-T changes in ECG, and increased troponin level. Echocardiography revealed abnormal left regional ventricular wall motion, and coronary angiography revealed no abnormalities. This suggested that DLBCL initially invaded the left heart, which is not a commonly reported tumor invasion site. This finding indicated that myocardial infarction was not caused by coronary artery disease but by direct invasion, compression, and damage of the ventricular muscles due to DLBCL. The increased wall tension of the ventricle may have led to coronary artery spasm and necrosis caused by compression, and myocardial necrosis was possibly induced by toxic substances released by the tumor, resulting in non-obstructive coronary artery disease. When DLBCL directly invaded and compressed the heart, echocardiography revealed local pericardial thickening and adhesion in the inferior and lateral walls of the left ventricle, leading to the diagnosis of left ventricular hypertrophy. However, the treatment effect for this disease was poor, PET-CT showed that the tumor caused diffuse cardiac invasion, there were apparent space-occupying lesions in the cardiac cavity, and DLBCL was later diagnosed through myocardial biopsy. After evaluating his severe cardiac insufficiency, radiotherapy was not used to alleviate the symptoms.
The symptoms of DLBCL invading the heart exhibit atypical characteristics. Depending on the tumor’s anatomical location within the heart, patients may present with various clinical manifestations, including arrhythmia due to compressed cardiac conduction systems and heart failure symptoms resulting from obstructed blood flow. 11 Currently, no specific biomarkers for early DLBCL diagnosis exist, and tumor biopsy or pathological examination of metastatic tissue is warranted for a definitive diagnosis. Transthoracic echocardiography is a non-invasive examination and the most convenient method for the early diagnosis of PCL. It can confirm the presence of cardiac masses or pericardial effusion.12,13 In this patient, transthoracic echocardiography reflected the progression of DLBCL during the diagnosis and treatment processes. Such indications can alert clinicians to avoid misdiagnosis and mistreatment that may significantly impact the prognosis of patients with DLBCL.
Cardiac involvement with hematologic neoplasms is of more than academic interest as this complication is likely to respond to radiotherapy. Proper irradiation dose and timing of treatment can relieve symptoms and provide opportunities for follow-up treatment of patients with DLBCL invading the heart. 14-16 Compared to patients with DLBCL with cardiac infiltration in previous literature, the patient in this case did not achieve long-term survival. Whether the treatment was related to the failure of radiotherapy required further exploration.
Conclusion
Diffuse large B-cell lymphoma invading the heart is rare and associated with a poor prognosis. We presented the case of a male patient with DLBCL who was successively diagnosed with acute myocardial infarction, left ventricular hypertrophy with heart failure, and ultimately with DLBCL based on myocardial biopsy and pathological examination. Early diagnosis and prompt treatment are crucial for improving the survival rate of patients. We found that transthoracic echocardiography can narrow the differential and guide further investigations and interventions, thereby improving the survival of these patients. Cardiac magnetic resonance imaging should be pursued after determining that obstructive coronary artery disease has been excluded or cannot fully account for the patient’s myocardial ischemia.2,8 Further investigations, such as PET-CT imaging and myocardial biopsy, may be necessary to confirm the diagnosis. This approach can help prevent misdiagnosis and mistreatment, which can severely impact the patient’s prognosis.
Footnotes
Author Contributions
J.M., T.D., and T.C. analyzed and interpreted the patient data regarding the hematological disease. C.C. and B.W. performed the transthoracic echocardiography examination of the heart. Z.L. and X.P. were major contributors to writing the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the First Batch of High-level Talent Scientific Research Projects of the Affiliated Hospital of Youjiang Medical University for Nationalities in 2022 (R202210305).
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Availability of Supporting Data
Data are available on request from the corresponding author.
