Abstract
The concurrent development of abducens nerve palsy and optic neuritis on the same side is rare. Here we presented an 82-year-old man who developed the combination of abducens nerve palsy and optic neuritis on the left side 2 months after the sixth inoculation of COVID-19 mRNA vaccine. In past history at 45 years old, he experienced subarachnoid hemorrhage and underwent surgery for the clipping of intracranial aneurysm. The patient had no systemic symptoms, such as general fatigue, fever, arthralgia, and skin rashes. Physical and neurological examinations were also unremarkable. Since the aneurysmal metal clip used at that time was not compatible with magnetic resonance imaging, he underwent computed tomographic (CT) scan of the head and showed no space-occupying lesion in the orbit, paranasal sinuses, and brain. As an old lesion, the anterior temporal lobe on the left side had low-density area with metallic artifact on the left side of the skull base, indicative of metal clipping. In 4 weeks of observation from the initial visit, he showed complete recovery of visual acuity and became capable of abducting the left eye in full degrees. We also reviewed 8 patients with the combination of abducens nerve palsy and optic neuritis in the literature to reveal that the combination of signs did occur in mild meningitis with rare infectious diseases and in association with preceding herpes zoster in the first branch of the trigeminal nerve. The course of the present patient suggested that the combination of signs might be vaccine-associated.
Keywords
Background
Isolated ocular motor nerve palsies including oculomotor, trochlear, and abducens nerve palsy are relatively common in ophthalmic practice.1-3 So-called ischemic palsies caused by microvascular accident of the nerves are most frequent in aged patients who usually have systemic risk factors, such as hypertension, diabetes mellitus, and dyslipidemia. The onset of ischemic palsies is abrupt and the main symptom is sudden diplopia. To make a diagnosis of the ischemic palsies, space-occupying lesions in the paranasal sinuses, orbit, and the brain have to be excluded at first by computed tomographic (CT) scan or magnetic resonance imaging. The abducens, oculomotor, and trochlear nerve palsies have been reported as adverse events for different kinds of vaccines. 4 In the recent pandemics of COVID-19 from the year 2020, vast numbers of people worldwide have undergone the vaccination more than once. In the consequence, the different kinds of adverse events related to the COVID-19 vaccinations have been accumulated in the literature. The adverse events can be roughly classified into two entities: immune-related events and cardiovascular events. In the field of ophthalmology, uveitis 5 and optic neuritis 6 would be developed as the immune-related events while unexpected retinal and vitreous hemorrhages 7 would be presented as the cardiovascular events. Abducens nerve palsy has been indeed reported as sequel to the vaccinations for COVID-19.8-11 and also in association with the infection of COVID-19. 12
In this study, we report a unique patient who developed concurrently the rare combination of abducens nerve palsy and optic neuritis on the same side 2 months after the sixth vaccination for COVID-19. We also reviewed 8 patients with the combination of abducens nerve palsy and optic neuritis in the literature to search for the clinical background.13-20
Case Report
An 82-year-old man had double vision a week previously and then noticed darkness in the vision of his left eye. At the initial visit, he had the left eye that deviated medially at front view and could not abduct the left eye, indicative of abducens nerve palsy on the left side. The eye movements in other directions appeared normal and pupillary light reaction in both eyes appeared minimal due probably to the old age and previous cataract surgeries. The best-corrected visual acuity in decimals was 1.0 (20/20) in the right eye and 0.3 (20/60) in the left eye. The intraocular pressure was 10 mmHg in both eyes. He had intraocular lens implantation in both eyes. Slit-lamp and fundus examinations were normal except for slight blurring of the optic disk on the left side. Optical coherence tomography showed the swollen optic disk on the left side, compared with the right side (Figure 1A and 1B). The macular structure was normal in both eyes.

Fundus photographs and horizontal macular sections of optical coherence tomography on initial visit (A, B), a week later (C, D), 2 weeks later (E, F), and 4 weeks later (G, H) from the initial visit. Right eye in left column (A, C, E, and G) and left eye in right column (B, D, F, and H). Note that the swollen optic disk (arrows) in the left eye has gradually subsided in the time course, compared with the optic disk in the right eye. Small images in the rightmost column show different horizontal slices and also depict the swollen optic disk (stars) on the initial visit and a week later.
Two months prior to the onset of symptoms, he had undergone the sixth inoculation of COVID-19 mRNA vaccine (elasomeran, Moderna). He had 5 prior vaccinations without incident: the first and second inoculation of COVID-19 mRNA vaccine (tozinameran, Pfizer-BioNTech) in the internal of 3 weeks, the third (elasomeran, Moderna), fourth, and fifth inoculation of COVID-19 mRNA vaccine (tozinameran, Pfizer-BioNTech) with each interval of about 6 months. In the past history at the age of 45 years, he experienced subarachnoid hemorrhage and underwent surgery for the clipping of intracranial aneurysm of the internal carotid-posterior communicating artery. At that time, he showed no neurological deficit as complications of subarachnoid hemorrhage and clipping surgery. He had no history of paranasal sinusitis. Currently, he took a combination drug of candesartan 8 mg and amlodipine 5 mg daily for hypertension, tamsulosin 0.2 mg and tadalafil 5 mg daily for dysuria caused by prostate hyperplasia.
The patient had no systemic symptoms, such as general fatigue, fever, arthralgia, and skin rashes. Physical examinations detected no particular findings. Neurological examinations were also unremarkable. Since the aneurysmal metal clip used at that time was not compatible with magnetic resonance imaging, he underwent CT scan of the head to look for any causative lesions for abducens nerve palsy and optic neuritis at the initial visit. No space-occupying lesions were detected in the orbit, paranasal sinuses, and brain. The intracranial hemorrhage was also absent (Figure 2A-2F). As an old lesion, the anterior temporal lobe on the left side had a low-density area of brain atrophy (Figure 2A) with metallic artifact (Figure 2D) on the left side of the skull base, indicative of metal clipping. There were no abnormalities detected in urinalysis and blood examinations including complete blood cell counts, blood glucose, liver, and kidney function tests.

Computed tomographic scans (inferior to superior axial slices in A-F) of the head at the initial visit. Note the old atrophic lesion of low-density area (arrow in A) in the anterior temporal lobe on the left side and metallic artifact (arrow in D) on the left skull base, indicative of metal clip.
In 4 weeks of observation from the initial visit, the best-corrected visual acuity in the left eye returned to 1.0 (20/20) and he became capable of abducting the left eye in almost full degrees from the front. The optic disk swelling depicted by optical coherence tomography in the left eye gradually subsided in the time course of 4 weeks (Figure 1D, 1F, and 1H). The optic disk in the right eye remained normal (Figure 1C, 1E, and 1G). He was also systemically well in the time course and showed no neurological signs in the follow-up of additional 3 months. The differential diagnoses that were listed at the initial presentation for the combination of abducens nerve palsy and optic neuritis on the left side, which after all subsided in a month were intracranial hemorrhage, brain infarction, scarring reaction due to the previous aneurysmal clipping surgery, multiple sclerosis, and vaccine-related complication in order of priority.
Discussion
This patient is unique to develop abducens nerve palsy and probable optic neuritis at the same time on the same side. We chose simple observation since the patient at the old age had no other neurological signs or symptoms. After space-occupying lesions and intracranial hemorrhage were confirmed to be absent by CT scan of the head at the initial visit, a working clinical diagnosis was vaccine-associated abducens nerve palsy and optic neuritis. Oral prednisolone was not given at the initial presentation because of the old age. In 1 month of observation, the visual acuity in the left eye returned to normal and he was capable of abducting in almost full degrees on the left side. The patient did not have systemic symptoms or signs and did not develop other neurological signs in the time course. All these facts suggest that abducens nerve palsy and optic neuritis on the same side in this patient might be related to the preceding vaccination for COVID-19 2 months previously.
The standard treatment strategy for the ischemic ocular motor nerve palsies is the observation to wait for the spontaneous recovery in 1 to 3 months up to 6 months. Abducens nerve palsy that involves only the lateral rectus muscle tends to recover at a higher rate and at a faster speed usually in a month, compared with oculomotor nerve palsy that involves different kinds of extraocular muscles. Trochlear nerve palsy that involves a single muscle of the superior oblique also tends to recover at a fast speed in 1 to 3 months. The complete recovery of abducens nerve palsy in a month in the present patient suggests that the palsy was of microvascular ischemic origin. Since the patient showed complete recovery of abducens nerve palsy and optic neuritis and did not develop other neurological and systemic symptoms in the weekly follow-up of 1 month, we did not perform additional blood tests, such as vitamin B12, autoantibodies, and pituitary hormones to search for other causes. Screening tests to exclude infectious diseases were also not done in the systemically healthy condition of the patient. The follow-up CT to search for the ischemic stroke was not done because the patient did not develop additional neurological symptoms in the course.
A major limitation in this study is that the patient could not be evaluated by magnetic resonance imaging due to the previous surgery with metal clipping of intracranial aneurysm. Furthermore, the aneurysmal clipping appeared to be located on the skull base on the left side that was consistent with the laterality of abducens nerve palsy and optic neuritis in the current presentation. The patient did not experience any neurological symptoms and signs for 37 years since the age of 45 years when he had undergone the surgery for subarachnoid hemorrhage. However, it could not be denied completely that the recent episode of abducens nerve palsy and optic neuritis might be related to this old surgical procedure. The aneurysmal clip might cause vascular or mass effect to develop the combination of abducens nerve palsy and optic neuritis in the present patient.
In our previous study, we reported a patient who developed lymphocytic hypophysitis and diabetes insipidus 1 month after the fourth vaccination for COVID-19 and then developed bilateral optic neuritis 2 months later. 6 In that study, we also reviewed 15 patients with idiopathic hypophysitis and optic neuritis before the pandemics of COVID-19, and noted that 6 of these 15 patients showed recoverable abducens nerve palsy. 6 Abducens nerve might be affected by inflammatory lesions around the pituitary gland in that series of patients. The present patient did not have any symptoms or signs of pituitary dysfunction, and thus, pituitary function tests were not done.
To analyze similar cases, PubMed and Google Scholar were searched for the key words: abducens nerve palsy, optic neuritis, COVID-19, and vaccine. The Japanese literature was searched for the same key words in the bibliographic database of medical literature in Japanese (Igaku Chuo Zasshi, Japana Centra Revuo Medicina, Ichushi-Web), published by the Japan Medical Abstracts Society (JAMAS, Tokyo, Japan). There has been no report regarding the combination of abducens nerve palsy and optic neuritis that would be related to COVID-19 or vaccination against COVID-19. Before the COVID-19 pandemics, the combination of abducens nerve palsy and optic neuritis has been reported as case reports: 8 patients with sufficient description13-20 are summarized, together with the present case, in Table 1.
Review of 9 Patients With Abducens Nerve Palsy and Optic Neuritis in Combination, Including the Present Patient.
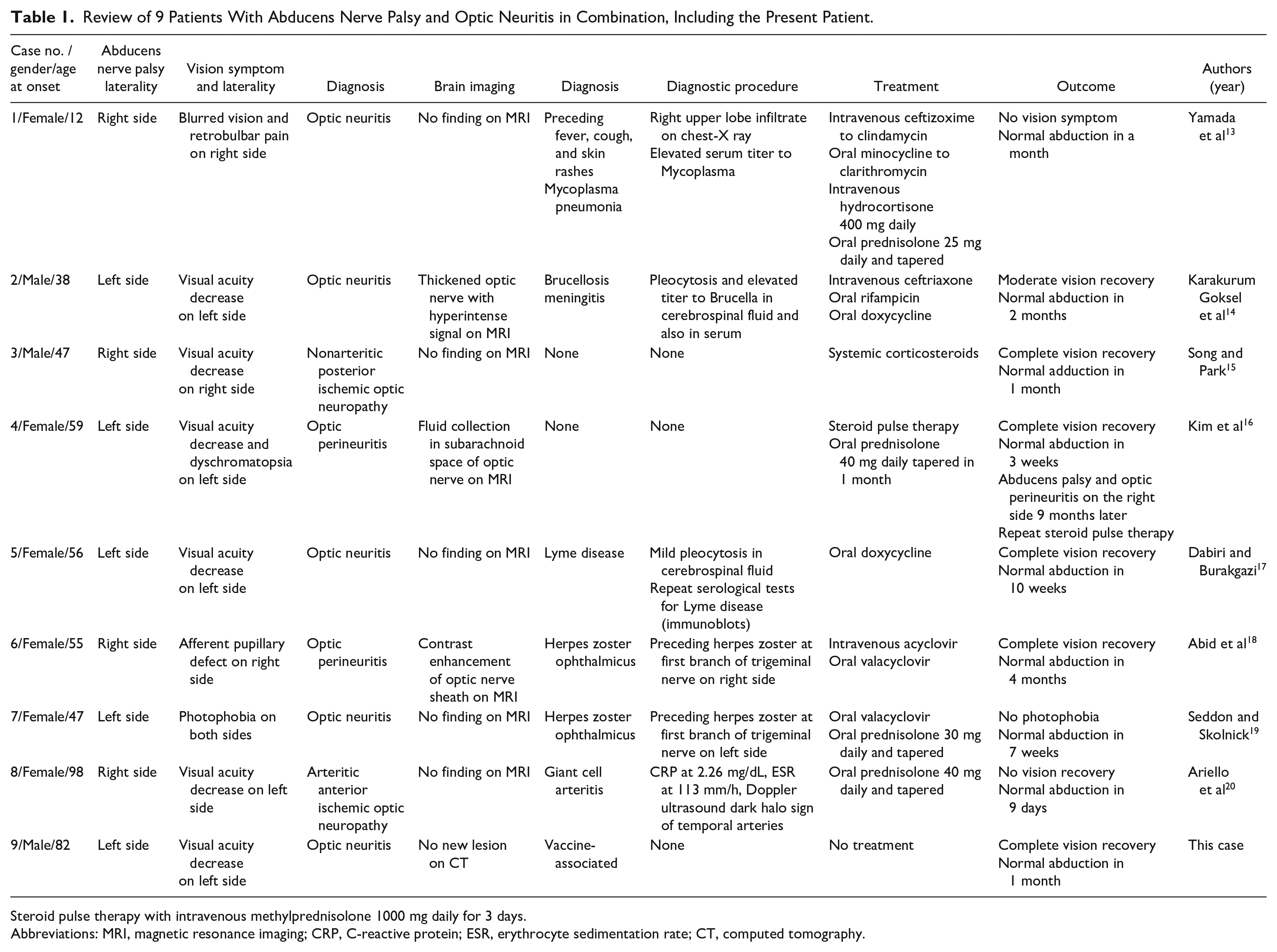
Steroid pulse therapy with intravenous methylprednisolone 1000 mg daily for 3 days.
Abbreviations: MRI, magnetic resonance imaging; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; CT, computed tomography.
The 9 patients including the present patients (Table 1) were 3 men and 6 women with the age at the initial visit ranging from 12 to 98 years (median, 55 years). All patients developed abducens nerve palsy and optic neuritis on the same side, except for one patient (Case 8) with giant cell arteritis who developed abducens nerve palsy and optic neuritis on the different side. Abducens nerve palsy and optic neuritis on the same side in 3 patients were caused probably by meningitis of infectious diseases: mycoplasma (Case 1), brucellosis (Case 2) and Lyme disease (Case 5). Abducens nerve palsy and optic neuritis on the same side in the other pair of 2 patients (Cases 6 and 7) were associated with the preceding onset of ipsilateral herpes zoster and thus were diagnosed as herpes zoster ophthalmicus. All patients except for one (Case 9, the present patient) underwent magnetic resonance imaging: 6 patients showed no particular findings. In contrast, 2 patients showed signs of optic perineuritis, such as subarachnoid fluid collection of the optic nerve (Case 4) and contrast enhancement of optic nerve sheath (Case 6). Another patient (Case 2) showed the thickening of the optic nerve with hyperintense signal on fluid-attenuated inversion recovery (FLAIR).
As for the treatment (Table 1), 3 patients with infectious meningitis had antimicrobials, 2 patients with herpes zoster ophthalmicus had antiherpetics with or without oral corticosteroids, and 3 patients had oral or intravenous corticosteroids. Only 1 patient (Case 9, the present patient) had no treatment and showed the spontaneous recovery. All patients regained the capability of full abduction in 1 to 2 months. Complete vision recovery was noted in 7 patients, moderate recovery in one (Case 2), and no recovery on one (Case 8) with giant cell arteritis. It should be noted that one patient (Case 4) developed the combination of abducens nerve palsy and optic neuritis on the contralateral side 9 months later and had favorable outcome with steroid pulse therapy.
The present patient had no preceding herpes zoster and no signs of meningitis. The other differential diagnoses include multiple sclerosis and Tolosa-Hunt syndrome. Abducens nerve palsy may be a presenting sign of multiple sclerosis. 21 In this context, the combination of abducens nerve palsy and optic neuritis might be signs of multiple sclerosis. The present patient did not show other neurological signs and thus, multiple sclerosis was denied. Tolosa-Hunt syndrome is defined as painful ophthalmoplegia caused by nonspecific inflammation in the superior orbital fissure to the cavernous sinus. 22 The present patient had no pain or headache. Abducens nerve palsy has been also reported in association with viral febrile illness, such as dengue fever 23 and COVID-19 infection. 12 Orbital apex syndrome caused by fungal infection in paranasal sinuses should be also considered in the list of differential diagnoses. 24
After the process of differential diagnosis as mentioned above, the underlying cause for the concurrent onset of abducens nerve palsy and optic neuritis is discussed again in the present patient. The diagnosis of abducens nerve palsy was definite while the evidence for the diagnosis of optic neuritis was somewhat limited. The fundus images were not so demonstrative of optic disk swelling. The evident but small changes on the optical coherence tomographic images were used to diagnose optic neuritis in this patient, together with the visual acuity decrease that showed the recovery in a month. Under the circumstances, it is rather difficult to make the connection of these symptoms and signs to the preceding vaccination unless thorough workup for the large number of other causes would be undertaken. Vaccination-related changes would be possible in this patient but would have to be discussed with equal emphasis on other causes since the patient had the limited evaluation for other typical causes.
In conclusion, the rare combination of abducens nerve palsy and optic neuritis on the same side did occur in mild meningitis with rare infectious diseases and in association with preceding herpes zoster in the first branch of the trigeminal nerve. The course of the present patient illustrated that the combination of abducens nerve palsy and optic neuritis on the same side that showed spontaneous recovery in a month might be vaccine-associated. Given the uncertainties and limitations in this patient who was not fully evaluated due to the old age, a diagnosis with vaccination-related changes should be balanced and treated equally with the other possibilities that are based on the review of the literature.
Footnotes
Author Contributions
T.M., as an ophthalmologist, diagnosed and followed the patient and wrote the manuscript. D.I., as an internist, examined and followed the patient. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethics committee review was not applicable due to the case report design, based on the Ethical Guidelines for Medical and Health Research Involving Human Subjects, issued by the Government of Japan.
Informed Consent
Verbal informed consent was obtained from the patient for his anonymized information to be published in this article.
Data Availability
Data are available upon reasonable request to the corresponding author.
